Effect of antibiotic prophylaxis during hysteroresectoscopy on the recurrence of endometrial polyps
Vanakova A.I., Dolgushina N.V., Denisov P.A., Goncharuk O.D., Muravieva V.V., Priputnevich T.V.
Background: Endometrial polyps (EP) are the most common type of benign intrauterine pathology and are characterized by a high risk of recurrence. One of the possible etiological factors for EPs is microbial infection. Administration of antibacterial drugs during hysteroresectoscopy (HRS) may reduce the frequency of EP recurrence.
Objective: To study the effect of antibiotic prophylaxis during hysteroresectoscopy for EPs on their recurrence rate.
Materials and methods: A prospective parallel-group study included 84 patients with EP confirmed by histological examination who were randomized into two groups based on the administration of antibiotic prophylaxis during HRS. Endometrial samples were obtained before the HRS. The composition of the endometrial microbiota was analyzed using culturomics with an extended set of selective and non-selective nutrient media. First-generation cephalosporins were administered intravenously in a single dose. The recurrence rate of EP was assessed 12–18 months after HRS.
Results: Chronic endometritis (CE) was associated with EP recurrence within 12–18 months after HRS. The overall recurrence rate was 25%, which was 2.8 times higher in the presence of CE (OR=2.8, 95% CI=0.9–8.4). Bacterial flora growth in the uterine cavity was observed in 40/84 (47.6%) patients with EP and in all 23 (100%) patients with CE. The predominant species were microorganisms of the Staphylococcus genus, which were detected 22 times more frequently in patients with CE (OR=22.3, 95% CI=7.3–68.4). Antibiotic prophylaxis during HRS did not reduce the overall recurrence rate of EP, but it did reduce the recurrence rate by five and seven times, respectively, in patients with CE and in those with microbial growth in the uterine cavity in addition to EP.
Conclusion: These findings confirm the role of microbial factors in the genesis of CE and associated EP, as well as the effectiveness of antibiotics in reducing the recurrence rate in patients with CE and bacterial microflora growth in the uterine cavity.
Authors' contributions: Dolgushina N.V. – conception and design of the study; Vanakova A.I. – material collection, drafting of the manuscript; Goncharuk O.D., Muravieva V.V. – laboratory part of the study; Denisov P.A. – statistical analysis, visualization; Muravieva V.V., Priputnevich T.V. – editing of the manuscript.
Conflicts of interest: The authors have no conflicts of interest to declare.
Funding: There was no funding for this study.
Ethical Approval: The study was reviewed and approved by the Research Ethics Committee of the I.M. Sechenov First MSMU, Ministry of Health of Russia (Sechenov University) (Ref. No: 04-23 of 02.03.2023).
Patient Consent for Publication: All patients provided informed consent for the publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available upon request from the corresponding author after approval from the principal investigator.
For citation: Vanakova A.I., Dolgushina N.V., Denisov P.A., Goncharuk O.D., Muravieva V.V., Priputnevich T.V.
Effect of antibiotic prophylaxis during hysteroresectoscopy on the recurrence of endometrial polyps.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2025; (2): 59-67 (in Russian)
https://dx.doi.org/10.18565/aig.2024.337
Keywords
Endometrial polyps (EP) are among the most prevalent forms of benign intrauterine pathology, with a prevalence ranging from 6 to 27% [1, 2]. They significantly affect the reproductive function [3] and quality of life [4] of women. Despite the introduction of new surgical treatments, the recurrence rate of EPs remains high, varying between 13% and 43%, depending on the follow-up duration and surgical technique employed [5–7]. This elevated risk of EP recurrence often necessitates repeated surgical interventions, which in turn increases the likelihood of developing intrauterine synechiae and infertility [8].
A potential etiological factor in the development of EPs is a microbial infection [9]. However, there is no consensus on the need for antibiotic (AB) prescriptions in cases of EP. According to the national clinical guidelines "Endometrial Polyps", routine administration of ABs is not recommended to mitigate the risk of recurrent EP [10]. This recommendation is supported by several studies demonstrating the ineffectiveness of AB prescriptions in patients with EP and chronic endometritis (CE) concerning recovery from polypectomy and subsequent pregnancy [11, 12]. Conversely, some studies suggest the appropriateness of prescribing ABs for the surgical treatment of EP [13], highlight an increased risk of EP recurrence in the presence of CE [14, 15], and indicate enhanced pregnancy rates with antibiotic therapy for CE in women with infertility [16] and multiple implantation failures [17]. Therefore, research evaluating the role of microbial factors in EP recurrence and the efficacy of ABs in reducing recurrence incidence is highly relevant.
This study aimed to investigate the impact of antibiotic prophylaxis during hysteroresectoscopy for EPs on their recurrence rate.
Materials and methods
The study was conducted at the Department of Innovative Oncology and Gynecology at V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, from 2022 to 2023. This prospective parallel-group study involved 84 patients with EPs who were randomized into two groups based on the administration of AB prophylaxis (1st generation cephalosporins intravenously once) during HRS: group 1 received AB prophylaxis (n=42), while group 2 did not (n=42). Randomization was performed using a simple number method.
