Anti-Müllerian hormone as an indicator of phenotypes in women of reproductive age with polycystic ovary syndrome in the Siberian population
Artymuk N.V., Elgina S.I., Beglova A.Yu.
Objective: To define reference ranges for serum anti-Müllerian hormone (AMH) in patients of reproductive age with polycystic ovary syndrome (PCOS), the residents of the Kemerovo region (in southwestern Siberia) and to perform subsequent differentiation between PCOS phenotypes. Comparative analysis of the obtained AMH levels and assessment of their diagnostic significance for verification of PCOS phenotypes was performed.
Materials and methods: AMH levels were evaluated in 400 women aged 18 to 35 – in 200 with confirmed diagnosis of PCOS (Group I) and in 200 without PCOS (Group II). After determining the frequency of PCOS phenotypes in women of reproductive age in the Siberian population, 4 subgroups were formed in Group I: Group I (A) – women with PCOS phenotype A (classical), Group I (B) – phenotype B (anovulatory), Group I (C) – phenotype C (ovulatory), and Group I (D) – phenotype D (non-androgenic). Anti-Müllerian hormone levels were measured for all participants of the study.
Results: The classical phenotype A (53%) and anovular phenotype B (27%) prevailed in women of reproductive age with PCOS in the Siberian population. Serum AMH levels in women with PCOS were significantly higher compared with healthy women. However, not all PCOS phenotypes were characterized by elevated AMH levels. Analysis showed extremely high AMH levels in phenotypes A (classical) and D (non-androgenic), that significantly exceeded AMH levels both in healthy women in the control group and patient population with PCOS. In contrast, AMH levels in B (anovulatory) and C (ovulatory) phenotypes were comparable with those in women without PCOS, and the difference between them was not statistically significant. The results of our study indicate feasibility of defining AMH levels to diagnose PCOS depending on the phenotype.
Conclusion: The practical outcome of our study is that it suggests phenotype-depended diagnostic role of AMH. This universal marker can be informative for all types of PCOS. However, its diagnostic accuracy varies and demonstrates maximum sensitivity in phenotypes C and D, and specificity in phenotypes A, В and D.
Authors' contributions: Artymuk N.V. – manuscript editing; Elgina S.I.– the study concept and design, manuscript writing; Beglova A.Yu. – material collection and processing, the study concept and design, manuscript writing.
Conflicts of interest: The authors confirm that they have no known competing financial interests or personal relationships that could be considered as potential competing interests and could have appeared to influence the work reported in this paper.
Funding: The study was supported by author own support.
Ethical Approval: The study was approved by the local Ethics Committee of Kemerovo State Medical University, Ministry of Health of Russia.
Patient Consent for Publication: The patients have signed informed consent for publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available on request from the corresponding author after approval from the principal investigator.
For citation: Artymuk N.V., Elgina S.I., Beglova A.Yu. Anti-Müllerian hormone as an indicator of phenotypes in
women of reproductive age with polycystic ovary syndrome in the Siberian population.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2026; (2): 104-110 (in Russian)
https://dx.doi.org/10.18565/aig.2025.262
Keywords
Polycystic ovary syndrome (PCOS) is one of the most common pathologies of the endocrine system in women of reproductive age.
Manifestations of PCOS can be observed for a long period of time – from puberty to postmenopause [1]. Despite the global prevalence of PCOS, affecting approximately 11–13% of the female population, the etiology and pathogenesis of PCOS remain the subject of research [2].
The key diagnostic criteria for PCOS are menstrual disorders (oligomenorrhea or amenorrhea), the clinical and/or biochemical signs of androgen excess, as well as ultrasound signs of polycystic ovarian morphology. According to the latest international recommendations, establishment of the diagnosis of PCOS requires the presence of at least two of the three of the mentioned criteria after excluding other endocrine diseases [2, 3]. These include: the signs of hyperandrogenism (clinically manifested or confirmed by laboratory tests), impaired ovulatory function, as well as specific morphology of the ovaries which is detected by ultrasound, or increased anti-Müllerian hormone (AMH) level [2].
Accordingly, the experts of the National Institutes of Health support the Rotterdam consensus on diagnostic criteria for PCOS (2003, amended in 2012), although insist on the importance of an individual approach. They propose to distinguish one of the four main PCOS phenotypes in patients: A (classical PCOS), B (anovulatory PCOS), C ovulatory (PCOS), and D (non-androgenic PCOS) [4].
