Lipid profile of fetal membrane tissue in premature rupture of membranes
Baisova A.R., Amiraslanov E.Yu., Frankevich V.E., Chagovets V.V., Tokareva A.O., Kulikova G.V., Shchegolev A.I., Каn N.E., Tyutyunnik V.L.
Objective: To study the characteristics of the lipid profile of fetal membrane tissue in premature rupture of membranes to clarify the pathways for pathogenesis.
Materials and methods: This prospective study included 91 patients who were divided into groups. Group 1 consisted of 33 pregnant women who had premature rupture of membranes (PROM): 10 patients had PROM between 220 and 366 weeks gestation and 23 patients had PROM at more than 370 weeks gestation. The control group included 58 patients with intact fetal membranes who delivered at term. Lipidomic profiling of fetal membrane tissue was performed as part of the study and isolated lipid extracts were analyzed using a Dionex UltiMate 3000 liquid chromatograph with a Maxis Impact qTOF mass spectrometer. The histological study of inflammatory reactions in the extraplacental membranes was also performed; the stage and degree of maternal inflammatory response were determined according to the criteria of Redline R.W. (2003) and the International Amsterdam Consensus (2016).
Results: A total of 172 lipids belonging to different classes were detected in fetal membrane tissues in positive and negative ion mode. The comparison of lipid composition in the fetal membrane tissues of patients who had preterm labor complicated by PROM showed that there were 28 lipids belonging to the classes of oxidized lipids and cholesterol esters; the number of the lipids was statistically significant. There were 44 lipids identified in patients with term pregnancies, and these lipids showed statistically significant differences. The histological study showed that the most severe inflammatory responses in the extraplacental membranes were observed in the group of patients with premature rupture at early gestation.
Conclusion: The lipid composition of the fetal membranes showed significant changes in the main lipid classes in patients with PROM, including lipids involved in the initiation of inflammatory processes and apoptosis. The activation of these processes leads to the structural changes in the fetal membranes and increases the risk of rupture. Thus, the alteration of the lipid profile in premature rupture of membranes may be one of the key components of the pathogenesis of this pregnancy complication and could serve as a potential biomarker for diagnosing and predicting this condition.
Authors’ contributions: Baisova A.R. – developing the study design, obtaining data for analysis, collecting biological materials for the study, drafting the manuscript; Amiraslanov E.Yu. – developing the study design, review of publications on the topic of the article, analysis of the obtained data, writing the text of the manuscript; Chagovets V.V. – conducting metabolomic analysis using mass spectrometry, statistical analysis; Tokareva A.O. – conducting the statistical analysis of the data; Kulikova G.V. – conducting the analysis of microscopic data, statistical analysis; Amiraslanov E.Yu., Frankevich V.E., Chagovets V.V., Kan N.E., Tyutyunnik V.L., Shchеgolev A.I. – editing the article.
Conflicts of interest: The authors declare that there are no conflicts of interest.
Funding: The study was supported by the Russian Science Foundation Grant No. 24-64-00006,
https://rscf.ru/project/24-64-00006/.
Ethical Approval: The study was approved by the Ethical Review Board of the Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Moscow (protocol No. 11 as of November 11, 2021).
Patient Consent for Publication: The patients signed informed consent for the publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available on request from the corresponding author after approval from the principal investigator.
For citation: Baisova A.R., Amiraslanov E.Yu., Frankevich V.E., Chagovets V.V.,
Tokareva A.O., Kulikova G.V., Shchegolev A.I., Каn N.E., Tyutyunnik V.L.
Lipid profile of fetal membrane tissue in premature rupture of membranes.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2025; (2): 40-51 (in Russian)
https://dx.doi.org/10.18565/aig.2025.37
Keywords
Premature rupture of membranes (PROM) is one of the most relevant and complex problems in modern obstetrics. It is a pathological condition characterized by the disruption of the integrity of the fetal membranes prior to the onset of labor. The condition can be classified as either term or preterm, depending on the time of onset and gestational age [1, 2]. PROM occurs in approximately 3% of all pregnancies; however, it is clinically significant because of its associated complications such as maternal infection [1, 3, 4] and high neonatal morbidity and mortality [5, 6]. The Federal State Statistics Service obtained the data for 2010 and 2019 in the Russian Federation as a whole and reported that placental pathology, including fetal membrane lesions, caused stillbirth in 45.3% and 45.1%, respectively, of the total number of stillbirths [7, 8] and contributed to the death of the newborn in 17.2% and 23.9%, respectively [9, 10].
Despite the considerable amount of research devoted to the study of this pathology, the underlying mechanism of its occurrence remains understudied. Current understanding of the PROM pathogenesis suggests that its development results from a complex interaction of structural, biochemical, inflammatory, and possibly genetic factors [11–14]. The impairment of mechanisms regulating the strength and integrity of fetal membranes may be due to infectious inflammatory processes, changes in the synthesis of collagen and matrix metalloproteinases [15, 16], cellular apoptosis [17–19] and immunological dysfunctions [12, 20, 21]. Interactions between fetal membrane tissues and the environment, including amniotic fluid and microbiota, also play a key role.
PROM is mainly caused by inflammation, collagen degradation and destruction of the extracellular matrix. The inflammatory response is initiated via different pathways, including NF-κB (nuclear factor κB) signaling pathway and circular RNA interactions [22]. Inflammation plays a crucial role in both term and preterm labor, and it also affects the fetal membranes [23]. The inflammatory response in PROM can be triggered by infection or sterile mechanisms involving damage-associated molecular patterns (DAMPs) and pattern recognition receptors (PRRs) such as RAGE inflammasome and NLRP7 [24–26]. In turn, the macrophage response may play a critical role by secreting matrix metalloproteinases (MMPs) that soften the cervix and damage the extracellular matrix [16, 22]. MMPs, particularly MMP-2, MMP-8 and MMP-9, are key enzymes in membrane degradation and can be induced by both sterile and non-sterile inflammation [16].