The inclusion criteria were being aged 18 years or older and pre-menopausal, providing informed voluntary consent for participation in the study, and having histologically confirmed EP.
Exclusion criteria included cancer, endometriosis or adenomyosis stage 3-4; submucosal uterine myoma or intramural uterine myoma with centripetal growth, acute inflammatory diseases, infectious diseases, and the use of antibacterial or hormonal medications within three months prior to study inclusion. All patients underwent pre-hospitalization examinations according to the clinical guideline "Endometrial Polyps". Given the correlation between the phase of the menstrual cycle and the microbial composition of the endometrium [18–21], materials were collected during the first phase of the menstrual cycle.
To investigate the microbiota of the cervical canal before hysteroscopy, we sampled the cervical canal contents using a sterile bacteriological Dacron swab, which was placed into a tube containing Amies transport medium (Copan, Italy). To prevent contamination of the uterine cavity contents by microflora from the lower genitalia, we treated different loci sequentially with antiseptic: first, we removed mucus from the cervix with a sterile swab and applied antiseptic; after collecting cervical canal secretions for examination, we treated the cervical canal twice with a bacteriologic swab moistened with antiseptic, with a 5-minute interval between treatments. Without prior dilation of the cervical canal, a surgical hysteroscope tube was inserted transcervically via the internal cervical os into the uterine cavity. Surgical forceps were inserted into the surgical canal and the material was collected during the first attempt. The material was then retrieved from the forceps using a sterile needle, placed in a special container, and delivered to the laboratory.
To isolate facultative anaerobic microorganisms, we employed a combination of universal and selective nutrient media: Colombian agar, chocolate agar, mannitol-salt agar (Sonda, Spain), Endo medium, and Sabouraud agar (FGUN "GICPM&B", Obolensk, Russia). Lactobacilli were cultured on Lactobacagar medium (FSUN "GICPM&B", Obolensk, Russia), while strict anaerobes were cultured on Schaedler pre-reduced agar (Sonda, Spain) with the necessary additives and Anaerob Basal Agar (Oxoid, UK). Strict anaerobes were grown in an anaerobic box (Whitley DG 250 Anaerobic Workstation, UK) in a three-component gas mixture (N2, 80%; CO2, 10%; H2, 10%) for 48 h. Species identification was performed using time-of-flight mass spectrometry (MALDI-TOF MS) on a MicroFlex mass spectrometer with the MALDI BioTyper software (Bruker Daltonics, Germany), version 5.0. The antibiotic sensitivity of the isolated microorganisms was assessed, which revealed that 100% of the cases were sensitive to cephalosporins.
A comprehensive morphological analysis of endometrial samples was conducted to interpret the morphological features according to established criteria. Following the standard procedure, the samples were embedded in paraffin, and the deparaffinized sections (4 μm thick) were stained with hematoxylin and eosin, as well as picrofuchsin by Van-Gieson. The diagnosis of CE was based on the following morphological criteria: the presence of plasma cells, identified by immunohistochemical study for CD138 (clone B-A38, Cell Marque) on the plasma cell membrane; inflammatory infiltrates characterized by "lymphoid follicles" in the basal layer and throughout the functional layer of the mucosa; stromal fibrosis; and sclerosis of spiral arteries. Diagnosis requires a combination of at least three of these criteria, with the obligatory presence of plasma cells [22].
The EP recurrence rate was evaluated 12–18 months after HRS based on the pelvic ultrasound examination data. The main criteria were: 1) the presence of a structure in the uterine cavity with increased echogenicity measuring 5 mm or more, and 2) the presence of a vascular pedicle.
Statistical analysis
Statistical analysis and visualization were conducted using OriginLab Pro 2021 (version 9.8.0.200; OriginLab Corporation, USA) and Statistica 10 (USA). The Kolmogorov–Smirnov test was used to assess the normality of the distribution. In cases of non-normal data distribution, the median with interquartile range (Me (Q25; Q75)) and Mann–Whitney test were used. Fractions (%) were calculated to evaluate qualitative data. The χ² test was used to compare categorical data and assess the significance of the differences. To compare binary data, the odds ratio (OR) with a 95% confidence interval (95% CI) was determined using logistic regression. The Margalef and Menchinick indices were used to calculate species richness, whereas the Simpson and Shannon indices were used for taxonomic diversity. The endpoints of the study included the OR of EP recurrence according to the presence of CE and the EP recurrence rate according to the prescription of AB prophylaxis. Differences between statistical values were considered significant at p<0.05.
Results
The median age of the patients was 38.5 years and their body mass index (BMI) was 21.7 kg/m2. A total of 33/84 (39%) patients had a history of polypectomies. The patients in the two groups did not differ significantly in any of the evaluated clinical and medical history data (Table 1).