The Russian medical literature also focuses on the need for phenotypic characterization of PCOS. However, the practical value of such classification for clinical decision making requires further study [5].
The incidence of PCOS varies depending on the diagnostic criteria used. However, on a global scale it demonstrates relative uniformity. At the same time, the epidemiological studies reported rising prevalence of PCOS in populations in Southeast Asia and the Eastern Mediterranean region [5].
The distribution of phenotypic variations in PCOS is also very different. Statistical data on the frequency of PCOS phenotypes are based mainly on information from medical facilities and show a specific picture in different countries. For example, phenotype A is most common in the United States. It is diagnosed in 48.0% of patients followed by phenotype B (25.0–27.0%), phenotype C (18.0–25.0%) and phenotype D (3.0–8.0%). The most common phenotype among the Indian women with PCOS is phenotype A. It reaches 56%. The distribution of other phenotypes in India is different: phenotype (C) is identified in 11.0% of women, phenotype (D) in 18.0%, and phenotype (B) in 1.0% [6].
The study by Lazareva L.M. et al. was devoted to exploration of the prevalence of PCOS phenotypes in Russia, especially in the Eastern Siberian and in the Republic of Buryatia. According to the data obtained by them, the most common phenotype is classical phenotype A, followed by C (ovulatory PCOS), B (anovulatory PCOS) and D (non-androgenic PCOS) [7]. At the same time, correlation was established between obesity, insulin resistance, cardiometabolic disorders, hyperandrogenism in patients and increased frequency of detection of phenotypes A and B [8, 9].
Latest research data confirm the influence of race and ethnicity on manifestation of PCOS phenotypes and concomitant metabolic disorders [10]. VanHise K. et al. reported that the prevalence and/or intensity of hirsutism in women in the Middle East, the Mediterranean, India and South Asia is higher compared with indigenous people of East Asia or Europe. In addition, this group is characterized by increased risk of developing metabolic syndrome, whereas Hispanic and Mexican women are most often diagnosed with insulin resistance, and African American women with arterial hypertension compared with white women [10].
The studies clearly confirm that the patients with PCOS have significantly increased serum AMH levels [11].
In modern clinical practice, the AMH level is proved to be a reliable laboratory indicator of PCOS and is a functional alternative to counting of antral follicles by ultrasound in women of reproductive age [12, 13]. Moreover, Anti- Müllerian hormone test is gradually replacing ultrasound diagnostics of polycystic ovarian morphology. Current international guidelines allow the use of any of these methods to detect polycystic transformation in adult patients, but emphasize the unacceptability of their simultaneous application to avoid overdiagnosis [1].
At the same time, diagnostic and prognostic value of AMH as well as other PCOS criteria apparently are different and depend on the geographical region and ethnicity. It is important to note that many ethnic groups, including Asian, Native American, Native Alaskan, Native Hawaiian, and Pacific Islander women, have been underrepresented in previous epidemiological studies on PCOS [5, 6, 10]
Our study is relevant due to the lack of data on the population frequency of PCOS phenotypes and AMH levels in different PCOS phenotypes in female residents of Western Siberia. Of particular scientific interest is assessment of AMH concentrations depending on the clinical type of PCOS in this population.
The objective of the study was to define reference ranges for serum AMH in patients of reproductive age with PCOS, the residents of the Kemerovo region (in Southwestern Siberia), to perform subsequent differentiation between PCOS phenotypes, and to do comparative analysis of the obtained AMH levels and assessment of their diagnostic significance for verification of PCOS phenotypes.
Materials and methods
The study was carried out at Women’s Health Clinic No. 1 at the premises of Kemerovo City Polyclinic No. 5 named after L.I. Temerkhanova from 2015 to 2021 Appoval for the study was obtained from the local Ethics Committee of Kemerovo State Medical University of the Ministry of Health of Russia (protocol No. 15/k of January 2015).
The study design is represented in Structure Chart (Fig. 1)
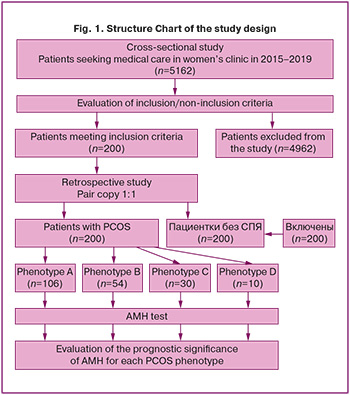
The study included two stages.
