A model for diagnosing fetal growth restriction using functional diagnostic methods
Volochaeva M.V., Timofeeva A.V., Fedorov I.S., Kan N.E., Tyutyunnik V.L., Ryzhova K.O., Gasymova Sh.R.
Objective: To enhance the differential diagnosis of fetal growth restriction (FGR) and small-for-gestational-age (SGA) fetuses using functional diagnostic methods.
Materials and methods: This case-control study included 13,232 patients who were divided into three groups. Group 1 comprised pregnant women with FGR (n=242), group 2 included pregnant women with SGA (n=395), and group 3 consisted of pregnant women without FGR and SGA (n=12,595). Postnatally, neonatal length and height indicators were assessed according to the INTERGROWTH-21 centile curve. The sample for model development and training consisted of 13,232 patients, while a test sample of 1,843 patients was used to evaluate the model's performance.
Results: The developed formula (1), which includes fetal abdominal circumference (mm) (AC), right uterine artery pulsatility index (RUtA-PI) (percentile), and cerebro-placental ratio (CPR) (percentile), enabled the diagnosis of FGR with a sensitivity of 84% and a specificity of 82%. To differentiate between SGA and FGR, a second formula (2) was created, incorporating AC (mm), uterine artery pulsatility index (UA PI) (percentile), and middle cerebral artery pulsatility index (MCA PI) (percentile). This formula allows for the diagnosis of SGA as distinct from FGR, with a sensitivity of 67% and specificity of 88%.
Conclusion: The developed formulas facilitate the diagnosis of FGR and SGA, enabling timely decision-making regarding pregnancy management strategies. This approach aims to reduce the incidence of iatrogenic interventions, neonatal morbidity, and mortality while improving the quality of medical care.
Authors' contributions: Volochaeva M.V., Timofeeva A.V., Fedorov I.S., Kan N.E., Tyutyunnik V.L., Ryzhova K.O.,
Gasymova Sh.R. – conception and design of the study, acquisition of data for analysis, review of relevant literature, material processing and analysis, statistical analysis, drafting of the manuscript, editing of the manuscript.
Conflicts of interest: The authors have no conflicts of interest to declare.
Funding: The study was conducted within the framework of the initiative project «Epigenetic Criteria for Diagnosing Fetal Growth Delay from the Perspective of Neurogenesis Dysfunction». Research Project No. 19-И23 dated December 8, 2022. Registration number in the EGISU NIOKTR system (state accounting) – 123060500032-8.
Ethical Approval: The study was reviewed and approved by the Research Ethics Committee of the V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia.
Patient Consent for Publication: All patients provided informed consent for the publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available upon request from the corresponding author after approval from the principal investigator.
For citation: Volochaeva M.V., Timofeeva A.V., Fedorov I.S., Kan N.E., Tyutyunnik V.L., Ryzhova K.O.,
Gasymova Sh.R. A model for diagnosing fetal growth restriction using functional diagnostic methods.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2025; (2): 31-39 (in Russian)
https://dx.doi.org/10.18565/aig.2025.15
Keywords
The prediction and diagnosis of fetal growth restriction (FGR) remains one of the most studied and unresolved issues in modern obstetrics. FGR is associated with an increased incidence of stillbirths, neonatal morbidity, and mortality [1, 2], as well as future health complications, including an elevated risk of neurologic diseases (characterized by decreased motor and cognitive performance due to reduced brain volume and cell death) and non-infectious chronic conditions (such as metabolic syndrome, obesity, and cardiovascular pathology), compared to children born with normal weight, ultimately leading to a decreased life expectancy [3, 4].
Diagnosis of FGR is based on clinical and functional investigations. The contemporary definition of FGR and low birth weight (SGA) hinges on the fetus's inability to achieve its genetically determined growth potential. It is widely accepted that a diagnosis is made when the estimated fetal weight falls below the 10th percentile; for FGR, this is accompanied by abnormal Doppler measurements, whereas for SGA, it occurs without abnormal blood flow in the uteroplacental and fetoplacental complex. The gestational age at which FGR is diagnosed allows for the classification of two clinically relevant phenotypes: early and late (before and after 32 weeks). This classification serves as a criterion for assessing the severity of FGR, guiding management strategies, and highlighting differences in pathogenesis [5, 6]. Notably, early FGR (diagnosed before 32 weeks) is identified more frequently than late FGR or SGA, when proper monitoring is in place. Currently, ultrasound and Doppler ultrasonography, along with the application of the Delphi criteria [7], are standard methods for diagnosing FGR and SGA. Although these techniques undoubtedly enhance the detection rates of at-risk fetuses, they also contribute to an increase in false-positive and false-negative results.