The main complaints of patients were menorrhagia, metrorrhagia, and infertility (Table 2). Menorrhagia occurred more often in patients with large polyps (r=0.22, p=0.04). Infertility was more frequently diagnosed in the presence of a polypectomy history (p=0.02).
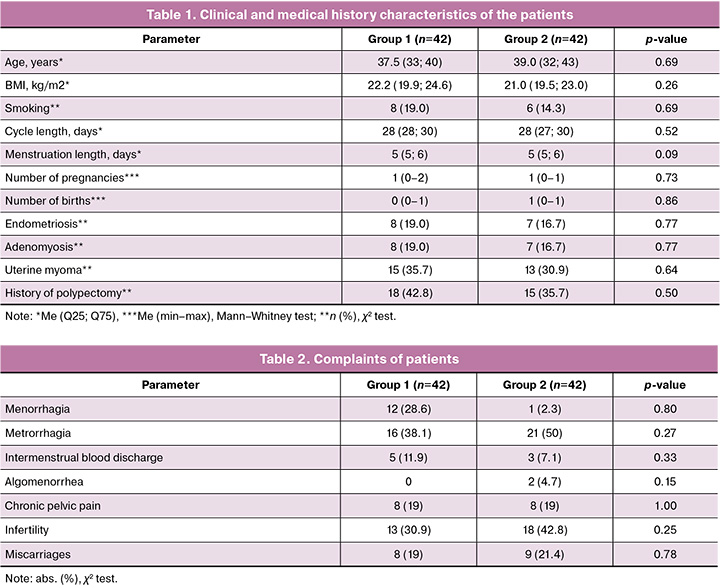
Bacterial microflora growth in the uterine cavity was observed in 40/84 (47.6%) patients. Twenty species of microorganisms belonging to nine genera were identified. The most frequently detected microorganisms were microorganisms of genus Staphylococcus (in 20/40 (50%) patients) and microorganisms of genus Lactobacillus (in 15/40 (37.5%) patients) (Fig. 1).
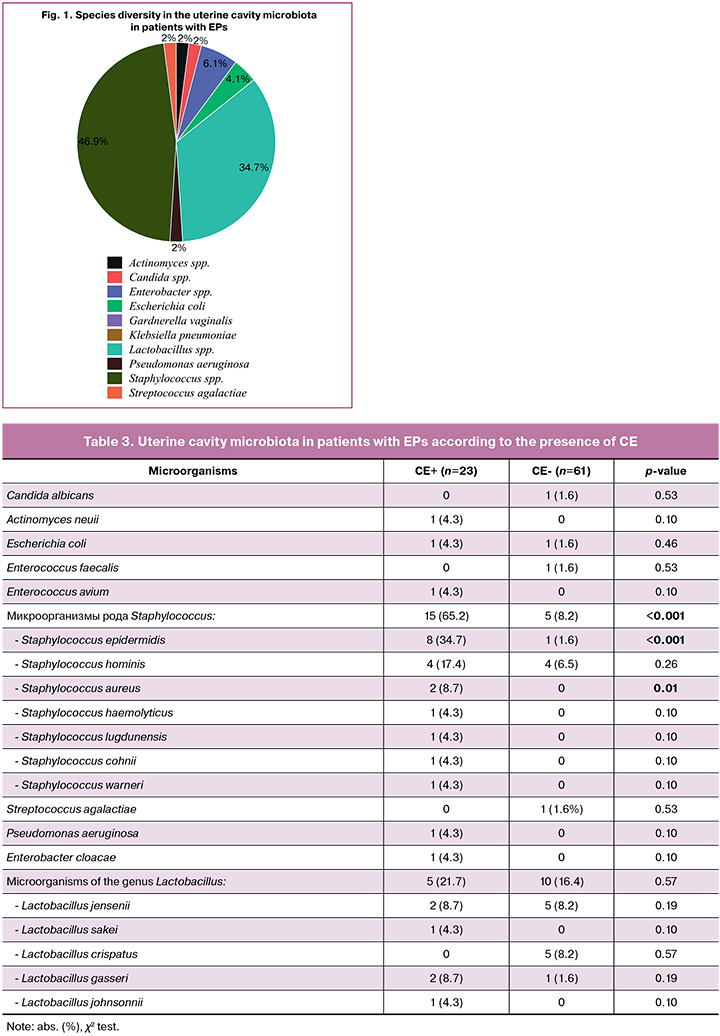
CE, according to a histological report, was diagnosed in 23/84 (27.4%) patients with EPs. All patients with CE showed an increase in microflora in the uterine cavity. When comparing the features of the uterine cavity microbiota in patients with EPs with and without CE, it was found that Staphylococcus microorganisms were significantly more frequently detected in patients with CE (p<0.001, OR=22.3, 95% CI 7.3–68.4), including Staphylococcus aureus (p=0.01) (Table 3).