Lipids are an essential structural component of cell membranes; they are involved in cell signaling and function as key regulators of various cellular processes such as cell division, proliferation and cell death. Increased lipid membrane permeability is a distinctive feature of apoptosis, which is characterized by an increase in membrane permeability and enables the release of apoptotic factors into the extracellular space [27, 28].
In addition, lipids function as mediators in the inflammatory process, with platelet-activating factor stimulating inflammatory pathways [29]. Saturated fatty acids can activate Toll-like receptors (TLRs), whereas omega-3 polyunsaturated fatty acids inhibit them [30]. Oxidized phospholipids also play a role in modulating inflammation through TLR activation [30].
Thus, the process of fetal membrane rupture is an integration of biochemical changes induced by hormonal and irritant agents as well as by mechanical forces acting on the tissue already weakened. These processes are interconnected and dynamic, making the study of ways to prevent premature rupture of membranes a relevant issue in obstetrics and perinatology.
Understanding the mechanisms underlying PROM is an important step for developing preventive and therapeutic interventions to reduce maternal and perinatal morbidity and mortality. This article presents a modern insight into the pathogenesis of PROM with the analysis of the lipid profile of the fetal membranes and histological findings in order to identify the key factors affecting the strength of the fetal membranes and the mechanisms of their sequential damage.
The aim of the study is to investigate the characteristics of the lipid profile of fetal membrane tissue in premature rupture of membranes to clarify the components of pathogenesis.
Materials and methods
This was a prospective study that included 91 patients. All pregnant women were divided into groups: group I (main group) consisted of 33 patients with PROM, which included 10 patients (subgroup Ia) at 220 to 366 weeks gestation and 23 patients (subgroup Ib) with gestation exceeding 370 weeks. Group II (control group) included women with intact fetal membranes who gave birth at term (n=58). There were the following inclusion criteria: signed informed consent from the patient to participate in the study, singleton pregnancy, age from 18 to 45 years, and gestational age of 22 weeks or more. The exclusion criteria were the presence of severe somatic pathology, lipid metabolism disorders, diabetes mellitus, arterial hypertension, multiple pregnancy, chromosomal anomalies or congenital fetal malformations, oncological diseases, hypertensive complications during pregnancy, and gestational diabetes mellitus. Premature rupture of the fetal membranes was diagnosed using the criteria listed in the approved clinical guidelines [2].
The patients were treated and monitored according to the clinical guidelines for normal pregnancy and preterm labor [2, 31].
In the course of the study, three 2×2 cm samples of fetal membranes were obtained from the patients after delivery (the biomaterial was taken directly at the edge of the fetal membrane rupture in the patients of the main group). The obtained tissue samples were washed in physiologic saline (0.9% NaCl) solution. After washing, the samples were placed in a labelled tube and underwent shock freezing in liquid nitrogen. The samples were further stored in the collection of the Biobank of the National Medical Research Centre for Obstetrics, Gynecology and Perinatology, Moscow.
Extracting lipids from tissues
The Folch method was used for lipid extraction. Fetal membrane tissues were thawed and homogenized in liquid nitrogen. A quantity of 50 mg of homogenate was placed into a 2-ml tube, followed by the addition of 700 μl of a chloroform/methanol solution (2:1). The sample was treated in an ultrasonic water bath at room temperature for 10 minutes, then 440 µl of purified water was added for mixing and centrifugation.
The lower organic layer was withdrawn and 730 μl of solution was added for the second extraction.
The organic layer was then dried in a stream of nitrogen and was redissolved in isopropanol/acetonitrile (500 μl) for analysis. The upper aqueous methanol layer was also dried and redissolved.
The lipid extracts were analyzed using a Dionex UltiMate 3000 liquid chromatograph with a Maxis Impact qTOF mass spectrometer. Separation was performed using Zorbax C18 column (150×2.1 mm) with a gradient of 15% to 45% eluent B for 2 minutes and then 45% to 99% for 15 minutes.
Lipid identification was performed with the use of tandem mass spectrometry and LipidMatch software, according to LipidMaps nomenclature.
Histological examination
The fragments of extraplacental membranes were used for histological examination. The material was fixed in 4% neutral formalin and embedded in paraffin. Paraffin sections were cut at a thickness of 4 μm and stained with hematoxylin and eosin. Microscopic examination was performed using Nikon eclipse 80i light microscope and NIS Elements Advanced Research 4.1. computer software for image analysis.
The stage and degree of maternal inflammatory response were determined according to the criteria of Redline R.W. [32] and the International Amsterdam Consensus [33].
Statistical analysis
Statistical processing of the data was performed on a personal computer using SPSS Statistics software version 21.0 for Windows. Normality of distribution was determined according to Shapiro–Wilk test and Kolmogorov–Smirnov test. In case the data had a normal distribution, calculations were performed using mean values (M) and standard deviations (SD). In the absence of normal distribution, descriptive statistics included the median (Me) and the lower and upper quartile values (Q1; Q3).
The comparative analysis of quantitative data between the two groups, which distribution deviated from normal, was performed using the Mann–Whitney U-test, using the Bonferroni correction. The comparative analysis of quantitative data among the three groups was performed using the Kruskal–Wallis test. The results of the histological study in groups were compared using the χ2 criterion (chi-square) and Fisher’s exact test. Student’s t-test was used for the comparative analysis of quantitative data with normal distribution. Differences were considered statistically significant at the level of p<0.05.
The odds ratio (OR) was used to quantify the effect in comparative analysis of relative performance. The OR values were obtained using the contingency tables. In order to illustrate ten obtained OR values for the general population, the limits of the 95% confidence interval (95% CI) were determined.
Results
The clinical and anamnestic characteristics of the examined women are presented in Table 1.