Stage I. One time screening test was performed in the study cohort, which was defined among 5962 who sought medical care due to complaints about PCOS symptoms (oligomenorrhea or amenorrhea, infertility, hirsutism) and underwent a full scope of medical examination following the protocol according to the clinical recommendations (2021).
Stage II. Retrospective case-control study was conducted. The main group included women aged 18–35 years with the diagnosis of PCOS, the native-born residents of the Kemerovo region (Kuzbass), who had signed informed consent for publication of their data. The diagnosis of PCOS was made based on at least two out of the three following criteria:
- Ovulatory dysfunction (menstrual cycle <21/>35 days, <8 cycles per year, progesterone level <3 ng/mL).
- Biochemical or clinical hyperandrogenism (hirsutism score ≥4–6 or testosterone level >1.7 nmol/L).
- Ultrasound signs of polycystic ovary syndrome (≥20 follicles 2–9 mm in diameter and/or ovarian volume ≥10 cm³ in the absence of dominant structures) [5].
Criteria for formation of the control group: the control group consisted of women aged 18–35 years without PCOS, the residents of the Kemerovo region (Kuzbass), who had signed informed consent for publication of their data.
Exclusion criteria for all women, participating in the study: age below 18 years and over 35 years, the presence of endocrinopathies (hyperprolactinemia, hypothyroidism, adrenal pathology), pelvic surgery in history, intake of estrogen- progestogen drugs for therapeutic or contraceptive purposes, as well as refusal to participate in the study.
Based on the above criteria, the main group was formed (group I). It included 200 patients. Based on pair copy, the control group (group II) was formed. It consisted of 200 women who had no PCOS.
Laboratory diagnostics: Serum AMH levels were measured. Venous blood samples (5 ml) were collected in the morning from the cubital vein. Blood samples were collected from women with regular menstrual cycle on days 2–3 of the cycle, and from women with oligomenorrhea or amenorrhea on a random day. In total, blood samples were collected from 400 participants.
Solid phase enzyme-linked immunosorbent assay with AMH Quantitative Test Kit (Beckman Coulter, Inc., USA) was used for the quantitative determination of AMH.
Lehr's formula was used to calculate the sample size.
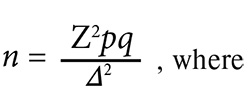
n – the sample size;
Z – the coefficient, the critical value for 95% confidence interval = 1.96;
p – the number of respondents with PCOS;
q=1-p – the number of respondents without PCOS;
∆ – the standardized difference.
The study included 200 patients who were selected among 13 283 women aged 18–35 years, and were at the dispensary registration at Women’s Health Clinic No. 1 at the premises of Kemerovo City Polyclinic No. 5 named after L.I. Temerkhanova. Sample size calculation was done with 95% confidence interval and 5% margin of error, that was representative for the explored pathology, the prevalence of which among women of reproductive age reaches 8–21%.
The main group (group I) was formed of patients with PCOS. It was divided into 4 subgroups based on PCOS phenotype:
IА (classical PCOS) – hyperandrogenism, oligo-/anovulation and polycystic ovarian morphology;
IВ (anovulatory PCOS) – hyperandrogenism and oligo-/anovulation;
IС (ovulatory PCOS) – hyperandrogenism and polycystic ovaries with preserved ovulation;
ID (non-androgenic PCOS) – oligo-/anovulation and polycystic ovaries in the absence of hyperandrogenism.
In the final phase, the prognostic significance of AMH for each phenotype was evaluated.
Statistical analysis
Statistical data processing was carried out using Microsoft Excel 2013 and IBM SPSS Statistics V.24. The Shapiro–Wilk test showed that the data deviated from a normal distribution. Therefore, the results are represented as the median (Me) and interquartile range (the 25th–75th percentiles).
AMH levels between the groups were compared using nonparametric methods – the Kruskal–Wallis test and subsequent pairwise comparison using the Mann–Whitney U test with Bonferroni correction.
The prognostic value of AMH was assessed using regression analysis and receiver operating characteristic (ROC) curves. The diagnostic performance of the model was determined by the area under the ROC curve (AUC), sensitivity, specificity, and accuracy.