The recurrence rate of EPs after 12–18 months in all patients was analyzed according to the prescription of AB prophylaxis, taking into account the presence of CE and uterine microbiota. Overall, there was no significant difference in the EP recurrence rate between groups 1 and 2 (8/42 (19%) and 13/42 (31%), respectively; p=0.21) (Fig. 2).
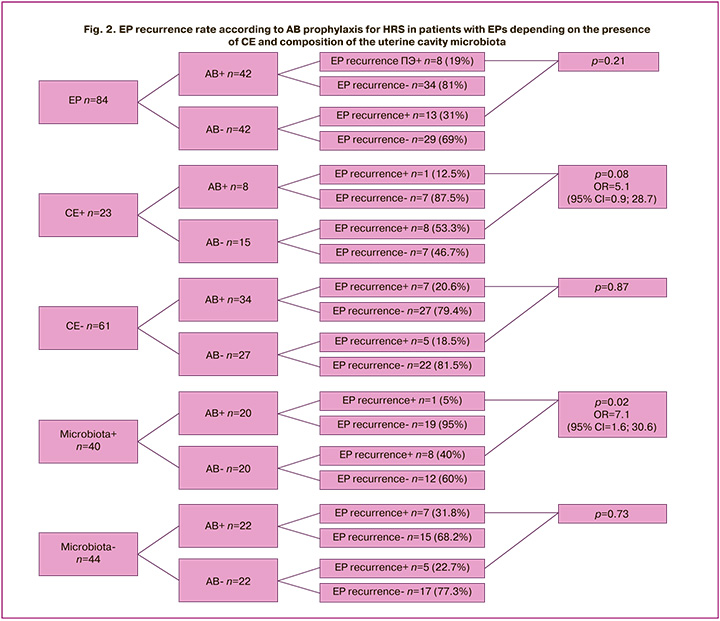
When stratified into subgroups according to the presence of CE, it was found that the recurrence rate did not differ in the group without CE, regardless of the prescription of AB prophylaxis (7/34 (20.6%) and 5/27 (18.5%), respectively), whereas it was five times higher in the group with CE in the absence of AB prophylaxis: 1/8 (12.5%) in the AB group and 8/15 (53.3%) in the group without AB (p=0.08).
When stratified into subgroups according to the presence of microbiota in the uterine cavity, similarly, the recurrence rate did not differ in the absence of microbiota, regardless of AB prescription (7/22 (31.8%) and 5/22 (22.7%), respectively). However, in the group with microflora, growth in the uterine cavity was seven times higher in the absence of AB prescription: 1/20 (5%) in the AB group and 8/20 (40%) in the group without AB (p=0.02).
Discussion
Chronic endometritis is associated with EPs, and according to the literature, it is diagnosed in 27% to 86% of cases [23–26]. This is supported by data from a 2021 meta-analysis, which revealed that the overall prevalence of CE was 51.3% (95% CI 27.2– 75.1%) and three times higher in women with EPs (OR 3.1, 95% CI 1.6–5.9) [27]. Additionally, evidence suggests that the risk of recurrence of EPs is 2.5 to 5.2 times higher in the presence of CE [14, 28, 29]. However, contrasting results emerged from a Mendelian randomization study and generalized genome-wide association study (GWAS) statistics in a European population, which did not find an association between CE and EPs [30]. In our study, CE was detected in 23/84 (27.4%) patients with EPs. The recurrence rate within 12 to 18 months was 21/84 (25%), of which 9/23 (39.1%) had CE compared to 12/61 (19.7%) in patients without CE (p=0.09, OR=2.8, 95% CI 0.9–8.4).
Colonization of the uterine cavity by microorganisms was observed in 40/84 (47.6%) patients with EPs. Among patients with CE, colonization was noted in 100% of the cases, consistent with the literature on non-sterility of the uterine cavity in both normal and pathological states [18, 31–37]. Several studies have demonstrated the significance of uterine microbiota in the development of female reproductive system diseases, particularly endometrial hyperplasia and adenomyosis [36, 38–42]. While there are limited studies specifically focusing on EPs, evidence indicates that compared to healthy women, those with EPs exhibit changes in the composition of the uterine cavity microbiota, primarily characterized by an increased frequency of vaginal bacteria such as Lactobacillus [43, 44]. This alteration may facilitate cell migration and proliferation, thus contributing to localized endometrial hyperplasia and polyp formation [45]. In our study, the predominant species found in EPs were microorganisms of the genus Staphylococcus (20/40 (50%) patients) and Lactobacillus (15/40 [37.5%] patients). In cases of CE, Staphylococcus was detected 22 times more frequently compared to women without CE (p<0.001, OR=22.3, 95% CI 7.3–68.4).