In obstetrics, the terms "fetus with FGR" and "fetus with SGA" are often used interchangeably; however, clinicians tend to underestimate the significance of SGA, which is frequently associated with a healthy, constitutionally small fetus. The primary challenge in diagnosing fetuses with FGR and SGA lies in the timely detection and differentiation of these conditions, underscoring the need to enhance ultrasound screening by developing more sensitive and specific tests through functional diagnostic methods.
This study aimed to improve the differential diagnosis of FGR and SGA fetuses by using functional diagnostic methods.
Materials and methods
The development and training of formulas for FGR and SGA diagnosis was performed using machine learning. This case-control study was carried out from 2021 to 2023 at the obstetric departments and ultrasound diagnostics department of the V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia. Based on the inclusion and non-inclusion criteria, 13,232 patients were included in the final sample, which was divided into three groups: group 1 comprised pregnant women with FGR (postnatally, the child was small for gestational age) (n=242); group 2 included pregnant women with SGA (postnatally, the child was underweight for gestational age) (n=395); and group 3 consisted of pregnant women without FGR and SGA (postnatally, the child appropriate for gestational age) (n=12,595).
The inclusion criteria for the study were as follows: age between 18 and 45 years, singleton pregnancy, and observation and delivery between 24 and 42 weeks of gestation at I. Kulakov NMRC for OG&P. Patients underwent ultrasound and Doppler examinations, which included assessments of abdominal circumference (AC) (mm, percentiles), determination of the uterine artery pulsatility index (UA PI), umbilical artery pulsatility index (UmA PI), and middle cerebral artery pulsatility index (MCA PI) (percentiles), as well as estimation of the cerebral-placental ratio (CPR) (percentiles) using Fetal Medicine Foundation calculators to convert the values of pulsatility indices into a centile score. The non-inclusion criteria included multiple pregnancies and incomplete ultrasound and Doppler assessments.
At the antenatal stage, the diagnoses of "fetal growth restriction," "fetus underweight for gestational age," and "fetus appropriate for gestational age" were stratified according to the Delphi diagnostic criteria [7] and the clinical guidelines of the Russian Ministry of Health "Fetal growth restriction requiring medical care for the mother" (fetal growth restriction)" [8]. Postnatally, all cases were evaluated according to the INTERGROWTH-21 centile curves to confirm the antenatal diagnosis of FGR and SGA as well as to verify normal body weight.
The study was reviewed and approved by the Research Ethics Committee of V.I. Kulakov NMRC for OG&P, and all patients provided informed consent.
Statistical analysis
The normality of the distribution of continuous variables was tested using the Shapiro–Wilk test (for samples with fewer than 50 subjects) or the Kolmogorov–Smirnov test (for samples with 50 or more subjects). Continuous variables with a normal distribution were described using arithmetic mean (M), standard deviation (SD), and 95% confidence interval (95% CI). Non-normally distributed variables were described using the median (Me) and the lower and upper quartiles [Q1, Q3]. Categorical variables were presented as counts and percentages. The 95% CI for percentages was calculated using the Klopper–Pearson method. Comparisons of three or more groups for continuous variables with a normal distribution were performed using one-way analysis of variance, with post-hoc comparisons conducted using Tukey's test (assuming equal variance) or the Games–Howell test (assuming unequal variance). Comparisons of three or more groups for non-normally distributed variables were performed using the Kruskal–Wallis test, with post-hoc comparisons using Dunn's test with Holm’s correction. Comparisons of percentages in the multi-way contingency table analyses were conducted using Pearson's chi-square test, with post-hoc comparisons using Pearson's chi-square test with Holm’s correction. Differences were considered statistically significant at p<0.05.