Results and discussion
The participants of the study were age-homogenous. The mean age of patients with PCOS in group I was 28.2 years (the interquartile range: 25.9–30.5), whereas 28.6 years in the control group II (the interquartile range: 26.6–30.3). The age difference between the groups was not statistically significant (р=0.92).
Distribution of PCOS phenotypes showed that phenotype A was most common in female residents of Western Siberia, and was identified in more than half of women – 53.0% (106/200). Other phenotypes were identified less common: В – in 27.0% (54/200), С (ovulatory PCOS) – in 15.0% (30/200) and D (non-androgenic PCOS) – in 5.0% of patients (10/200).
Comparative analysis of AMH levels in the study groups is represented in the Table.
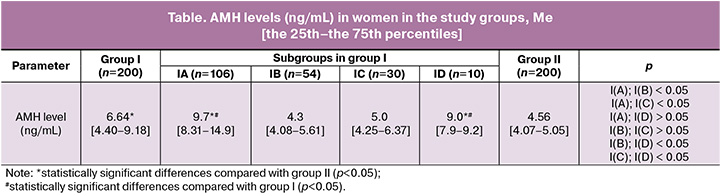
Women with PCOS had significantly higher AMH levels compared with healthy women. However, not all PCOS phenotypes were characterized by elevated AMH levels. Extremely high AMH levels were in phenotype A (classical PCOS) and phenotype D (non-androgenic PCOS), and significantly exceeded AMH levels not only in the control group of healthy women, but also in the general patient population with PCOS. However, AMH levels in phenotype B (anovulatory PCOS) and phenotype C (ovulatory PCOS) were comparable with those in women without PCOS and did not show statistically significant increase.
Given a wide reference range of AMH levels during the reproductive years, as well as the lack of clearly established standard threshold for AMH levels in women with PCOS, we determined cut-off values of AMH as diagnostic criteria of PCOS using ROC analysis (Fig. 2).
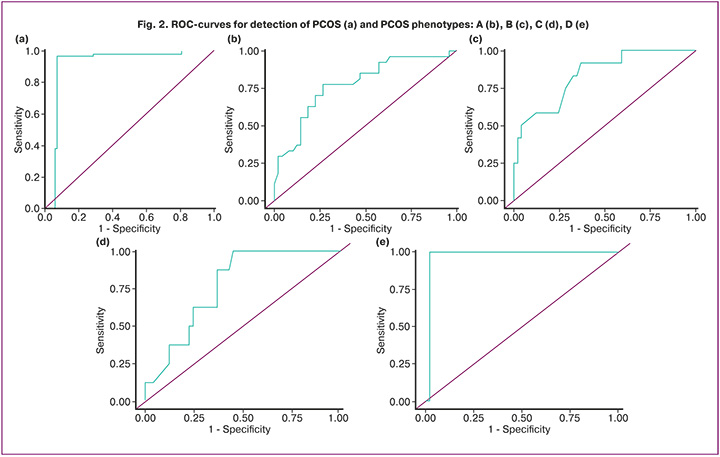
The diagnostic threshold of AMH level in women with PCOS was 5.2 ng/mL (sensitivity – 87.7%, specificity – 66.7%, accuracy – 77.9%, AUC=0.912±0.027 [0.859; 0.966], p<0.001). This diagnostic threshold can help make differential diagnosis in controversial cases between women with PCOS and without PCOS.
Figure 2 (b), (c), (d), (e) shows ROC curves of AMH levels in different PCOS phenotypes.
The diagnostic thresholds of AMH level in women with PCOS were the following: 6.2 ng/mL (sensitivity – 77.8%, specificity – 73.5, 7.5%, accuracy – 77.9%, AUC=0.779±0.059 [0.664; 0.895], р<0.001) in phenotype A; 3.06 ng/mL (sensitivity – 63.3%, specificity – 73.5%, accuracy – 83.8%, AUC=0.8383±0,.75 [0.690; 0.985], p<0.001) in phenotype B; 3.18 ng/mL (sensitivity – 100.0%, specificity – 55.1%, accuracy – 76.9%, AUC=0.769±0.120 [0.568; 0.970], р=0.015) in phenotype С; and 5.9 ng/mL, (sensitivity – 100.0%, specificity – 98.0%, accuracy – 98.0%, AUC=0.980±0.050 [0.881; 1.00], р=0.002) in phenotype D.