Despite the common association between EPs and CE, as well as the established non-sterility of the uterine cavity in this condition, the prescription of AB prophylaxis or AB therapy during the surgical removal of EPs is not recommended by domestic and international professional associations [10, 46–48]. Numerous studies have demonstrated the ineffectiveness of AB therapy in patients with EPs and CE, in terms of recovery after polypectomy and future pregnancies [11]. Conversely, some data suggest the potential benefits of prescribing ABs during the surgical treatment of EPs [13], including an increased risk of recurrence in the presence of CE [14], improved pregnancy rates with AB treatment for CE in women with infertility [16], and multiple implantation failures [17]. In our study, no significant difference in the recurrence rate of EPs was observed in the overall group of patients (group 1, 8/42 (19%); group 2, 13/42 (31%); p=0.21), which aligns with existing literature. However, when stratified into subgroups according to the presence of CE and microflora growth in the uterine cavity, the recurrence rate was five times higher in the presence of CE in the absence of AB prescription (p=0.08) and seven times higher in the presence of microflora growth in the uterine cavity in the absence of AB prescription (p=0.02).
Conclusion
Chronic endometritis was associated with the recurrence of EPs within 12–18 months after HRS. The recurrence rate was 25% and occurred 2.8 times more frequently in the background of CE (OR=2.8, 95% CI 0.9–8.4). Colonization of the uterine cavity by microorganisms was observed in 47.6% of patients with EPs and in 100% of patients with CE. The predominant microbial species were of the genus Staphylococcus, which were detected 22 times more frequently in patients with CE (OR=22.3, 95% CI 7.3–68.4). AB prophylaxis during HRS in the overall patient group did not reduce the recurrence rate of EPs; however, in patients with CE and microbial growth in the uterine cavity, it reduced the recurrence rate by five and seven times, respectively. These findings underscore the role of microbial factors in the genesis of CE and associated EPs as well as the potential efficacy of ABs in reducing the recurrence rate of EPs in patients with CE and microbial growth in the uterine cavity.
References
- Nijkang N.P., Anderson L., Markham R., Manconi F. Endometrial polyps: Pathogenesis, sequelae and treatment. SAGE Open Med. 2019; 7: 2050312119848247. https://dx.doi.org/10.1177/2050312119848247.
- Fatemi H.M., Kasius J.C., Timmermans A., van Disseldorp J., Fauser B.C., Devroey P. et al. Prevalence of unsuspected uterine cavity abnormalities diagnosed by office hysteroscopy prior to in vitro fertilization. Hum. Reprod. 2010; 25(8): 1959-65. https://dx.doi.org/10.1093/humrep/deq150.
- Di Spiezio Sardo A., Di Carlo C., Minozzi S., Spinelli M., Pistotti V., Alviggi C. et al. Efficacy of hysteroscopy in improving reproductive outcomes of infertile couples: a systematic review and meta-analysis. Hum. Reprod. Update. 2016; 22(4): 479-96. https://dx.doi.org/10.1093/humupd/dmw008.
- Clark T.J., Stevenson H. Endometrial Polyps and Abnormal Uterine Bleeding (AUB-P): What is the relationship, how are they diagnosed and how are they treated? Best Pract. Res. Clin. Obstet. Gynaecol. 2017; 40: 89-104. https://dx.doi.org/10.1016/j.bpobgyn.2016.09.005.
- Paradisi R., Rossi S., Scifo M.C., Dall'O' F., Battaglia C., Venturoli S. Recurrence of endometrial polyps. Gynecol. Obstet. Invest. 2014; 78(1): 26-32. https://dx.doi.org/10.1159/000362646.
- AlHilli M.M., Nixon K.E., Hopkins M.R., Weaver A.L., Laughlin-Tommaso S.K., Famuyide A.O. Long-term outcomes after intrauterine morcellation vs hysteroscopic resection of endometrial polyps. J. Minim. Invasive Gynecol. 2013; 20(2): 215-21. https://dx.doi.org/10.1016/j.jmig.2012.10.013.
- Yang J.H., Chen C.D., Chen S.U., Yang Y.S., Chen M.J. Factors influencing the recurrence potential of benign endometrial polyps after hysteroscopic polypectomy. PLoS One. 2015; 10(12): e0144857. https://dx.doi.org/ 10.1371/journal.pone.0144857.
- Tchente N.C., Brichant G., Nisolle M. [Asherman's syndrome: management after curettage following a postnatal placental retention and literature review]. Rev. Med. Liege. 2018; 73(10): 508-12. (in French).
- Cicinelli E., Bettocchi S., de Ziegler D., Loizzi V., Cormio G., Marinaccio M. et al. Chronic endometritis, a common disease hidden behind endometrial polyps in premenopausal women: first evidence from a case-control study. J. Minim. Invasive Gynecol. 2019; 26(7): 1346-50. https://dx.doi.org/10.1016/j.jmig.2019.01.012.
- Министерство здравоохранения Российской Федерации. Клинические рекомендации. Полипы эндометрия. 2023. [Ministry of Health of the Russian Federation. Clinical guidelines. Endometrial polyps. 2023. (in Russian)].