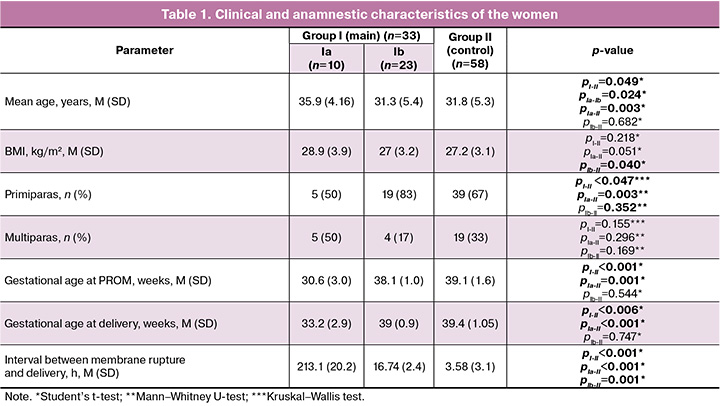
The mean age of pregnant women included in the study was 32.0 (5.3) years. The rupture of fetal membranes was significantly more frequent in the age group above 30 years (20/33 (60%), pI-II=0.049). Body mass index (BMI) in patients of all groups was comparable and had no statistically significant differences: it was 28.9 (3.9) kg/m2 in patients of subgroup Ia, 27.0 (3.2) kg/m2 in subgroup Ib, and 27.2 (3.1) kg/m2 in the control group (pI-II=0.218).
Primiparous women accounted for 50% (5/10) of patients who had preterm labor complicated by PROM, and their number was significantly lower than in the control group, where the proportion of primiparous women was as high as 67% (52/58; pIa-II=0.002). Most patients were also primiparous in the group of term delivery with PROM, namely 83% (19/23), and their number was higher than that of the control group, 67% (39/58), but there was no statistically significant difference (pIb-II=0.352). Multiparous patients predominated in the control group (33% (19/58); pI-II =0.155).
Analysis of extragenital morbidity showed that the incidence of chronic pyelonephritis was significantly more frequent among patients with PROM, especially in subgroup Ia (2/10 (20%); pIa-II=0.044); however, this difference between subgroup Ib (2/23 (8.6%)) and the control group (1/58 (1.7%)) did not reach the level of statistical significance (pIb-II=0.153). Chronic cystitis was also reported significantly more frequently in subgroup Ia (2/10 (20%); pIa-II=0.025), subgroup Ib (3/23 (13%); pIb-II=0.049) and the control group (3/58 (3.4%)) had the same frequency. Chronic gastritis was reported more frequently in the group with PROM, namely in subgroup Ia (1/10 (10%); pIa-II=0.175) and subgroup Ib (4/23 (17%); pIb-II=0.089) compared to the control group (5/58 (8.6%)), but the differences did not reach a statistically significant level. No significant differences were found among the groups for other extragenital diseases.
The analysis of the course of pregnancy in trimesters revealed the following characteristics: subgroup Ia had a high risk of threatened miscarriage more often in the first trimester (OR=7.2, 95% CI: 1.7–30.2; pIa-II=0.022), the incidence of cervical incompetence was significantly higher in the second trimester (OR=8.7, 95% CI: 1.9–38.2; pIa-II=0.002), and there was a significant increase in the risk of preterm labor in the third trimester (OR=2.5, 95% CI: 0.19–21.8; pIa-II=0.049) compared to the control group. Subgroup Ib was more likely to have nonspecific vulvovaginitis (OR=5.43, 95% CI: 0.47–63.04; pIb-II=0.001) in the second trimester and this subgroup was significantly more likely to have large fetal size estimated by ultrasound (OR=1.74, 95% CI: 0.5–6.0; pIb-II=0.003) compared to the control group.
Spontaneous vaginal delivery was predominant in subgroup Ib (18/23 (78%); pIb-II<0.383) compared to the control group (50/58 (86%); pI-II=0.028). Subgroup Ia had a high rate of operative delivery, 70% (7/10; pIa-II=0.001). The high proportion of urgent surgeries in subgroup Ia, namely 100% (7/7) of cases, is particularly worth noting. The main indications for operative delivery were uterine scar in combination with other obstetric conditions, increasing laboratory markers of inflammation in patients with PROM, fetal distress, poor progress of labor, and fetal malpresentation.
Results of lipidomic analysis of fetal membranes
A total of 172 lipids were detected in fetal membrane tissues, in both positive and negative ion mode. These lipids belonged to different classes including cholesterol esters, ceramides, cardiolipins, diacylglycerols, (lyso-) phosphatidylcholines, simple ester-linked lipids, phosphatidylinositols and glycerols, oxidized lipids, sphingomyelins and triacylglycerols.
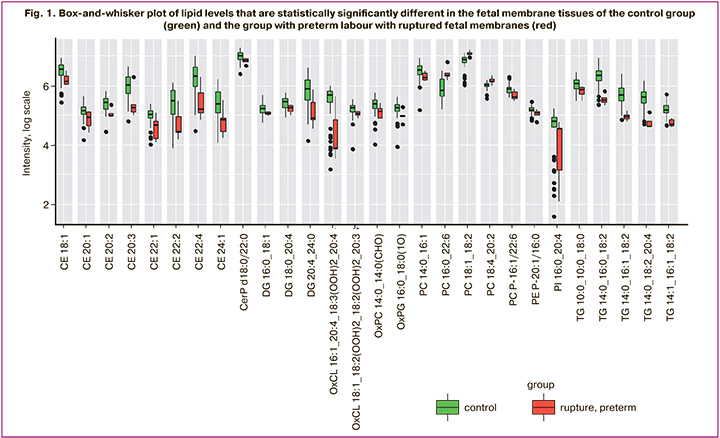
The comparison of lipid composition in the fetal membrane tissues of patients from the control group and the group with preterm labor complicated by PROM showed statistically significant 28 lipids (Fig. 1, Table 2). There was an increase in the levels of lipids such as PC 16:0 22:6, PC 18:1 18:2, PC 18:1 18:2, PC 18:4 20:2 in the group of patients who had preterm labor with PROM, while there was a decrease in the levels of other lipids.
