Preservation of reproductive material in oncology patients: the role of anti-Müllerian hormone and possibilities of Random Start protocols using corifollitropin alfa
Nazarenko T.A., Dzhanashvili L.G., Biryukova A.M., Krylova E.I.
Objective: To perform retrospective assessment of the effectiveness of using corifollitropin alfa according to Random Start protocol for ovarian stimulation regardless of the day or both follicular and luteal phases of the menstrual cycle taking into account anti-Müllerian hormone (AMH) levels in oncology patients.
Materials and methods: Retrospective analysis of medical records of 190 patients with oncologic diseases was performed. The patients were referred by oncologist to the IVF clinic for fertility preservation, where they underwent examination and treatment from 2024 to 2025.
Results: Analysis of clinical data of 160 patients showed that the AMH level is a key factor determining ovarian response to controlled ovarian stimulation. The groups with AMH levels >1.5 ng/ml demonstrated significantly higher number of oocytes 12.7 (5.9) and 11.8 (3.1) versus. 2.9 (1.6) and 3.0 (1.2); (p<001) and blastocysts 3.2 (1.7) and 3.4 (1.8) versus 0.9 (0.4) and 0.9 (0.4); (p<0.01) compared with the groups with AMH levels <1.5 ng ml. The initiation of stimulation in a phase of the cycle did not influence significantly these indicators.
A positive correlation was found between AMH level and the number of antral follicles (r=0.85–0.91), oocytes (r=0.84–0.94), and blastocysts (r=0.76–0.94). The need for additional gonadotropin stimulation and its duration directly depended on the initial ovarian reserve.
Conclusion: Thus, patient stratification based on AMH level helps to optimize the stimulation protocol, whereas the choice of follicular or luteal phase of the cycle for initiation of ovarian stimulation does not significantly influence treatment effectiveness. The use of long-acting gonadotropin (corifollitropin alfa) in ovarian stimulation protocols for oncology patients is of significant practical interest due to the possibility of a single injection against the backdrop of psychological stress related to diagnosis and treatment. However, the effectiveness of this approach, especially in Random Start protocols is understudied. Based on the obtained data, p corifollitropin alfa can be recommended for patients with preserved ovarian reserve during the follicular phase of the cycle. With diminished ovarian reserve, particularly in the luteal phase, the conventional protocol with daily gonadotropin administration can be preferable, since it is more predictable. Our study provides the physicians with substantiated criteria for selection of the optimal stimulation protocol for oncology patients.
Authors' contributions: Nazarenko T.A. – the study concept and design, manuscript editing and final approval; Dzhanashvili L.G., Birukova A.M., Krylova E.I. – material collection, statistical data analysis and processing, manuscript writing.
Conflicts of interest: The authors confirm that they have no conflicts of interest to declare
Funding: The study was carried out without any sponsorship.
Ethical Approval: The study was approved by the local Ethics Committee of Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia.
Patient Consent for Publication: The patients have signed informed consent for publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available on request from the corresponding author after approval from the principal investigator.
For citation: Nazarenko T.A., Dzhanashvili L.G., Birukova A.M., Krylova E.I. Preservation of reproductive material in oncology patients: the role of anti-Müllerian hormone and possibilities of Random Start protocols using corifollitropin alfa.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2026; (4): 149-158 (in Russian)
https://dx.doi.org/10.18565/aig.2025.363
Keywords
In recent years, preservation of reproductive material in young women with cancer has become increasingly important [1, 2]. On the one hand, this is due, to the increased incidence of oncologic diseases among young women, on the other hand, due to significant increase in the survival rate of oncologic patients with preserved reproductive function. This became possible thanks to improved diagnostic methods and timely treatment. However, side effect of cancer treatment, especially chemotherapy and/or radiation therapy, is often premature depletion of ovarian reserve or fertility loss, that is especially important for patients who have not yet realized their reproductive function [3, 4]. In this regard, the issues of timely and optimal approach to preserving fertility have become an integral part of treatment and rehabilitation strategies. This requires active involvement of reproductologists in early diagnosis before starting antitumor therapy and, subsequently, after achieving disease remission.
One of the most effective methods of fertility preservation is mature oocyte or embryo cryopreservation after controlled ovarian stimulation [5–8]. The optimal solution for patients requiring immediate cancer treatment is the use of modified stimulation regimens, in particular, Random Start protocols, that provides initiation of ovarian stimulation regardless of the phase of the menstrual cycle – both in the follicular phase (FP) and in the luteal phase (LP) [9, 10]. These protocols are based on modern concept of multiple waves of follicle development during the menstrual cycle, that make is possible to avoid delay in preparation for stimulation [11, 12].