- Kuroda K., Takamizawa S., Motoyama H., Tsutsumi R., Sugiyama R., Nakagawa K. et al. Analysis of the therapeutic effects of hysteroscopic polypectomy with and without doxycycline treatment on chronic endometritis with endometrial polyps. Am. J. Reprod. Immunol. 2021; 85(6): e13392. https://dx.doi.org/10.1111/aji.13392.
- Дубровина С.О., Божинская Д.М., Гимбут В.С., Богунова Д.Ю., Циркунова Н.С., Циркунова К.Г. Хронический эндометрит. Акушерство и гинекология. 2024; 6: 147-52. [Dubrovina S.O., Bozhinskaya D.M., Gimbut V.S., Bogunova D.Yu., Tsirkunova N.S., Tsirkunova K.G. Chronic endometritis. Obstetrics and Gynecology. 2024; (6): 147-52 (in Russian)]. https://dx.doi.org/10.18565/aig.2024.137.
- Liu Y., Yu X., Huang J., Du C., Zhou H., Yang Y. et al. Additional dydrogesterone for the treatment of chronic endometritis treated with antibiotic in premenopausal women with endometrial polyps: a retrospective cohort study. BMC Womens Health. 2022; 22(1): 435. https://dx.doi.org/10.1186/s12905-022-02033-0.
- Qu D., Liu Y., Zhou H., Wang Z. Chronic endometritis increases the recurrence of endometrial polyps in premenopausal women after hysteroscopic polypectomy. BMC Womens. Health. 2023; 23: 88. https://dx.doi.org/10.1186/s12905-023-02232-3.
- Асатурова А.В., Чернуха Г.Е., Иванов И.А., Куземин А.А. Клинико-морфометрические особенности полипов эндометрия и механизмы возникновения аномальных маточных кровотечений. Акушерство и гинекология. 2019; 7: 64-70. [Asaturova A.V., Chernukha G.E., Ivanov I.A., Kuzemin A.A. Clinical and morphometric characteristics of endometrial polyps and underlying mechanisms of abnormal uterine bleeding. Obstetrics and Gynecology. 2019; (7): 64-70 (in Russian)]. https://dx.doi.org/10.18565/aig.2019.7.64-70.
- Vitagliano A., Saccardi C., Noventa M., Di Spiezio Sardo A., Saccone G., Cicinelli E. et al. Effects of chronic endometritis therapy on in vitro fertilization outcome in women with repeated implantation failure: a systematic review and meta-analysis. Fertil. Steril. 2018; 110(1): 103-112.e1. https://dx.doi.org/10.1016/j.fertnstert.2018.03.017.
- Kitaya K., Matsubayashi H., Takaya Y., Nishiyama R., Yamaguchi K., Takeuchi T. et al. Live birth rate following oral antibiotic treatment for chronic endometritis in infertile women with repeated implantation failure. Am. J. Reprod. Immunol. 2017; 78(5). https://dx.doi.org/10.1111/aji.12719.
- Zhu N., Yang X., Liu Q., Chen Y., Wang X., Li H. et al. "Iron triangle" of regulating the uterine microecology: endometrial microbiota, immunity and endometrium. Front. Immunol. 2022; 13: 928475. https://dx.doi.org/10.3389/fimmu.2022.928475.
- Critchley H.O.D., Babayev E., Bulun S.E., Clark S., Garcia-Grau I., Gregersen P.K. et al. Menstruation: science and society. Am. J. Obstet. Gynecol. 2020; 223(5): 624-64. https://dx.doi.org/10.1016/j.ajog.2020.06.004.
- Santiago G.L., Tency I., Verstraelen H., Verhelst R., Trog M., Temmerman M. et al. Longitudinal qPCR Study of the Dynamics of L. crispatus, L. iners, A. vaginae, (Sialidase Positive) G. vaginalis, and P. bivia in the vagina. PLoS One. 2012; 7(9): e45281. https://dx.doi.org/10.1371/journal.pone.0045281.
- Pelzer E.S., Willner D., Buttini M., Huygens F. A role for the endometrial microbiome in dysfunctional menstrual bleeding. Antonie Van Leeuwenhoek. 2018; 111(6): 933-43. https://dx.doi.org/10.1007/s10482-017-0992-6.
- Кузнецова А.В. Хронический эндометрит. Архив патологии. 2000; 62(3): 48-52. [Kuznetsova A.V. Chronic endometritis. Arkhiv Patologii. 2000; 62(3): 48-52. (in Russian)].
- Carvalho F.M., Aguiar F.N., Tomioka R., de Oliveira R.M., Frantz N., Ueno J. Functional endometrial polyps in infertile asymptomatic patients: a possible evolution of vascular changes secondary to endometritis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013; 170(1): 152-6. https://dx.doi.org/10.1016/j.ejogrb.2013.05.012.
- Vaduva C.C., Constantinescu C., Serbanescu M., Dara L., Oancea M.D., Carp-Veliscu A. The association between endometrial polyps, chronic endometritis, and IVF outcomes. Eur. Rev. Med. Pharmacol. Sci. 2023; 27(18): 8895-904. https://dx.doi.org/10.26355/eurrev_202309_33810.