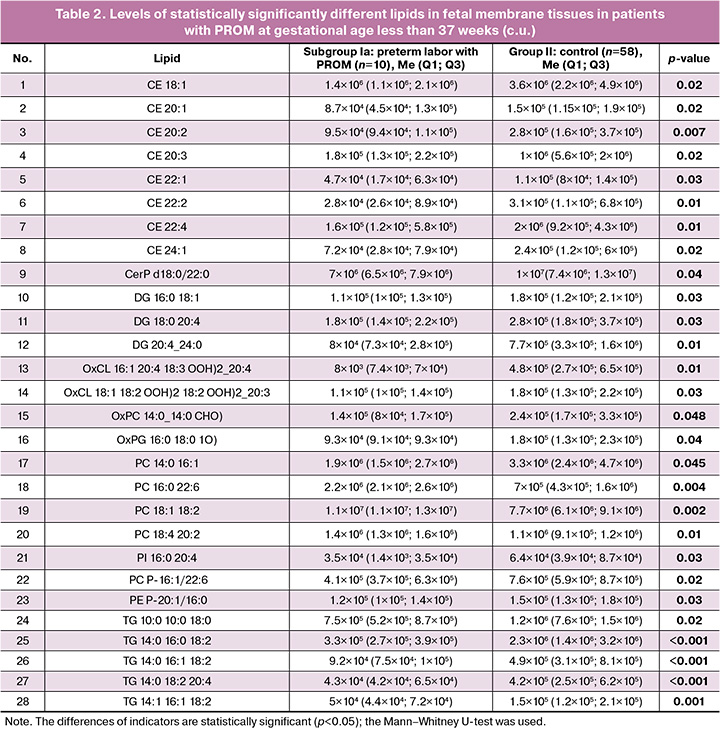
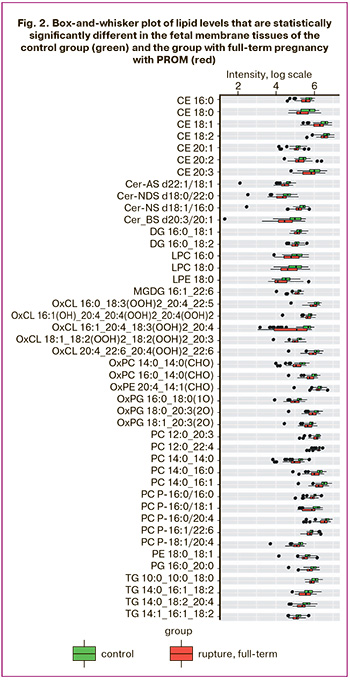
The comparative analysis of the lipid composition of fetal membrane tissues between patients in the control group and the group with full-term pregnancies with PROM revealed 44 lipids that showed statistically significant differences (Fig. 2, Table 3). All cases in the group with PROM showed a decrease in the levels of these lipids.
The results of the histological examination
The obtained data showed the presence of polymorphonuclear neutrophils in the extraplacental membranes of the patients in all study groups; these neutrophils are a histological marker of acute inflammation. Acute inflammation was detected in two cases, accounting for 20% (2/10) in subgroup Ia, and in one case, 4.34% (1/23) in group Ib, while in the control group it was noted in 1.7% (1/58) of the observations; the differences were statistically significant when subgroup Ia was compared with the control group (χ2=4.77, pIa-II=0.0290).
Stage 1 acute inflammation was found to be present only in subgroup Ia (10% (1/10)) and absent in the other groups; the differences were not statistically significant when the groups were compared. Stage 2 acute inflammation was detected predominantly in subgroup Ia, namely in 10% (1/10) of patients, which was 2.3 times higher than in subgroup Ib (4.3% (1/23)) and 5.8 times higher than in the control group (1.7% (1/58)); the differences were statistically significant (χ2=4.09, pIa-Ib=0.0431; χ2=4.77, pIa-II=0.0290). Figure 3 shows micrographs of the most typical maternal inflammatory responses.
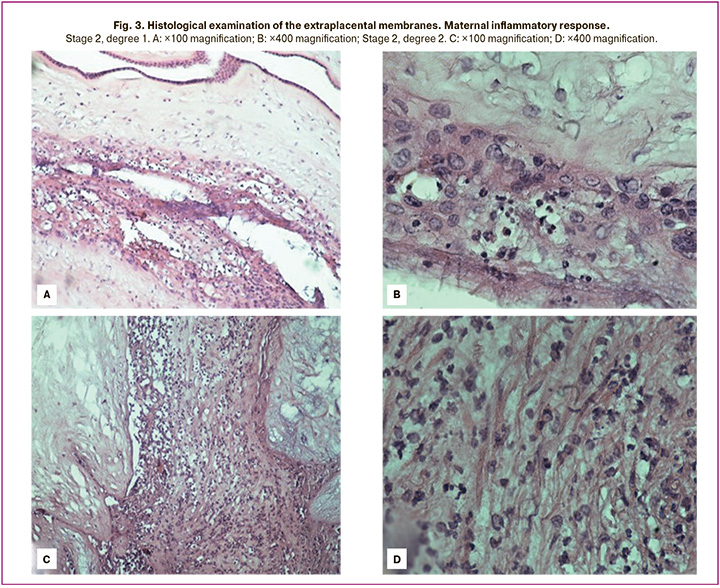
Chronic inflammation represented by chorioamnionitis with mononuclear infiltration was observed in 10% (1/10) of patients in subgroup Ia; it had a low incidence in the control group, 1.7% (1/10), and it was absent in subgroup Ib (χ2=4.83, pIa-Ib=0.0280; χ2=5.43, pIa-II=0.0198).
Thus, signs of both acute and chronic inflammation in the extraplacental membranes were mainly present in the group of patients who had preterm birth with PROM, in comparison with subgroup Ib and the control group.
Discussion
Fetal membranes are a multilayered structure that can experience various mechanical stresses during the course of pregnancy [34]. Fetal membranes are subjected to pressure and stretching as the fetus grows and the volume of amniotic fluid increases. This pressure becomes more intense as labor approaches. The mechanical stresses can contribute to the rupture of membranes, especially in their weakened sections.