The flexibility of Random Start protocols ensures sufficient follicle growth and obtaining mature oocytes in a short timeframe, that is critical taking into consideration very limited time before starting cancer treatment [11]. Clinical research demonstrates that the effectiveness of these protocols is comparable with conventional cancer therapies. At the same time, Random Start protocols can be adapted for individualized clinical approach to treatment and rehabilitation of oncologic patients [9]. However, in vitro fertilization (IVF) protocol for this category of patients is associated with a number of clinical and organizational difficulties. So, ovarian stimulation should be completed within 10–14 days from the time of referral. This timeframe coincides with the time period of active diagnostic investigation, psychological distress, and somatic instability [1, 11]. Daily administration of gonadotropins can pose an additional burden, reducing patient adherence to treatment and creating the risk of incomplete compliance with IVF protocol. It is known that the level of anti-Müllerian hormone (AMH) is an important factor in stimulation planning, since AMH is a stable biomarker of ovarian reserve produced by the granulosa cells of preantral and small antral follicles [12].
AMH level does not depend on the phase of the menstrual cycle. For this reason, it is a convenient and reliable tool for urgent evaluation of fertility potential. High AMH levels correlate with a good response to ovarian stimulation, whereas low AMH levels are associated with the risk of low number of mature oocytes [9, 12]. In the context of using Random Start protocols, AMH level is especially important for predicting reproductive outcomes and personalizing treatment, especially in the luteal phase stimulation or in cases of low ovarian reserve [9].
In modern IVF programs, the use of corifollitropin alfa (CFT-α), a long-acting recombinant gonadotropin is of particular interest. This medication provides stable stimulation of folliculogenesis for 7 days after a single injection, that helps significantly reduce the injection burden and improve patient comfort and adherence to treatment [13]. This is especially important for oncologic patients who experience physical and emotional tension. However, despite apparent advantages, there is lack of data in the available literature on the effectiveness of using CFT-α in Random Start protocol, particularly in the luteal phase of the menstrual cycle. However, this regimen may represent a valuable clinical option that could improve the effectiveness and convenience of emergency ovarian stimulation in patients with oncologic diseases.
The purpose of the study was to perform retrospective assessment of the effectiveness of using CFT-α in Random Start protocol for ovarian stimulation in oncologic patients regardless of the follicular or luteal phase of the menstrual cycle taking into account anti-Müllerian hormone (AMH) levels.
Hypotheses:
1) The use of CFT-α for ovulation stimulation, regardless of the phase (FP or LP) of the menstrual cycle allows to attain comparable ovarian response in both phases of the menstrual cycle within Random Start protocols in oncologic patients.
2) The effectiveness of ovulation stimulation within Random Start protocols in oncologic patients depends on AMH levels, regardless of the phase of the menstrual cycle.
Materials and methods
To ensure sample homogeneity, exclude the risks to health and life associated with cancer, improve the effectiveness of evaluation of ovarian stimulation method, and reliability of the study results, the following inclusion criteria were used: age; the presence of oncologic disease; the oncologist’s conclusion about appropriateness of using IVF programs to preserve reproductive material based on the stage of the disease, prognosis for survival based on the time required for treatment.
Inclusion criteria were age of 38 years and higher, diminished ovarian reserve; contraindications associated with worsening of the course of the underlying disease; severe concomitant diseases.
Retrospective analysis of medical records of 190 patients with oncologic diseases was performed. The patients were referred by oncologist to the IVF clinic for fertility preservation (Figure), where they underwent examination and treatment from 2024 to 2025.
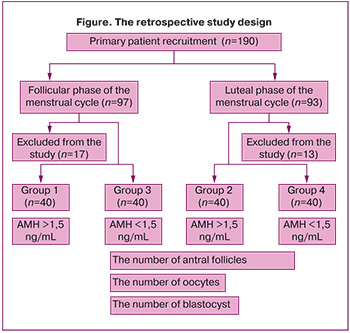
The patients were diagnosed with the following oncologic diseases: Hodgkin lymphoma – 68/190 (35.7%); non-Hodgkin lymphoma – 28/190 (14,7%); breast cancer – 82/190 (43%); colorectal cancer – 12/190 (6,6%).