- Kuroda K., Yamanaka A., Takamizawa S., Nakao K., Kuribayashi Y., Nakagawa K. et al. Prevalence of and risk factors for chronic endometritis in patients with intrauterine disorders after hysteroscopic surgery. Fertil. Steril. 2022; 118(3): 568-75. https://dx.doi.org/10.1016/j.fertnstert.2022.05.029.
- Peng J., Guo J., Zeng Z., Liang X., Zeng H., Li M. Endometrial polyp is associated with a higher prevalence of chronic endometritis in infertile women. Int. J. Gynaecol. Obstet. 2022; 159(2): 563-7. https://dx.doi.org/10.1002/ijgo.14207.
- Vitagliano A., Cialdella M., Cicinelli R., Santarsiero C., Greco P., Buzzaccarini G. et al. Association between endometrial polyps and chronic endometritis: Is it time for a paradigm shift in the pathophysiology of endometrial polyps in pre-menopausal women? Results of a systematic review and meta-analysis. Diagnostics. 2021; 11(12): 2182. https://dx.doi.org/10.3390/diagnostics11122182.
- Huang J., You X., Zhao Z., Jiang X., Qu D. Chronic endometritis multiplies the recurrence risk of endometrial polyps after transcervical resection of endometrial polyps: a prospective study. BMC Womens Health. 2024; 24(1): 372. https://dx.doi.org/10.1186/s12905-024-03221-w.
- Hosseini S., Abbasi H., Salehpour S., Saharkhiz N., Nemati M. Prevalence of chronic endometritis in infertile women undergoing hysteroscopy and its association with intrauterine abnormalities: A cross-sectional study. JBRA Assist. Reprod. 2024; 28(3): 430-4. https://dx.doi.org/10.5935/1518-0557.20240011.
- Wei L., Zhao Y., Xu S., Zhang C. Association between endometritis and endometrial polyp: A Mendelian randomization study. Int. J. Womens Health. 2023; 15: 1963-70. https://dx.doi.org/10.2147/IJWH.S434299.
- Ansbacher R., Boyson W.A., Morris J.A. Sterility of the uterine cavity. Am. J. Obstet. Gynecol. 1967; 99(3): 394-6. https://dx.doi.org/10.1016/s0002-9378(16)34549-5.
- Mitchell C.M., Haick A., Nkwopara E., Garcia R., Rendi M., Agnew K. et al. Colonization of the upper genital tract by vaginal bacterial species in nonpregnant women. Am. J. Obstet. Gynecol. 2015; 212(5): 611.e1-9. https://dx.doi.org/10.1016/j.ajog.2014.11.043.
- Verstraelen H., Vilchez-Vargas R., Desimpel F., Jauregui R., Vankeirsbilck N., Weyers S. et al. Characterisation of the human uterine microbiome in non-pregnant women through deep sequencing of the V1-2 region of the 16S rRNA gene. PeerJ. 2016; 4: e1602. https://dx.doi.org/10.7717/peerj.1602.
- Giudice L.C. Challenging dogma: the endometrium has a microbiome with functional consequences! Am. J. Obstet. Gynecol. 2016; 215(6): 682-3. https://dx.doi.org/10.1016/j.ajog.2016.09.085.
- Тапильская Н.И., Будиловская О.В., Крысанова А.А., Толибова Г.Х., Копылова А.А., Цыпурдеева Н.Д., Гзгзян А.М., Савичева А.М., Коган И.Ю. Микробиота эндометрия женщин с хроническим эндометритом и идиопатическим бесплодием. Акушерство и гинекология. 2020; 4: 72-81. [Tapilskaya N.I., Budilovskaya O.V., Krysanova A.A., Tolibova G.Kh., Kopylova A.A., Tsypurdeeva N.D., Gzgzyan A.M., Savicheva A.M., Kogan I.Yu. Еndometrial microbiota of women with chronic endometritis and idiopathic infertility. Obstetrics and Gynecology. 2020; (4): 72-81. (in Russian)]. https://dx.doi.org/10.18565/aig.2020.4.72-81.
- Chen C., Song X., Wei W., Zhong H., Dai J., Lan Z. et al. The microbiota continuum along the female reproductive tract and its relation to uterine-related diseases. Nat. Commun. 2017; 8(1): 875. https://dx.doi.org/10.1038/s41467-017-00901-0.
- Miles S.M., Hardy B.L., Merrell D.S. Investigation of the microbiota of the reproductive tract in women undergoing a total hysterectomy and bilateral salpingo-oopherectomy. Fertil. Steril. 2017; 107(3): 813-820.e1. https://dx.doi.org/10.1016/j.fertnstert.2016.11.028.
- Margulies S.L., Flores V., Parkash V., Pal L. Chronic endometritis: A prevalent yet poorly understood entity. Int. J. Gynaecol. Obstet. 2022; 158(1): 194-200. https://dx.doi.org/10.1002/ijgo.13962.