Results
The data from the complex analysis of ultrasound, Doppler examination, and length and weight indicators of newborns at the time of delivery in the study groups are presented in Table 1. The median period of ultrasound and Doppler examination in group 1 was 258.00 [245.25; 269.00] days, in group 2 – 265.00 [256.50; 273.00] days, in group 3 – 266.00 [255.00; 274.00] days (p<0.001). Significant differences were found between the groups when comparing the AC (p<0.001) and estimated weight of the fetus (p<0.001). When assessing Doppler parameters, significant differences were observed when comparing pulsatility indices in the left and right uterine arteries (p<0.001 and p=0.008, respectively), umbilical artery (p<0.001), middle cerebral artery (p<0.001), and CPR (p<0.001). When assessing the length and weight indicators of newborns, the groups showed significant differences in birth weight (p<0.001), length (p<0.001), and head circumference (p<0.001).
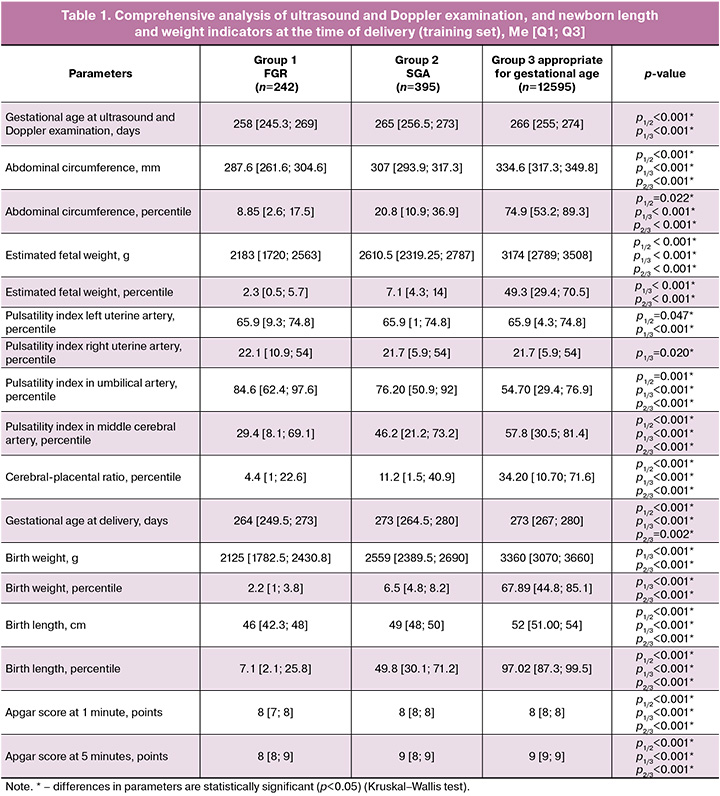
A comparative analysis of antenatal and postnatal diagnostics of early and late FGR was conducted using the criteria of the international Delphi consensus, postnatal assessment of newborns according to the INTERGROWTH-21 centile tables [9, 10] (Table 2).
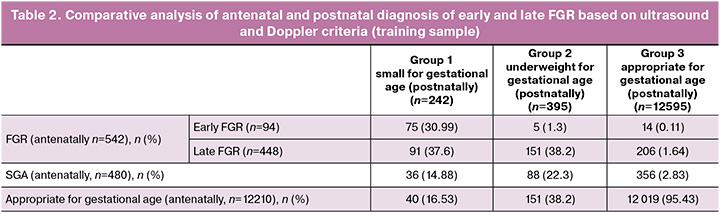
FGR was diagnosed in 542 cases at the antenatal stage, which was 2.2 times higher than the number of postnatally confirmed SGA births, indicating overdiagnosis of this pregnancy complication. A similar trend was noted during the SGA verification (1.2 times).