Ovarian stimulation was perfomed in the follicular phase in 97 patients, and in the luteal phase in 93 patients. After primary analysis of medical records, 17 and 13 women in both groups, respectively, were excluded from further analysis in accordance with the exlusion criteria.
Thus, medical records of 80 women undergoing ovulation stimulation in the FP, and 80 in the LP were distributed in the following 4 groups: group one – medical records of 40 women with AMH level >1.50 ng/mL who underwent stimulation in the FP of the menstrual cycle; group 2 – medical records of 40 women with AMH level >1.50 ng/mL who underwent stimulation in the LP; group 3 –medical records of 40 women with AMH level < 1.50 ng/mLмл who underwent stimulation in the FP; and group 4 – medical records of 40 women with AMH level < 1.50 ng/mL who underwent stimulation in the LP.
Subsequently comprehensive analysis of patient data was performed including age, anthropometric data (height, weight, body mass index), the length of the menstrual cycle, duration of ovarian stimulation, and AMH level.
The effectiveness of ovarian stimulation depending on the AMH level and the phase of the menstrual cycle was evaluated by analysis of the number of antral follicles, oocytes, and blastocysts obtained at this stage of rehabilitation of reproductive function in young women with oncologic diseases.
MH levels were assessed based on AMH test reports submitted by patients upon admission to the IVF clinic. In the absence of test reports, AMH levels were measured using enzyme-linked immune sorbent assay technique. Ovarian condition was evaluated, and antral follicle count was measured by transvaginal ultrasound using the BK Medical device on the day of admission to the IVF clinic.
The quality of obtained oocytes was evaluated by assessing oocyte maturation, and the quality of blastocysts was classified according to Gardner's grading system.
Stimulation protocols
Ovarian stimulation in the follicular phase of the menstrual cycle
On days 2–3 of the menstrual cycle, a single injection of long-acting gonadotropin (CFT-α) at a dose of 150 mcg was used daily. Ultrasound was performed on days 6–7 of the menstrual cycle. In the presence of follicles 14 mm in diameter, gonadotropin-releasing hormone antagonist at a dose of 0.25 mg was additionally administered. Repeat ultrasound was performed on days 9–10 of the menstrual cycle. In the presence of preovulatory follicles 17–19 mm in diameter, the trigger dose of recombinant human chorionic gonadotropin (hCG) (250 mcg) was administered on the same or next day, 36–37 hours prior to transvaginal ovarian puncture. If the size of growing follicles was small, recombinant follicle-stimulating hormone (FSH) was additionally administered at a dose of 150–225 IU per day for 2–3 days to reach the preovulatory follicle size.
Stimulation in the luteal phase of the menstrual cycle
The presence of corpus luteum in the ovary was detected on days 16–18 of the menstrual cycle. A single injection of CFT-α was used at a dose of 150 mcg. After 7 days, repeat ultrasound was performed. If the follicle size was less than 17 mm, recombinant FSH injection at a dose of 150–225 IU was additionally used for 3–4 days. When the follicles reached preovulatory size, the trigger dose of recombinant hCG (250 mcg) was administered 36–37 hours prior to transvaginal ovarian puncture.
Statistical analysis
Nonparametric data are represented as median (Me) and the quartiles [Q1; Q3]. When necessary, nonparametric data are represented as arithmetic mean (M) and standard deviation (SD). The normtest package for choosing the correct statistical test (D' Agostino's K-squared & Pearson’s chi-squared tests, the Anderson-Darling test, Shapiro–Wilk test, Kolmogorov–Smirnov test) was used to assess normality of dataset distribution. Nonparametric tests (the Kruskal–Wallis test and the Dunn's test) for multiple comparisons were used to test the quantitative data with non-normal distribution. The Mann–Whitney test using the Hodges–Lehmann estimator was done for robust estimation of the median of differences between the groups. Two-way ANOVA and the Tukey test for multiple comparisons were used for quantitative data with normal distribution. Correlation analysis was done using the Spearman rank correlation coefficient for the AMH level (alpha=0.05) to be compared with all other parameters. All comparisons were considered significant at p<0.05.
Results
The age of patients was 26 – 37 years. The median age was 31.1. (2.9), 31.1 (3.1), 32.4 (3.3) and 32,5 (2.8) years in groups 1 – 4, respectively (Table 1). Nonparametric tests for multiple comparisons (the Kruskal–Wallis and the Dunn’s tests) found not significant differences both in general and separate comparisons between the groups (p=NS) with regard to the age and anthropometric parameters (Table 1). Also, there was no significant difference in the length of the menstrual cycle between the groups (p=NS).