- Kubyshkin A.V., Aliev L.L., Fomochkina I.I., Kovalenko Y.P., Litvinova S.V., Filonenko T.G. et al. Endometrial hyperplasia-related inflammation: its role in the development and progression of endometrial hyperplasia. Inflamm. Res. 2016; 65(10): 785-94. https://dx.doi.org/10.1007/s00011-016-0960-z.
- Cai L. Preliminary study on the mechanism of uterine flora on the occurrence and development of endometrial hyperplasia. Jinan University. 2019; 2: 44. https://dx.doi.org/10.27167/d.cnki.gjinu.2019.000458.
- Khan K.N., Fujishita A., Masumoto H., Muto H., Kitajima M., Masuzaki H. et al. Molecular detection of intrauterine microbial colonization in women with endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016; 199: 69-75. https://dx.doi.org/10.1016/j.ejogrb.2016.01.040.
- Khan K.N., Fujishita A., Kitajima M., Hiraki K., Nakashima M., Masuzaki H. Intra-uterine microbial colonization and occurrence of endometritis in women with endometriosis. Hum. Reprod. 2014; 29(11): 2446-56. https://dx.doi.org/10.1093/humrep/deu222.
- Fang R.L., Chen L.X., Shu W.S., Yao S.Z., Wang S.W., Chen Y.Q. Barcoded sequencing reveals diverse intrauterine microbiomes in patients suffering with endometrial polyps. Am. J. Transl. Res. 2016; 8(3): 1581-92.
- Cicinelli E., De Ziegler D., Nicoletti R., Tinelli R., Saliani N., Resta L. et al. Poor reliability of vaginal and endocervical cultures for evaluating microbiology of endometrial cavity in women with chronic endometritis. Gynecol. Obstet. Invest. 2009; 68(2): 108-15. https://dx.doi.org/10.1159/000223819.
- Teame T., Wang A., Xie M., Zhang Z., Yang Y., Ding Q. et al. Paraprobiotics and postbiotics of probiotic Lactobacilli, their positive effects on the host and action mechanisms: a review. Front. Nutr. 2020; 22(7): 570344. https://dx.doi.org/10.3389/fnut.2020.570344.
- American Association of Gynecologic Laparoscopists. AAGL practice report: practice guidelines for the diagnosis and management of endometrial polyps. J. Minim. Invasive Gynecol. 2012; 19(1): 3-10. https://dx.doi.org/10.1016/j.jmig.2011.09.003.
- Endometrial polyps. National Guideline approved by The Norwegian Society of Obstetrics and Gynecology. 2015. Available at: https://nfog.org/files/guidelines/NFOG_Guideline_NOR_160419%20Endometrial%20polyp%20NO%20merged.pdf (accessed 24.02.2025).
- Royal College of Obstetricians & Gynaecologists. Management of Endometrial Hyperplasia. Green-top Guideline No.67. RCOG/BSGE Joint Guideline. February 2016. Available at: https://www.rcog.org.uk/guidance/browse-all-guidance/green-top-guidelines/management-of-endometrial-hyperplasia-green-top-guideline-no-67/
Received 28.12.2024
Accepted 12.02.2025
About the Authors
Angelina I. Vanakova, obstetrician-gynecologist, oncologist at the Department of Innovative Oncology and Gynecology, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4; PhD student at the Department of Obstetrics, Gynecology, Perinatology and Reproductology, IPE, I.M. Sechenov First MSMU, Ministry of Health of Russia (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8-2, +7(903)108-17-90, angelinavanakova@gmail.com,https://orcid.org/0009-0007-8048-1682
Nataliya V. Dolgushina, Dr. Med. Sci., Professor, Deputy Director, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4; Professor at the Department of Obstetrics, Gynecology, Perinatology and Reproductology, IPE, I.M. Sechenov First MSMU, Ministry of Health of Russia (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8-2, n_dolgushina@oparina4.ru, https://orcid.org/0000-0003-1116-138X
Pavel A. Denisov, Researcher at the Laboratory of Bioinformational Analysis, Institute of Microbiology, Antimicrobial Therapy and Epidemiology, V.I. Kulakov NMRC
for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, https://orcid.org/0000-0003-1813-6718
Olga D. Goncharuk, Head of the Laboratory of Medical Microbiology, Institute of Microbiology, Antimicrobial Therapy and Epidemiology, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, https://orcid.org/0000-0001-5876-8424
Vera V. Muravieva, PhD (Bio), Senior Researcher at the Laboratory of Molecular Microbiology, Institute of Microbiology, Antimicrobial Therapy and Epidemiology,
V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, https://orcid.org/0000-0003-0383-0731
Tatiana V. Priputnevich, Corresponding Member of the RAS, Dr. Med. Sci., Associate Professor, Director of the Institute of Microbiology, Antimicrobial Therapy and Epidemiology, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, +7(910)414-56-16, priput1@gmail.com,
https://orcid.org/0000-0002-4126-9730