The diagnosis of FGR was established in 68.59% of observations (166/242) ( early FGR in 79.9% of observations, late FGR in 20.3% of observations). However, in every third case, underestimation of ultrasound and Doppler characteristics was noted (FGR was not detected in 31.41% of observations (76/242): 16.53% of fetuses were assessed as appropriate for gestational age (40/242) and 14.88% as SGA (36/242)). In the "low birth weight for gestational age" group (postnatally), only 22.3% of fetuses were correctly diagnosed antenatally (88/395). SGA was not suspected in 77.7% of cases (307/395). It should be noted that the maximum accuracy of ultrasound and Doppler diagnostics was noted in the group of newborns "appropriate for gestational age,” where the coincidence of ante- and postnatal assessment of the length and weight indicators of the newborn was 95.43% of cases (12019/12595). In the RStudio program, the method of stepwise inclusion and exclusion using machine learning was used to find optimal combinations of functional research method indicators associated with the diagnosis of FGR or SGA (ultrasound and Doppler criteria used according to the Delphi consensus to verify the diagnosis of early, late FGR and SGA), in accordance with their contribution to the construction of logistic regression models, where the dependent variable (response variable) was the absence or presence of pregnancy complications confirmed by postnatal assessment of length and weight indicators in newborns according to the INTERGROWTH-21 centile curves, Russian and international neonatal criteria for making the diagnoses of "small for gestational age" and "low birth weight for gestational age " (0 – no complication; 1 – presence of complication). The following models were created to allow the diagnosis of FGR based on the norm (Fig. 1A), and FGR from SGA (Fig. 1B).
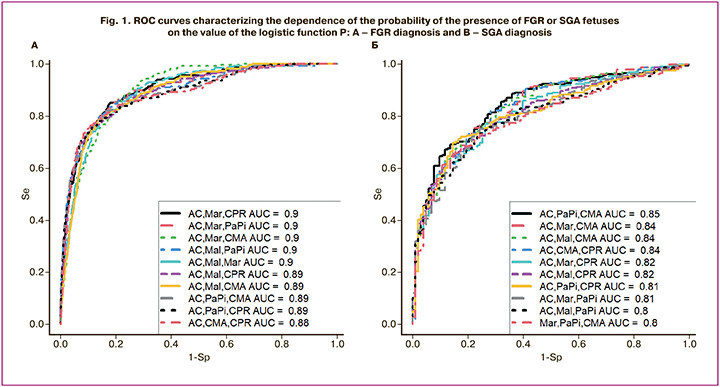
Formula (1), including AC (mm), RUtA-PI (percentile), CPR (percentile), allows diagnosing FGR with a sensitivity of 84% and a specificity of 82% (Fig. 2).
The observed relationship can be described by the following equation:
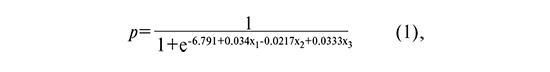
where p is the probability of FGR, x1 is the fetal AC (mm), x2 is the RUtA-PI (percentile), and x3 is the CPR (percentile).
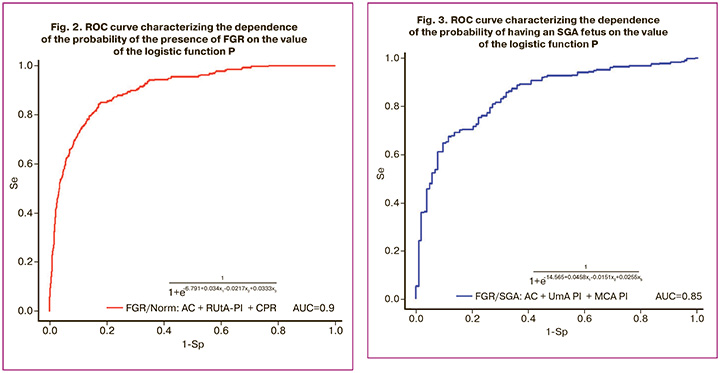
The cut-off level (p) was 0.0247. At p<0.0247, a conclusion is made regarding the absence of FGR, and at p>0.0247, regarding the presence of FGR.
For the differential diagnosis between SGA and FGR, formula (2) was created, including AC (mm), UmA PI (percentile), and MCA PI, allowing the diagnosis of SGA from FGR with a sensitivity of 67% and a specificity of 88% (Fig. 3).
The observed relationship can be described by the following equation:
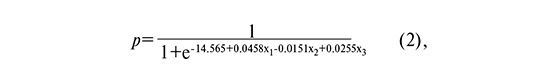
where p is the probability of SGA, x1 is the fetal AC (mm), x2 is the UmA PI (percentile), and x3 is the MCA PI (percentile).
The cut-off level (p) was 0.7733. At p<0.7733, a conclusion is made that the fetus is SGA, at p>0.7733, FGR is observed.