Medical records of 17 and 13 patients in the FP and LP of the menstrual cycle in each group, respectively, were excluded from analysis. Further, medical records of 40 patients in each group were analyzed – AMH levels in group 1 (FP) >1.5 ng/mL; in group 2 (LP) >1.5 ng/Ml; in group 3 (FP) <1.5 ng/mL; in group 4 (LP) <1.5 ng/mL.
Analysis found significant difference (p<0.01) between all groups in the duration of ovarian stimulation using the Kruskal–Wallis test. Similarly, significant difference (p<0.01) in the duration of ovarian stimulation was found between groups 1 and 3, 1 and 4, 2 and 3, as well 3 and 4 using the Dunn’s post hoc-test for multiple comparisons. At the same time, no statistically significant differences were found between groups 1 and 2, as well as 3 and 4 in the duration of ovarian stimulation (p=NS).
The median AMH level was 4.7 (1.9), 4.6 (1.7), 1.3 (0.2) and 1.2 (0.2) ng/mL in groups 1, 2, 3, 4, respectively. Comparative analysis of AMH levels using the Kruskal–Wallis test found significant difference (p<0.01) between the 4 groups. Further analysis using the Dunn’s test for multiple comparisons showed no differences between groups 1 and 2, as well as 3 and 4 (р=NS). However, comparison of AMH levels between groups 1,2 and 3,4 (р<0.01) showed significant difference.
In 80 women with high AMH levels, the number of antral follicles visible on ultrasound ranged from 3 to 25; the median antral follicle count (AFC) was 14.4 (6.3) and 13.5 (6.9), respectively, in groups 1 and 3 (Table 2). At the same time, in 80 women with low levels of AMH, from 1 to 6 antral follicles were detected; the median AFCr was 4 (1.4) and 3.9 (1.7), respectively, in groups 2 and 4. The number of oocytes depended on AMH level. So, in women with high AMH levels, up to 23 oocytes and in women with low AMH levels, up to 7 oocytes were detected. At the same time the median number of oocytes was 12.7 (5.9), 3.3 (1.8), 2.8 (1.6) and 3 (1.2) oocytes in groups 1, 2, 3, 4, respectively.

Similarly, up to 6–7 blastocysts were found in groups with high AMH levels, whereas single blastocysts or their absence were found in groups with low AMH levels. The median number of blastocysts was 3.2 (1.7), 3.3 (1.8), 0.9 (0.3) and 0.8 (0.4) in group1 1, 2, 3, 4, respectively
Statistical analysis of the number of antral follicles, oocytes and blastocysts using the Kruskal–Wallis test found significant differences (р<0.01) in all explored parameters between the four groups (Table 2). Further analysis using the Dunn's test for multiple comparisons showed absence of differences between the groups 1 and 2, as well as between 3 and 4 (p=NS) in all explored parameters (the number of antral follicles, oocytes and blastocysts). Comparison of the number of antral follicles, oocytes and blastocysts showed significant differences (p<0.01) between the groups 1, 2 and 3, 4.
Subsequently, robust estimation of the median of differences between the groups in AMH levels, the number of antral follicles and oocytes, as well as blastocysts was done using the Mann–Whitney test and the Hodges–Lehmann estimator (Table 3). No significant difference was found between the groups 1 and 2, as well as 3 and 4 in the ovarian parameters (Table 3). Comparison of other parameters showed significant differences between the 4 groups (p<0.01).
At the same time, large negative shifts were found (-11.0 and -12.0) between groups 1 and 3, 4, as well as between groups 2 and 3, 4 in the number of antral follicles and oocytes. However, moderate negative shifts (-2.0 and -4.15) were found between groups 1 and 3, 4, as well as between groups 2 and 3, 4 in AMH levels and the number of blastocysts. Absence of shift (0) or low shift (from -0.02 to -1.0) was noted between groups 1 and 2, as well as between groups 3 and 4 in the absence of significant differences between the compared groups using the Mann–Whitney test (p=NS).
Given significant impact of age and AMH on all aspects of women's reproductive function, correlation was identified between AMH concentrations, age, body mass index, the length of the menstrual cycle, and the outcome of ovulation stimulation – the number of oocytes and blastocysts (Table 4).