Recent studies have examined the relationship between lipid profiles, inflammation, and preterm labor outcomes. The studies of lipid composition have revealed significant changes in the levels of diverse lipid classes, including phospho- and sphingolipids, triacylglycerols and lipid oxidation products, which affect the structural integrity and functional activity of fetal membrane tissues [17, 30, 35–40].
According to our study, lipids belonging to different classes were statistically significant. These lipids are mainly part of bilipid membranes and are responsible for membrane stability and resilience; they are cholesterol esters, ceramides, cardiolipins, diacylglycerols, (lyso-) phosphatidylcholines, simple ester-linked lipids, phosphatidylinositols and glycerols, oxidized lipids, and triacylglycerols.
In the group of patients who had preterm labor with PROM, there was a decrease in the concentrations of the main lipid classes; these results are quite evident, given that the tissue was taken from the site of rupture. PROM is characterized by activation of inflammatory mediators such as cytokines and chemokines, leading to redistribution of lipid molecules involved in inflammatory signaling pathways. Oxidized lipids and sphingolipids can interact with membrane receptors such as Toll-like receptors, enhancing the production of pro-inflammatory molecules [30]. These processes trigger a cascade of inflammatory reactions that contribute to the degradation of the extracellular matrix and impairment of the integrity of the fetal membranes [16, 23].
The results of the lipid profile are confirmed by the results of the histological examination: there was a more intense activation of the inflammatory response in early pregnancy which manifested as the presence of polymorphonuclear neutrophilic infiltration of the fetal membranes; and this may play an important role in preterm labor and PROM. Inflammation can occur due to various mechanisms including cellular senescence and epithelial-mesenchymal junctions, which may contribute to membrane destabilization and onset of labor [23, 41]. According to the literature, rupture of the fetal membranes and preterm labor are most often preceded by the development of infection [6, 42]. In turn, rupture of the fetal membranes for any cause can also predispose to the development of secondary ascending infection [43].
It should be noted that the current international classification of placental lesions does not include the section ‘premature rupture of the membranes’, but it contains the part about inflammatory lesions of the placenta, including changes in the membranes [44].
In addition to inflammatory changes, changes in the lipid profile in PROM are associated with activation of apoptosis, i.e. programmed cell death in amniotic and chorionic tissues. The imbalance of lipids such as phosphatidylserines and ceramides causes the induction of apoptosis by activating caspases and other signaling molecules [18, 19]. These processes lead to the structural weakening of the membranes and increase the risk of rupture.
During pregnancy, especially closer to term, a complex process occurs: the fetal membranes undergo biochemical and mechanical changes that result in a decrease in their strength.
These data are consistent with the results of lipid profile studies in patients with PROM at full-term pregnancy: lysophosphatidylcholine (LPC), lysophosphatidylethanolamine (LPE), ceramides are also structural components of cell membranes and may play a role in cell signaling pathways, as they participate in the regulation of various biological processes such as cell proliferation and apoptosis [11, 27, 28, 30].
Programmed cell death increases in weak areas of the fetal membranes closer to labor. This process leads to the release of bioactive substances that may further stimulate the destruction of collagen fibrils and weakening of the fetal membranes. Therefore, apoptosis not only characterizes tissue restructuring but also contributes to the weakening of membrane density in the rupture area [23, 45, 46].
According to the results of histological examination, the proportion of chronic inflammation in the decidual tissue of the placenta is most represented in the group without PROM, which is probably associated with the process of placental ‘aging’ in normal pregnancy as the gestational age increases. Chronic inflammation can occur due to a variety of mechanisms, which can also contribute to the destabilization of membranes closer to term [47].
Each of the above lipid classes plays a specific role in maintaining the structure, activity, or function of cell membranes. Impairments of their levels or function in the fetal membranes in PROM contribute to membrane degradation, inflammation and increased vulnerability of tissues to mechanical rupture. The lipidomic profile in PROM plays a key role in understanding the pathogenesis of this condition. It has been shown that lipid changes are associated with inflammation and apoptotic activation in fetal membrane cells.
These results indicate a complex relationship between lipid metabolism, inflammation and pathogenetic processes in the fetal membranes at different gestational ages; therefore, further investigation of potential biomarkers and preventive strategies is necessary.
Conclusion
PROM is associated with a combined interaction of biochemical and mechanical changes, such as impaired lipid profile, inflammation and programmed cell death (apoptosis), which lead to destruction of the fetal membranes. The study of the lipid composition of fetal membranes revealed significant changes in the levels of key lipid classes, including oxidized lipids, sphingo- and phospholipids, which play an important role in the development of inflammation and apoptosis. The intensification of these processes disrupts the structure of the membranes, increasing the risk of rupture. Thus, the lipid profile in PROM can be considered not only as a key component of pathogenetic mechanisms, but it can also serve as a potential biomarker for diagnosis and prognosis of this condition, as well as for the development of new approaches in treatment.
References
- Castro D., Norwitz E.R. Preterm premature rupture of membranes. DeckerMed Obstet. Gynecol. 2021. https://dx.doi.org/10.2310/OBG.19056.
- Министерство здравоохранения Российской Федерации. Клинические рекомендации. Преждевременные роды. М.; 2024. 65 с. [Ministry of Health of the Russian Federation. Clinical guidelines. Preterm birth. Moscow; 2024. 65 p. (in Russian)].
- Romero R., Mazor M. Infection and preterm labor. Clin. Obstet. Gynecol. 1988; 31(3): 553-84. https://dx.doi.org/10.1097/00003081-198809000-00006.
- Romero R., Dey S.K., Fisher S.J. Preterm labor: one syndrome, many causes. Science. 2014; 345(6198): 760-5. https://dx.doi.org/10.1126/science.1251816.