The next stage of the study was the formation of a test sample, which included cases of discrepancy between the antenatal diagnosis of FGR and the fetus appropriate for gestational age according to the postnatal criteria for assessing the length and weight indicators of newborns INTERGROWTH-21 (cases of hypo- or hyper-diagnosis). The test sample consisted of 1843 pregnant women with a singleton pregnancy who were also observed and delivered at V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia from 2019 to 2023. A comprehensive analysis of ultrasound and Doppler findings of the fetus and the length and weight indicators of newborns in the study groups is presented in Table 3.
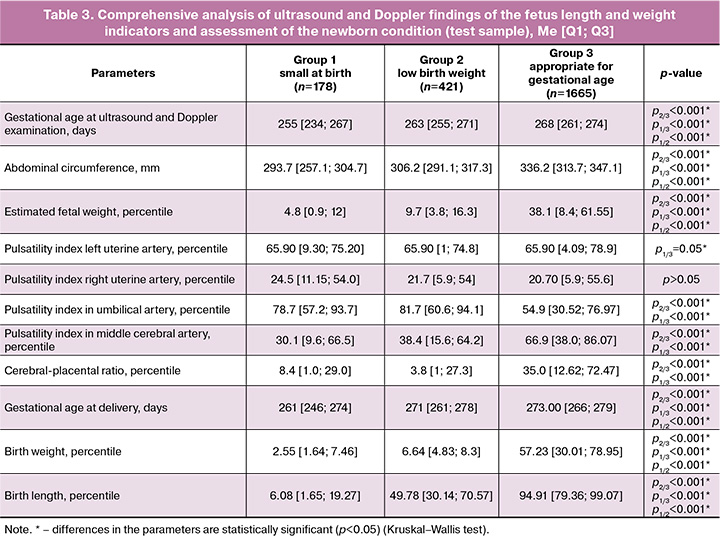
In the test sample, FGR was postnatally confirmed in 178 newborns. A comparative analysis of antenatal and postnatal FGR diagnoses based on ultrasound and Doppler criteria revealed the following findings: antenatally, using routine ultrasound/Doppler, the FGR detection rate was 22.5% (40/178) for fetuses appropriate for gestational age of 60% (1000/1665). The antenatal FGR detection rate using the proposed model was 61% (108/178), of fetuses appropriate for gestational age – 76% (1258/1665). The implementation of the developed model will improve the accuracy of FGR diagnostics by reducing the number of false-negative and false-positive results by 38.5% and 15.9 %, respectively.
Discussion
The relevance of this study lies in the pursuit of ultrasound markers that improve the efficiency of intrauterine fetal growth parameter assessment. The ultimate goal is to reduce the frequency of false-positive and false-negative results as well as the incidence of iatrogenic interventions and delayed provision of qualified medical care.
Various models for predicting and diagnosing FGR have been developed based on the clinical characteristics of pregnant women, ultrasound data, Doppler studies, and placental markers. However, their applications remain limited [11]. Ultrasound and Doppler examinations are the primary standards for diagnosing FGR and SGA phenotypes [7, 8]. Notably, there is currently no universally accepted growth chart or specific ultrasound indicator that distinguishes between a fetus with "normal" growth and one with pathological growth, contributing to delayed diagnosis [12]. The only "gold" standard for confirming these pregnancy complications is the assessment of birth weight and height indicators of children according to the INTERGROWTH-21 standards, along with neonatal protocols based on these standards [9].
According to Benson-Cooper S. et al. [13] and Dudley N.J. et al. [14], the primary challenge in conducting ultrasound diagnostics for the early detection of FGR is the tendency to underestimate fetal weight. This underestimation hampers timely identification of FGR, increasing the risk of antenatal death, neonatal morbidity, and mortality. Ridha N. et al. [15] further note that the accuracy of ultrasound diagnostics diminishes when detecting SGA fetuses.
The analysis revealed that at the antenatal stage of observation, there was an issue of overdiagnosis for both FGR (2.2 times more frequent compared to postnatal assessments) and SGA (1.2 times more frequent compared to postnatal assessments). The frequency of FGR diagnosis was 68.59% (early FGR: 79.9%; late FGR: 20.3%), whereas SGA was diagnosed at a rate of 22.3%. These findings are consistent with those of Miranda J. et al. [16], which indicate that ultrasound examination in the third trimester of pregnancy for assessing fetal biometry parameters has a sensitivity of 52%–57% for diagnosing FGR. In contrast, Haragan A. et al. [17] and Relph S. et al. [18] suggested that 75–78.7% of FGR or SGA cases are not diagnosed antenatally until delivery.