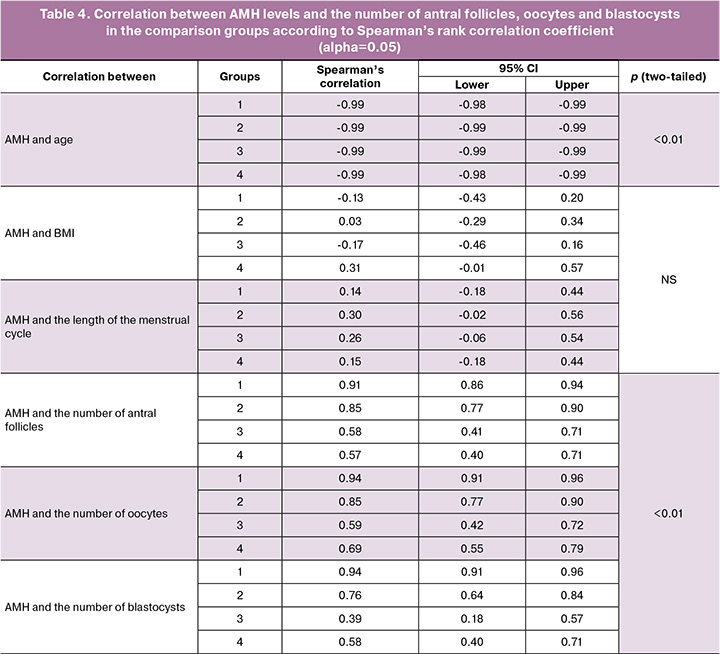
Strong negative correlation between age and AMH levels (-0.98/-0.99) was found in all groups, both in the FP and LP of ovarian stimulation. Weak positive correlation was found only in group 4 between AMH and BMI, whereas there was none in groups 1, 2, and 3. Similarly, no correlation was found between AMH and the length of the menstrual cycle. According to the Spearman rank correlation coefficient, strong positive correlation was found between AMH and the number of antral follicles in groups 1 and 2 (0.91 and 0.85), and moderate positive correlation in groups 3 and 4 (0.58 and 0.57). Similarly, strong positive correlation was found between AMH and the number of oocytes in groups 1 and 2 (0.94 and 0.85), and moderate correlation in groups 3 and 4 (0.59 and 0.69). Also, strong positive correlation was found between AMH level and the number of blastocysts in groups 1 and 2 (0.94 and 0.76). Weak and moderate positive correlation was found in groups 3 and 4 (0.39 and 0.58).
Analysis of characteristics of ovarian stimulation protocols (Table 5) showed that for patients with a high ovarian reserve (AMH = 4.7 (1.7) ng/mL) one injection of CFT-α was sufficient for ovarian stimulation in the FP to obtain the optimal number of mature oocytes – 16 (2.1). In 76% of patients with normal ovarian reserve (AMH = 2.9 (0.9) ng/mL), gonadotropins at a dose of 150–225 IU/l were used for 1–2 days. As a result, 11.7 (2.8) mature oocytes were obtained. With low ovarian reserve (AMH = 1.7 (0.8) ng/ml), additional gonadotropin injections at a dose of 150–225 IU/l for ovarian stimulation in the follicular phase were required for all patients for 3–4 days. As a result, 5.9 (2.4) mature oocytes were obtained.
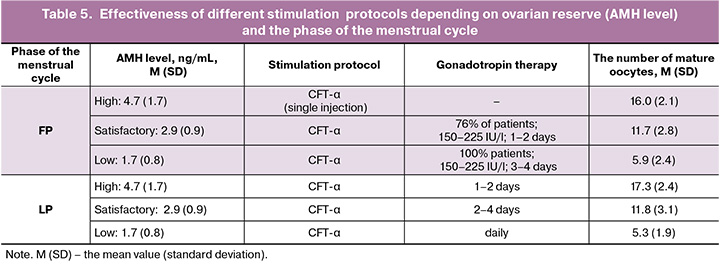
For ovarian stimulation in the luteal phase of the menstrual cycle, all patients additionally received gonadotropin injections. The outcomes were the following: with high ovarian reserve 17.3 (2.4) mature oocytes were obtained within 1–2 days. With normal ovarian reserve, 11.8 (3.1) mature oocytes were obtained within 2–4 days. The use of long-acting gonadotropin in women with low ovarian reserve did not lead to sufficient stimulation of ovarian function. For this reason, daily gonadotropin injections were indicated. As a result, 5.3 (1.9) mature oocytes were obtained.