The represented diagnostic test thresholds of AMH level enable differential diagnosis between phenotypes in patients with and without PCOS.
For these diagnostic tests the area under the ROC curve (AUC) was >0.75, and according to expert assessment this model can be rated as a good model. The prognostic significance of AMH demonstrated high efficiency in diagnosis of PCOS including the clinical phenotypes associated with PCOS in patients of reproductive age. The highest diagnostic sensitivity was observed in phenotypes C and D, while maximum specificity was in phenotypes A, B, and D.
Discussion
Among different PCOS phenotypes in women in the Kemerovo region (Southwestern Siberia) phenotype A (classical PCOS) prevailed, that is consistent with the epidemiological data from other regions of Russia and other countries of the world. Distribution of other phenotypes is similar with distribution of those in Western Siberian but significantly differ in populations in the Western Hemisphere and South Asia. This variability is presumably due to a combination of factors, including geographical location, racial and ethnic characteristics of populations [10].
Multiple studies confirm that serum AMH concentrations in patients with PCOS are significantly higher compared with women without this endocrine disorder [1, 5, 11, 14, 15, 16].
Therefore, determination of AMH level is recognized as a promising diagnostic tool for PCOS verification. Current clinical guidelines recommend to use this marker either as a standalone diagnostic test or as an alternative to ultrasound for diagnosing polycystic ovarian morphology [1, 5, 12, 13].
Research data confirm the heterogeneity in AMH concentration depending on the PCOS phenotype. Patients with phenotype A have elevated AMH levels, that is confirmed in some studies [13, 17]. Comparative dynamics of AMH levels between phenotypes B, C, and D remains controversial. According to some data, moderate AMH levels are in carriers of phenotype C, which are lower than those in phenotype A, but can differ from AMH levels in B and D in the absence of hyperandrogenism [18]. In contrast, another study demonstrates statistically significant elevation in AMH levels in patients with phenotype D, both in comparison with healthy women and in relation to phenotype B [19]. All PCOS phenotypes are characterized by elevated AMH levels compared with those in the control group [13, 17, 20, 21].
The variability can probably be explained in the underexplored mechanisms of excessive AMH secretion in PCOS. Increased AMH production is considered as a marker of corticosteroid dysregulation, that contributes to anovulation along with hyperandrogenism, excess of luteinizing hormone, or hyperinsulinemia. Furthermore, genetic factors can play a significant role. In particular, heterozygous mutations in the AMH gene which reduce its biological activity have been found in some women with PCOS. Carriers of these mutations tend to have lower serum AMH levels, although gonadotropin and testosterone levels are similar in women with PCOS and other patients [22, 23].
Our analysis found statistically higher values in serum AMH levels in women of reproductive age with PCOS, the residents of the Kemerovo region (Southwestern Siberia) compared with the control group without PCOS. The key observation is that phenotypes A and D are associated with significantly higher AMH levels versus phenotypes B and C.
Conclusion
The distribution of PCOS phenotypes in the region showed predominance of phenotypes A (classical PCOS) and B (anovulatory PCOS). The practical implications of this study suggest phenotype-dependent diagnostic role of AMH. This marker can be universally informative in diagnosing all types of PCOS. However, its diagnostic accuracy varies and demonstrates maximum sensitivity in phenotypes C and D, and specificity in phenotypes A, В and D.
References
- Teede H.J., Tay C.T., Laven J.J.E., Dokras A., Moran L.J., Piltonen T.T. et al. Recommendations from the 2023 international evidence-based guideline for the assessment and management of polycystic ovary syndrome. J. Clin Endocrinol Metab. 2023; 108(10): 2447-69. https://dx.doi.org/10.1210/clinem/dgad463
- Stener-Victorin E., Teede H., Norman R.J., Legro R., Goodarzi M.O., Dokras A. et al. Polycystic ovary syndrome. Nat. Rev. Dis. Primers. 2024; 10(1): 27. https://dx.doi.org/10.1038/s41572-024-00511-3
- Neven A.C.H., Laven J., Teede H.J., Boyle J.A. A summary on polycystic ovary syndrome: diagnostic criteria, prevalence, clinical manifestations, and management according to the latest international guidelines. Semin. Reprod. Med. 2018; 36(1): 5-12. https://dx.doi.org/10.1055/s-0038-1668085
- National Institutes of Health. Evidence-based Methodology Workshop on Polycystic Ovary Syndrome. December 3-5, 2012. Executive summary. Available at: https://www.physicsforums.com/attachments/finalreport-pdf.112236/
- Министерство здравоохранения Российской Федерации. Клинические рекомендации. Синдром поликистозных яичников. 2021. [Ministry of Health of the Russian Federation. Clinical guidelines. Polycystic ovary syndrome. 2021 (in Russian)].