- Щеголев А.И., Туманова У.Н., Шувалова М.П., Фролова О.Г. Врожденная пневмония как причина перинатальной смертности в Российской Федерации. Неонатология: новости, мнения, обучение. 2016; 2(12): 61-6. [Shchegolev A.I., Tumanova U.N., Shuvalova M.P., Frolova O.G. Congenital pneumonia as a cause of perinatal mortality in the Russian Federation. Neonatology: News, Opinions, Training. 2016; 2(12): 61-6. (in Russian)].
- Щеголев А.И., Туманова У.Н., Серов В.Н. Хориоамнионит: диагностика и роль в развитии осложнений беременности и плода. Акушерство и гинекология. 2024; 2: 5-14. [Shchegolev A.I., Tumanova U.N., Serov V.N. Chorioamnionitis: diagnosis and role in complications during pregnancy and fetal development. Obstetrics and Gynecology. 2024; (2): 5-14 (in Russian)]. https://dx.doi.org/10.18565/aig.2024.10.
- Щеголев А.И., Павлов К.А., Дубова Е.А., Фролова О.Г. Мертворождаемость в субъектах Российской Федерации в 2010 году. Архив патологии. 2013; 2: 20-4. [Shchegolev A.I., Pavlov K.A., Dubova E.A., Frolova O.G. Stillbirth rates in the subjects of the Russian Federation in 2010. Archiv pathologii. 2013; (2): 20-4 (in Russian)].
- Щеголев А.И., Туманова У.Н., Чаусов А.А., Шувалова М.П. Мертворождение в Российской Федерации в 2020 году (год пандемии COVID-19). Акушерство и гинекология. 2022; 11: 131-40. [Shchegolev A.I., Tumanova U.N., Chausov A.A., Shuvalova M.P. Stillbirths in the Russian Federation in 2020 (COVID-19 pandemic year). Obstetrics and Gynecology. 2022; (11): 131-40. (in Russian)]. https://dx.doi.org/10.18565/aig.2022.11.
- Щеголев А.И., Павлов К.А., Дубова Е.А., Фролова О.Г. Ранняя неонатальная смертность в Российской Федерации в 2010 г. Архив патологии. 2013; 75(4): 15-9. [Shchegolev A.I., Pavlov K.A., Dubova E.A., Frolova O.G. Early neonatal mortality in the Russian Federation in 2010. Arkhiv Patologii. 2013; 75(4): 15-9. (in Russian)].
- Tumanova U.N., Schegolev A.I., Chausov A.A., Shuvalova M.P. Analysis of causes of early neonatal mortality during covid-19 pandemic in 2020 in Russia. Bulletin of RSMU. 2021; (5): 71-7. https://dx.doi.org/10.24075/vrgmu.2021.045.
- Афанасиева М.Х., Болотских В.М., Полякова В.О. Преждевременное излитие околоплодных вод (современные взгляды на этиологию и патогенез, перспективы прогнозирования). Журнал акушерства и женских болезней. 2014; 63(3): 4-11. [Afanasyeva M.Kh., Bolotskikh V.M., Polyakova V.O. Premature rupture of membranes (modern view to etiology and pathogenesis, prediction perspectives). Journal of Obstetrics and Women’s Diseases. 2014; 63(3): 4-11. (in Russian)]. https://dx.doi.org/10.17816/JOWD6334-11.
- Баисова А.Р., Амирасланов Э.Ю., Франкевич В.Е., Чаговец В.В., Тютюнник В.Л. Современные представления об этиологии и патогенезе преждевременного разрыва плодных оболочек. Акушерство и гинекология. 2023; 10: 21-7. [Baisova A.R., Amiraslanov E.Yu., Frankevich V.E., Chagovets V.V., Tyutyunnik V.L. Modern concepts on the etiology and pathogenesis of premature rupture of membranes. Obstetrics and Gynecology. 2023; (10): 21-7. (in Russian)]. https://dx.doi.org/10.18565/aig.2023.199.
- Benyuk V.O., Korniets N.G, Oleshko V.F. Premature rupture of membranes in preterm pregnancy – a modern view on etiology and pathogenesis. Health of woman. 2019; 2(138): 8-13. https://dx.doi.org/10.15574/HW.2019.138.8.
- Luo X., Shi Q., Gu Y., Pan J., Hua M., Liu M. et al. LncRNA pathway involved in premature preterm rupture of membrane (PPROM): an epigenomic approach to study the pathogenesis of reproductive disorders. PLoS One. 2013; 8(11): e79897. https://dx.doi.org/10.1371/journal.pone.0079897.
- Maymon E., Romero R., Pacora P., Gomez R., Athayde N., Edwin S. et al. Human neutrophil collagenase (matrix metalloproteinase 8) in parturition, premature rupture of the membranes, and intrauterine infection. Am. J. Obstet. Gynecol. 2000; 183(1): 94-9. https://dx.doi.org/10.1067/mob.2000.105344.
- Nguyen L.M., Aronoff D.M., Eastman A.J. Matrix metalloproteinases in preterm prelabor rupture of membranes in the setting of chorioamnionitis: A scoping review. Am. J. Reprod. Immunol. 2023; 89(1): e13642. https://dx.doi.org/10.1111/aji.13642.
- Ishutina N.A., Andrievskaya I.A. The role of some lipids and their metabolites in programmed cell death (lipoapoptosis). Acta Biomed. Sci. 2022; 7(4): 12-22. https://dx.doi.org/10.29413/ABS.2022-7.4.2.
- Tang S.H., Tang X., Liu L., Peng L., Zhang H.M., Zhang H. et al. Role of NF-κB p65/TNF-α in cell apoptosis in the fetal membranes of pregnant women with preterm premature rupture of membranes. Ann. Clin. Lab. Sci. 2024; 54(1): 26-34.
- Negara K.S., Suwiyoga K., Sudewi R., Astawa N.M., Arijana G.N.K., Tunas K. et al. The role of caspase-dependent and caspase-independent pathways of apoptosis in the premature rupture of the membranes: A case-control study. Int. J. Reprod. Biomed. 2020; 18(6): 439-48. https://dx.doi.org/10.18502/ijrm.v13i6.7285.