Discussion
A single injection of long-lasting gonadotropin CFT-α can replace daily FSH injections for 7 days of ovarian stimulation. This effect is achieved due to the unique medication structure. The formula of CFT-α consists of the α-subunit of FSH and carboxy-terminal peptide, a chimeric structure synthesized on the basis of the β-subunit of hCG. The unique chemical structure of this medication ensures a long biological half-life – 69 hours, while that of FSH is approximately 34 hours, that provides prolonged ovarian stimulation by FSH for 7 days with a single injection of the hormone. It should be noted that, despite the presence of modified hCG molecule, CFT-α acts as FSH and does not bind to LH receptors [14]. Three large studies: Engage, Ensure and Trust [15–17] assessed the tolerability, the effectiveness, methods of medication administration, the features of folliculogenesis, steroid genesis and oogenesis of CFT-α in the protocol with gonadotropin-releasing hormone antagonist. It was concluded that the effectiveness of CFT-α is equivalent to recombinant FSH in achieving successful pregnancies, and has a simplified treatment regimen with fewer injections. However, the effectiveness of CFT-α was explored in the patient group that included young women aged 18–36 years, who had good ovarian reserve, tuboperitoneal factor of infertility, and a history of no more than two IVF attempts. The evidence of the effectiveness of CFT-α in complex clinical situations has not been defined. At the same time, long-acting gonadotropin is of interest for evaluation of the feasibility of ovarian stimulation for fertility preservation in cancer patients. The advantage of this protocol is obvious. A single injection can help avoid daily gonadotropin injections and multiple clinic visits of patients undergoing evaluation with the diagnosed disease and experiencing acute stress. However, despite the obvious advantages, there is no data in published papers on the effectiveness of using CFT-α in Random Start protocols, particularly for ovarian stimulation in the luteal phase of the menstrual cycle. The purpose of our study was to assess the feasibility of using corifollitropin alfa in Random Start protocols for ovarian stimulation in cancer patients.
Analysis of the study results showed that there were was no significant difference between the groups in patients' age, anthropometric parameters, and the length of the menstrual cycle, that indicates homogeneity of the patient population in these parameters. Further comparative analysis of AMH levels, duration of ovarian stimulation by using CFT-α, and the number of antral follicles, oocytes, and blastocysts showed identical patterns in using different statistical approaches (the Kruskal–Wallis test, the Dunn's test for multiple comparisons, and the Mann–Whitney test using the Hodges–Lehmann estimator). Moreover, no significant differences were found between groups 1 and 2, as well as between groups 3 and 4, virtually with 0 shift between the compared parameters. Moderate shift (-2.0 to - 4.15) was found between groups 1 and 3–4, as well as between groups 2 and 3-4, in AMH levels and the number of blastocysts. At the same time, significant negative shift (-11.0 and -12.0) was found between the groups 1 and 3, 4, as well as between groups 2 and 3, 4, in the number of antral follicles and oocytes. In general, these results support our hypothesis that “the use of CFT-α for ovulation stimulation regardless of phase (FF or LF) of the menstrual cycle phase helps to obtain comparable ovarian response in both phases of the menstrual cycle within Random Start protocols in cancer patients”. Furthermore, based on these data, as well as correlation analysis of AMH with other parameters, our second hypothesis is confirmed that “the effectiveness of ovulation stimulation within Random Start protocols in cancer patients depends on AMH levels regardless of the phase of the menstrual cycle phase”.
Conclusion
The results of the study showed that treatment effectiveness is determined by patients’ status of ovarian reserve, in particular, AMH levels. For ovarian stimulation in the follicular phase in patients with high and normal ovarian reserve, one injection of CFT-α was sufficient for ovarian stimulation, and additional administration of gonadotropin for no more than 2 days in patients with normal ovarian reserve, that resulted in obtaining the optimal number of mature oocytes. In patients with poor ovarian reserve, administration of long-lasting gonadotropin in the follicular cycle was less effective, and gonadotropin was additionally administered for 4 days. In these cases, it is necessary to take into account the patient’s characteristics in choosing the most appropriate protocol for ovarian stimulation. Indication of long-lasting gonadotropin cannot be excluded. However, daily injections can be a more predictive regimen for ovarian stimulation. For ovarian stimulation in the luteal phase, daily gonadotropins were indicated additionally for all patients. However, in women with high AMH levels administration of gonadotropins was indicated for 1–2 days. This enables us to consider this stimulation regimen to be effective. In cases of normal ovarian reserve, additional gonadotropin administration lasted 3–4 days, that has no obvious advantages over daily injections. In such cases, it is necessary to assess the patient's characteristics. In women with low ovarian reserve, ovarian stimulation by using long-lasting gonadotropin in the luteal phase of the menstrual cycle is not recommended.