- Bozdag G., Mumusoglu S., Zengin D., Karabulut E., Yildiz B.O. The prevalence and phenotypic features of polycystic ovary syndrome: a systematic review and meta-analysis. Hum. Reprod. 2016; 31(12): 2841-55. https://dx.doi.org/10.1093/humrep/dew218
- Лазарева Л.М., Шарифулин Э.М., Беленькая Л.В., Сутурина Л.В. СПКЯ в репродуктивном возрасте: фенотипическое разнообразие и диагностические подходы (обзор литературы). Доктор.Ру. 2020; 19(6): 50-6. [Lazareva L.M., Sharifulin E.M., Belenkaya L.V., Suturina L.V. Polycystic ovary syndrome in women of reproductive age: phenotypic variety and diagnostic approaches (review of literature). Doctor.Ru. 2020; 19(6): 50-6 (in Russian)]. https://dx.doi.org/10.31550/1727-2378-2020-19-6-50-56
- Mayer S.B., Evans W.S., Nestler J.E. Polycystic ovary syndrome and insulin: our understanding in the past, present and future. Women's Health (Lond). 2015; 11(2): 137-49. https://dx.doi.org/10.2217/whe.14.73
- Li L., Feng Q., Ye M., He Y., Yao A., Shi K. Metabolic effect of obesity on polycystic ovary syndrome in adolescents: a meta-analysis. J. Obstet. Gynaecol. 2017; 37(8): 1036-47. https://dx.doi.org/10.1080/01443615.2017.1318840
- VanHise K., Wang E.T., Norris K., Azziz R., Pisarska M.D., Chan J.L. Racial and ethnic disparities in polycystic ovary syndrome. Fertil. Steril. 2023; 119(3):348-54. https://dx.doi.org/10.1016/j.fertnstert.2023.01.031
- Pellatt L., Hanna L., Brincat M., Galea R., Brain H., Whitehead S. et al. Granulosa cell production of anti-Müllerian hormone is increased in polycystic ovaries. J. Clin. Endocrinol. Metab. 2007; 92(1): 240-5. https://dx.doi.org/10.1210/jc.2006-1582
- Bani Mohammad M., Majdi Seghinsara A. Polycystic ovary syndrome (PCOS), diagnostic criteria, and AMH. Asian Pac. J. Cancer Prev. 2017; 18(1): 17-21. https://dx.doi.org/10.22034/APJCP.2017.18.1.17
- Fraissinet A., Robin G., Pigny P., Lefebvre T., Catteau-Jonard S., Dewailly D. Use of the serum anti-Müllerian hormone assay as a surrogate for polycystic ovarian morphology: impact on diagnosis and phenotypic classification of polycystic ovary syndrome. Hum. Reprod. 2017; 32(8): 1716-22. https://dx.doi.org/10.1093/humrep/dex239
- Yin W.W., Huang C.C., Chen Y.R., Yu D.Q., Jin M., Feng C. The effect of medication on serum anti-müllerian hormone (AMH) levels in women of reproductive age: a meta-analysis. BMC Endocr. Disord. 2022; 22(1): 158. https://dx.doi.org/10.1186/s12902-022-01065-9
- Zhou Z., Chen H., Chu L., Zou Q., Chen Q., Yang J. et al. The effects of metformin on anti-Müllerian hormone levels in patients with polycystic ovary syndrome: a systematic review and meta-analysis. J. Ovarian. Res. 2023; 16(1): 123. https://dx.doi.org/10.1186/s13048-023-01195-1
- Sivanandy M.S., Ha S.K. The role of serum anti-mullerian hormone measurement in the diagnosis of polycystic ovary syndrome. Diagnostics (Basel). 2023; 13(5): 907. https://dx.doi.org/10.3390/diagnostics13050907
- Belenkaia L.V., Lazareva L.M., Walker W., Lizneva D.V., Suturina L.V. Criteria, phenotypes and prevalence of polycystic ovary syndrome. Minerva Ginecol. 2019; 71(3): 211-23. https://dx.doi.org/10.23736/S0026-4784.19.04404-6
- Гафарова Е.А. Клинико-лабораторные проявления СПКЯ в зависимости от фенотипических особенностей и уровня витамина Д в крови. Практическая медицина. 2016; 1(93): 81-4. [Gafarova E.A. Clinical and laboratory manifestations of PCOS depending on phenotypic characteristics and vitamin D blood level. Practical Medicine. 2016; 1(93): 81-4 (in Russian)].