- Li W., Zhao X., Li S., Chen X., Cui H., Chang Y. et al. Upregulation of TNF-α and IL-6 induces preterm premature rupture of membranes by activation of ADAMTS-9 in embryonic membrane cells. Life Sci. 2020; 260: 118237. https://dx.doi.org/10.1016/j.lfs.2020.118237.
- Zahorodnia O.S., Motsyuk Yu.B., Amerkhanova T.V. Labour as a manifestation of systemic inflammatory reaction (literature review). Reproductive Health of Woman. 2023; (4): 79-84. https://dx.doi.org/10.30841/2708-8731.4.2023.285769.
- Gao Y., Mi N., Zhang Y., Li X., Guan W., Bai C. Uterine macrophages as treatment targets for therapy of premature rupture of membranes by modified ADSC-EVs through a circRNA/miRNA/NF-κB pathway. J. Nanobiotechnology. 2022; 20(1): 487. https://dx.doi.org/10.1186/s12951-022-01696-z.
- Menon R., Behnia F., Polettini J., Richardson L.S. Novel pathways of inflammation in human fetal membranes associated with preterm birth and preterm pre-labor rupture of the membranes. Semin. Immunopathol. 2020; 42(4): 431-50. https://dx.doi.org/10.1007/s00281-020-00808-x.
- Choltus H., Lavergne M., De Sousa Do Outeiro C., Coste K., Belville C., Blanchon L. et al. Pathophysiological implication of pattern recognition receptors in fetal membranes rupture: RAGE and NLRP inflammasome. Biomedicines. 2021; 9(9): 1123. https://dx.doi.org/10.3390/biomedicines9091123.
- Choltus H., Lavergne M., Belville C., Gallot D., Minet-Quinard R., Durif J. et al. Occurrence of a RAGE-Mediated inflammatory response in human fetal membranes. Front. Physiol. 2020; 11: 581. https://dx.doi.org/10.3389/fphys.2020.00581.
- Yan H., Zhu L., Zhang Z., Li H., Li P., Wang Y. et al. HMGB1-RAGE signaling pathway in pPROM. Taiwan J. Obstet. Gynecol. 2018; 57(2): 211-6. https://dx.doi.org/10.1016/j.tjog.2018.02.008.
- Agmon E., Stockwell B.R. Lipid homeostasis and regulated cell death. Curr. Opin. Chem. Biol. 2017; 39: 83-9. https://dx.doi.org/10.1016/J.CBPA.2017.06.002.
- Flores-Romero H., Ros U., García-Sáez A.J. A lipid perspective on regulated cell death. Int. Rev. Cell. Mol Biol. 2020; 351: 197-236. https://dx.doi.org/10.1016/BS.IRCMB.2019.11.004.
- Wahid H.H., Anahar F.N., Isahak N.H., Mohd Zoharodzi J., Mohammad Khoiri S.N.L., Mohamad Zainal N.H. et al. Role of platelet activating factor as a mediator of inflammatory diseases and preterm delivery. Am. J. Pathol. 2024; 194(6): 862-78. https://dx.doi.org/10.1016/j.ajpath.2024.01.018.
- Кытикова О.Ю., Новгородцева Т.П., Денисенко Ю.К., Антонюк М.В., Гвозденко Т.А. Роль липидов в механизмах сигнализации толл-подобных рецепторов. Вестник РАМН. 2020; 75(6): 585-93. [Kytikovа O.Yu., Novgorodtseva T.P., Denisenko Yu.K., Antonyuk M.V., Gvozdenko T.A. The role of lipids in the signaling mechanisms of toll-like receptors. Annals of the Russian Academy of Medical Sciences. 2020; 75(6): 585-93. (in Russian)]. https://dx.doi.org/10.15690/vramn1179.
- Министерство здравоохранения Российской Федерации. Клинические рекомендации. Нормальная беременность. М.; 2024. 90 с. [Ministry of Health of the Russian Federation. Clinical guidelines. Normal pregnancy. Moscow; 2024. 90 p. (in Russian)].
- Redline R.W., Faye-Petersen O., Heller D., Qureshi F., Savell V., Vogler C.; Society for Pediatric Pathology, Perinatal Section, Amniotic Fluid Infection Nosology Committee. Amniotic infection syndrome: nosology and reproducibility of placental reaction patterns. Pediatr. Dev. Pathol. 2003; 6(5): 435-48. https://dx.doi.org/10.1007/s10024-003-7070-y.
- Khong T.Y., Mooney E.E., Ariel I., Balmus N.C., Boyd T.K., Brundler M.A. et al. Sampling and definitions of placental lesions: Amsterdam Placental Workshop Group Consensus Statement. Arch. Pathol. Lab. Med. 2016; 140(7): 698-713. https://dx.doi.org/10.5858/arpa.2015-0225-CC.
- Щеголев А.И., Дубова Е.А., Павлов К.А. Морфология плаценты. М.; 2010. 46 с. [Shchegolev A.I., Dubova E.A., Pavlov K.A. Morphology of the placenta. Moscow; 2010. 46 p. (in Russian)].
- Zhao Q., Ma Z., Wang X., Liang M., Wang W., Su F. et al. Lipidomic biomarkers of extracellular vesicles for the prediction of preterm birth in the early second trimester. J. Proteome Res. 2020; 19(10): 4104-13. https://dx.doi.org/10.1021/acs.jproteome.0c00525.
- Aung M.T., Ashrap P., Watkins D.J., Mukherjee B., Rosario Z., Vélez-Vega C.M. et al. Maternal lipidomic signatures in relation to spontaneous preterm birth and large-for-gestational age neonates. Sci. Rep. 2021; 11(1): 8115. https://dx.doi.org/10.1038/s41598-021-87472-9.