Therefore, the doctors should have a strong sense of responsibility in performing ovarian stimulation for cryopreservation of reproductive material in cancer patients, that necessitates selecting the most effective treatment protocol. The results of our study will help the doctors in using long-lasting gonadotropin for ovarian stimulation in cancer patients.
References
- Su H.I., Lacchetti C., Letourneau J., Partridge A.H., Qamar R., Quinn G.P. et al. Fertility preservation in people with cancer: ASCO guideline update. J. Clin. Oncol. 2025; 43(12): 1488-515. https://dx.doi.org/10.1200/JCO-24-02782
- Sonmezer M., Sacinti K.G., Oktay K.H. Female fertility preservation: 25 years of progress, expanding indications and future prospects. Hum. Reprod. Update. 2025; 32(2): 231-59. https://dx.doi.org/10.1093/humupd/dmaf026
- Gotschel F., Sonigo C., Becquart C., Sellami I., Mayeur A., Grynberg M. New insights on in vitro maturation of oocytes for fertility preservation. Int. J. Mol. Sci. 2024; 25(19): 10605. https://dx.doi.org/10.3390/ijms251910605
- Takae S., Harada M., Nakamura K., Furuyama S., Ono M., Osuga Y. et al. Reproductive outcomes of embryo cryopreservation and transfer at the start-up phase of fertility preservation in Japan. Reprod. Med. Biol. 2024; 23(1): e12581. https://dx.doi.org/10.1002/rmb2.12581
- Буняева Е.С., Кириллова А.О., Назаренко Т.А., Джанашвили Л.Г., Гаджимагомедова К.К., Хабас Г.Н., Бирюкова А.М., Гависова А.А. Показания и эффективность технологии получения незрелых ооцит-кумулюсных комплексов из ткани яичника с последующим их дозреванием in vitro. Акушерство и гинекология. 2022; 6: 75-82. [Bunyaeva E.S., Kirillova A.O., Nazarenko T.A., Dzhanashvili L.G., Gadzhimagomedova K.K., Khabas G.N., Biryukova A.M., Gavisova A.A. Indications and effectiveness of technique for immature oocyte-cumulus complexes retrieval from ovarian tissue followed by their in vitro maturation. Obstetrics and Gynecology. 2022; (6): 75-82 (in Russian)]. https://dx.doi.org/10.18565/aig.2022.6.75-82
- Chen C.N., Chang L.T., Chen C.H., Tam K.W. Fertility preservation for women with breast cancer before chemotherapy: a systematic review and meta-analysis. Reprod. Biomed. Online. 2022; 44(2): 357-69. https://dx.doi.org/10.1016/j.rbmo.2021.08.003
- Nazarenko T.A., Martirosyan Y.O., Birukova A.M., Korneeva I.E., Sokolova J.V., Khubaeva D.G. Outcomes of ovarian stimulation in the follicular and luteal phases of the menstrual cycle in cancer patients. Gynecol. Endocrinol. 2021; 37(sup1): 13-16. https://dx.doi.org/10.1080/09513590.2021.2006458
- Turan V., Bedoschi G., Lee D.Y., Barbosa C.P., de Oliveira R., Sacinti K.G. et al. Trends and regional differences for fertility preservation procedures in women with breast cancer. Clin. Breast Cancer. 2025; 25(2): 108-16.e1. https://dx.doi.org/10.1016/j.clbc.2024.09.011
- Sönmezer M., Şükür Y.E., Ateş C., Saçıntı K.G., Sönmezer M., Aslan B. et al. Random start ovarian stimulation before gonadotoxic therapies in women with cancer: a systematic review and meta-analysis. Reprod. Biomed. Online. 2023; 47(6): 103337. https://dx.doi.org/10.1016/j.rbmo.2023.103337
- Сыркашева А.Г., Доброхотова Ю.Э., Гохберг Я.А., Трошина М.Н., Сорокин Ю.А., Лапина И.А. In vitro maturation как стратегия сохранения фертильности у пациенток с онкологическими заболеваниями. Акушерство и гинекология. 2025; 10: 83-9. [Syrkasheva A.G., Dobrokhotova Yu.E., Gokhberg Ya.A., Troshina M.N., Sorokin Yu.A., Lapina I.A. In vitro maturation as a fertility preservation strategy in cancer patients. Obstetrics and Gynecology. 2025; (10): 83-9 (in Russian)]. https://dx.doi.org/10.18565/aig.2025.210