- Абашова Е.И., Шалина М.А., Мишарина Е.В., Ткаченко Н.Н., Булгакова О.Л. Клинические особенности фенотипов синдрома поликистозных яичников у женщин с нормогонадотропной ановуляцией в репродуктивном возрасте. Журнал акушерства и женских болезней. 2019; 68(3): 7-14. [Abashova E.I., Shalina M.A., Misharina E.V., Tkachenko N.N., Bulgakova O.L. Clinical features of polycystic ovary syndrome phenotypes in women with normogonadotropic anovulation in reproductive age. Journal of Obstetrics and Women’s Diseases. 2019; 68(3): 7-14 (in Russian)]. https://dx.doi.org/10.17816/JOWD6837-14
- Romualdi D., Di Florio C., Tagliaferri V., De Cicco S., Gagliano D., Immediata V. et al. The role of anti-müllerian hormone in the characterization of the different polycystic ovary syndrome phenotypes. Reprod. Sci. 2016; 23(5): 655-61. https://dx.doi.org/10.1177/1933719115611751
- Хащенко Е.П., Уварова Е.В., Иванец Т.Ю., Мамедова Ф.Ш., Высоких М.Ю., Боровиков П.И., Балашов И.С. Ключевые критерии диагностики синдрома поликистозных яичников у подростков с учетом уточненных возрастных нормативов гормонального и метаболического статуса. Акушерство и гинекология. 2019; 9: 102-10. [Khashchenko E.P., Uvarova E.V., Ivanets T.Yu., Mamedova F.Sh., Vysokikh M.Yu., Borovikov P.I., Balashov I.S. Key diagnostic criteria for adolescent polycystic ovary syndrome adjusted for age-specific hormonal and metabolic status standards. Obstetrics and Gynecology. 2019; (9): 102-10 (in Russian)]. https://dx.doi.org/10.18565/aig.2019.9.102-110
- Hoyos L.R., Visser J.A., McLuskey A., Chazenbalk G.D., Grogan T.R., Dumesic D.A. Loss of anti-Müllerian hormone (AMH) immunoactivity due to a homozygous AMH gene variant rs10417628 in a woman with classical polycystic ovary syndrome (PCOS). Hum. Reprod. 2020; 35(10): 2294-302. https://dx.doi.org/10.1093/humrep/deaa199
- Moolhuijsen L.M.E., Louwers Y.V., McLuskey A., Broer L., Uitterlinden A.G., Verdiesen R.M.G. et al. Association between an AMH promoter polymorphism and serum AMH levels in PCOS patients. Hum. Reprod. 2022; 37(7): 1544-56. https://dx.doi.org/10.1093/humrep/deac082
Received 22.09.2025
Accepted 29.01.2026
About the Authors
Natalia V. Artymuk, Dr. Med. Sci., Professor, Head of the Department of Obstetrics and Gynecology named after Professor G.A. Ushakova, Kemerovo State Medical University, Ministry of Health of Russia, 650029, Russia, Kemerovo, Voroshilova str., 22a, artymuk@gmail.com, https://orcid.org/0000-0001-7014-6492Svetlana I. Elgina, Dr. Med. Sci., Professor at the Department of Obstetrics and Gynecology named after G.A. Ushakova, Kemerovo State Medical University, Ministry of Health of Russia, 650029, Russia, Kemerovo, Voroshilova str., 22a, +7(905)065-16-13, Elginas.i@mail.ru, https://orcid.org/0000-0002-6966-2681
Angelika Yu. Beglova, obstetrician-gynecologist, Kuzbass Clinical Oncological Dispensary named after M.S. Rappoport, 650029, Russia, Kemerovo, Voroshilova str., 22a, angelik-1986@mail.ru, https://orcid.org/0000-0001-5574-4275
Corresponding author: Svetlana I. Elgina, elginas.i@mail.ru