- Maddipati K.R., Romero R., Chaiworapongsa T., Chaemsaithong P., Zhou S.L., Xu Z. et al. Lipidomic analysis of patients with microbial invasion of the amniotic cavity reveals up-regulation of leukotriene B4. FASEB J. 2016; 30(10): 3296-307. https://dx.doi.org/10.1096/fj.201600583R.
- Hong S.H., Lee J.Y., Seo S., Shin B., Jeong C.H., Bae E. et al. Lipidomic analysis of cervicovaginal fluid for elucidating prognostic biomarkers and relevant phospholipid and sphingolipid pathways in preterm birth. Metabolites. 2023; 13(2): 177. https://dx.doi.org/10.3390/metabo13020177.
- Morillon A.C., Yakkundi S., Thomas G., Gethings L.A., Langridge J.I., Baker P.N. et al. Association between phospholipid metabolism in plasma and spontaneous preterm birth: a discovery lipidomic analysis in the cork pregnancy cohort. Metabolomics. 2020; 16(2): 19. https://dx.doi.org/10.1007/s11306-020-1639-6.
- Makide K., Kitamura H., Sato Y., Okutani M., Aoki J. Emerging lysophospholipid mediators, lysophosphatidylserine, lysophosphatidylthreonine, lysophosphatidylethanolamine and lysophosphatidylglycerol. Prostaglandins Other Lipid Mediat. 2009; 89 (3-4): 135-9. https://dx.doi.org/10.1016/j.prostaglandins.2009.04.009.
- Musilova I., Kutová R., Pliskova L., Stepan M., Menon R., Jacobsson B. et al. Intraamniotic inflammation in women with preterm prelabor rupture of membranes. PLoS One. 2015; 10(7): e0133929. https://dx.doi.org/10.1371/journal.pone.0133929.
- Ogunyemi D., Murillo M., Jackson U., Hunter N., Alperson B. The relationship between placental histopathology findings and perinatal outcome in preterm infants. J. Matern. Fetal Neonatal. Med. 2003; 13(2): 102-9. https://dx.doi.org/10.1080/jmf.13.2.102.109.
- Щеголев А.И., Серов В.Н. Клиническая значимость поражений плаценты. Акушерство и гинекология. 2019; 3: 54-62. [Shchegolev A.I., Serov V.N. Clinical significance of placental lesions. Obstetrics and Gynecology. 2019; (3): 54-62. (in Russian)]. https://dx.doi.org/10.18565/aig.2019.3.54-62.
- Щеголев А.И. Современная морфологическая классификация повреждений плаценты. Акушерство и гинекология. 2016; 4: 16-23. [Shchegolev A.I. Current morphological classification of damages to the placenta. Obstetrics and Gynecology. 2016; (4): 16-23. (in Russian)]. http://dx.doi.org/10.18565/aig.2016.4.16-23.
- Menon R., Richardson L.S., Lappas M. Fetal membrane architecture, aging and inflammation in pregnancy and parturition. Placenta. 2019; 79: 40-5. https://dx.doi.org/10.1016/j.placenta.2018.11.003.
- Richardson L.S., Radnaa E., Urrabaz-Garza R., Lavu N., Menon R. Stretch, scratch, and stress: Suppressors and supporters of senescence in human fetal membranes. Placenta. 2020; 99: 27-34. https://dx.doi.org/10.1016/j.placenta.2020.07.013.
- Carroll A., Lee S., Desforges M., Heazell A. A review of the evidence for placental ageing in prolonged pregnancy. Med. Res. Arch. 2020; 8(8). https://dx.doi.org/10.18103/mra.v8i8.2188.
Received 17.02.2025
Accepted 25.02.2025
About the Authors
Almira R. Baisova, PhD student, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, +7(932)099-10-04, almira.baisova@mail.ru, https://orcid.org/0009-0004-4546-2388Elrad Yu. Amiraslanov, PhD, Head of the Department of Obstetrics, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, eldis@mail.ru, https://orcid.org/0000-0001-5601-1241
Vladimir E. Frankevich, Dr. Sci. (physico-mathematical), Head of the Department of Systems Biology in Reproduction, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4; Leading Researcher at the Laboratory
of Translational Medicine, Siberian State Medical University, Ministry of Health of Russia, 634050, Russia, Tomsk, Moskovsky tract, 2, v_frankevich@oparina4.ru,
https://orcid.org/0000-0002-9780-4579
Vitaliy V. Chagovets, PhD, Senior Researcher at Proteomics of Human Reproduction Department, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, v_chagovets@oparina4.ru, https://orcid.org/0000-0002-5120-376X
Alisa O. Tokareva, PhD, Specialist at the Laboratory of Clinical Proteomics, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, alisa.tokareva@phystech.edu, https://orcid.org/0000-0001-5918-9045
Galina V. Kulikova, PhD, Senior Researcher at the Pathology and Anatomical Department, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, g_kulikova@oparina4.ru, Researcher ID: G-6159-2014,
Scopus Author ID: 7003966947, https://orcid.org/0000-0003-0594-955X
Alexander I. Shchegolev, Dr. Med. Sci., Professor, Head of the 2nd Pathology Department, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, ashegolev@oparina4.ru, https://orcid.org/0000-0002-2111-1530
Natalia E. Kan, Professor, Dr. Med. Sci., Honored Scientist of the Russian Federation, Deputy Director for Research – Director of the Institute of Obstetrics,
Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow,
Ac. Oparin str., 4, kan-med@mail.ru, Researcher ID: B-2370-2015, SPIN code: 5378-8437, Authors ID: 624900, Scopus Author ID: 57008835600,
https://orcid.org/0000-0001-5087-5946
Victor L. Tyutyunnik, Professor, Dr. Med. Sci., Leading Researcher at the Center for Scientific and Clinical Research, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, tioutiounnik@mail.ru,
Researcher ID: B-2364-2015, SPIN-code: 1963-1359, Authors ID: 213217, Scopus Author ID: 56190621500, https://orcid.org/0000-0002-5830-5099
Corresponding author: Almira R. Baisova, almira.baisova@mail.ru