- Назаренко Т.А., Мартиросян Я.О., Бирюкова А.М., Джанашвили Л.Г., Иванец Т.Ю., Сухова Ю.В. Опыт стимуляции яичников в режиме «random-start» протоколов для сохранения репродуктивного материала онкологических больных. Акушерство и гинекология. 2020; 4: 52-8. [ Nazarenko T.A., Martirosyan Ya.O., Biryukova A.M., Dzhanashvili L.G., Ivanets T.Yu., Sukhova Yu.V. Experience in random-start ovarian stimulation for preserving reproductive material of cancer patients. Obstetrics and Gynecology. 2020; (4): 52-8 (in Russian)]. https://dx.doi.org/10.18565/aig.2020.4.52-58
- Panattoni A., Montt Guevara M.M., Marzi I., Saçıntı K.G., Papini F., Maggiorano C. et al. Effectiveness of controlled ovarian stimulation for oocyte preservation in oncologic patients: insights from DuoStim protocol. J. Clin. Med. 2025; 14(22): 8062. https://dx.doi.org/10.3390/jcm14228062
- García-Velasco J.A., Fernández-Sánchez M., Larsson P., Mannaerts B. A single dose of corifollitropin alfa is equipotent to a daily dose of 300 IU recombinant FSH regardless of ovarian reserve. Reprod. Biomed. Online. 2025; 51(3): 104987. https://dx.doi.org/10.1016/j.rbmo.2025.104987
- LaPolt P.S., Nishimori K., Fares F.A., Perlas E., Boime I., Hsueh A.J. Enhanced stimulation of follicle maturation and ovulatory potential by long acting follicle-stimulating hormone agonists with extended carboxyl-terminal peptides. Endocrinology. 1992; 131(6): 2514-20. https://dx.doi.org/10.1210/endo.131.6.1446593
- Devroey P., Boostanfar R., Koper N.P., Mannaerts B.M., Ijzerman-Boon P.C., Fauser B.C. A double-blind, non-inferiority RCT comparing corifollitropin alfa and recombinant FSH during the first seven days of ovarian stimulation using a GnRH antagonist protocol. Hum. Reprod. 2009; 24(12): 3063-72. https://dx.doi.org/10.1093/humrep/dep291
- Corifollitropin alfa Ensure Study Group. Corifollitropin alfa for ovarian stimulation in IVF: a randomized trial in lower-body-weight women. Reprod. Biomed. Online. 2010; 21(1): 66-76. https://dx.doi.org/10.1016/j.rbmo.2010.03.019
- Norman R.J., Zegers-Hochschild F., Salle B.S., Elbers J., Heijnen E., Marintcheva-Petrova M. et al. Repeated ovarian stimulation with corifollitropin alfa in patients in a GnRH antagonist protocol: no concern for immunogenicity. Hum. Reprod. 2011; 26(8): 2200-8. https://dx.doi.org/10.1093/humrep/der163
Received 09.12.2025
Accepted 09.04.2026
About the Authors
Tatyana A. Nazarenko, Dr. Med. Sci., Professor, Head of the Institute of Reproductive Medicine, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of the Russian Federation, 117997, Russia, Moscow, Ac. Oparin str., 4, t_nazarenko@oparina4.ru,https://orcid.org/0000-0002-5823-1667
Lana G. Dzhanashvili, PhD, obstetrician-gynecologist at the Scientific and Clinical Department of ART named after F. Paulsen, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of the Russian Federation, l_dzhanashvili@oparina4.ru, 117997, Russia, Moscow,
Ac. Oparin str., 4, https://orcid.org/0000-0002-2891-3974
Almina M. Birukova, PhD, Head on Clinical Work at the Scientific and Clinical Department of ART named after F. Paulsen, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of the Russian Federation, a_birukova@oparina4.ru, 117997, Russia, Moscow,
Ac. Oparin str., 4, https://orcid.org/0009-0004-5113-6239
Ekaterina I. Krylova, obstetrician-gynecologist at the Scientific and Clinical Department of ART named after F. Paulsen, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of the Russian Federation, kr.katrin00@gmail.com, 117997, Russia, Moscow, Ac. Oparin str., 4. https://orcid.org/0000-0002-0220-0474