MacDonald T.M. et al. [19] and Rizzo G. et al. [20] demonstrated the diagnostic potential of combined Doppler parameters in predicting FGR and adverse perinatal outcomes, serving as prototypes for the presented method of diagnosing FGR. However, these methods also have significant limitations. For example, MacDonald T.M. et al. (n=347) noted a strong correlation between cerebroplacental-uterine ratio and FGR at birth. Nevertheless, the applicability of this parameter is constrained by gestational age (36 weeks) and the lack of a test sample, making it unsuitable for diagnosing early FGR [19]. Conversely, Rizzo G. et al. (n=243) found a better diagnostic value in determining the ratio of blood flow velocity in the umbilical artery to fetal AC (AUC=0.723) than in determining CPR and/or PI MA (AUC=0.6) in late FGR [20]. The small sample size and lack of validation in these studies limits the reliability of the results. Furthermore, studies by MacDonald T.M. et al. and Rizzo G. et al. inadequately addressed the problem of diagnosing FGR and necessitated improved methodology, including larger sample sizes and validation with independent samples. To predict the development of FGR during pregnancy, obstetric practice employs biochemical markers (such as pregnancy-associated protein A (PAPP-A), β-human chorionic gonadotropin, α-fetoprotein, placental growth factor, and soluble fms-like tyrosine kinase-1) in conjunction with ultrasound data [21, 22]. This underscores the need to optimize ultrasound diagnostic approaches and introduce new noninvasive and/or minimally invasive molecular methods for verifying diagnoses and differentiating between FGR, SGA, and fetuses that appropriate for gestational age. We recommend combining ultrasound and molecular biological indicators to create a comprehensive algorithm for diagnosing FGR and SGA. The application of omics technology offers new opportunities to study the pathogenesis of FGR, potentially enhancing the prediction, diagnosis, and treatment of this pregnancy complication.
Conclusion
The developed formulas enable a highly accurate diagnosis of FGR or SGA, facilitating timely decisions regarding pregnancy management strategies and delivery timing. This approach aims to reduce the incidence of iatrogenic interventions and neonatal morbidity and mortality, ultimately enhancing the quality of medical care.
References
- Jarvis S., Glinianaia S.V., Torrioli M.G., Platt M.J., Miceli M., Jouk P.S. et al.; Surveillance of Cerebral Palsy in Europe (SCPE) collaboration of European Cerebral Palsy Registers. Cerebral palsy and intrauterine growth in single births: European collaborative study. Lancet. 2003; 362(9390): 1106-11. https://dx.doi.org/10.1016/S0140-6736(03)14466-2.
- Солдатова Е.Е., Кан Н.Е., Тютюнник В.Л., Волочаева М.В. Задержка роста плода с позиции фетального программирования. Акушерство и гинекология. 2022; 8: 5-10. [Soldatova E.E., Kan N.E., Tyutyunnik V.L., Volochaeva M.V. Fetal growth retardation in the context of fetal programming. Obstetrics and Gynecology. 2022; (8): 5-10. (in Russian)]. https://dx.doi.org/10.18565/aig.2022.8.5-10.
- Blair E.M., Nelson K.B. Fetal growth restriction and risk of cerebral palsy in singletons born after at least 35 weeks’ gestation. Am. J. Obstet. Gynecol. 2015; 212(520): e521-e527. https://dx.doi.org/10.1016/j.ajog.2014.10.1103.
- Misan N., Michalak S., Kapska K., Osztynowicz K., Ropacka-Lesiak M. Blood-brain barrier disintegration in growth-restricted fetuses with brain sparing effect. Int. J. Mol. Sci. 2022; 23(20): 12349. https://dx.doi.org/10.3390/ijms232012349.
- Lees C.C., Romero R., Stampalija T., Dall'Asta A., DeVore G.A., Prefumo F. et al. Clinical Opinion: The diagnosis and management of suspected fetal growth restriction: an evidence-based approach. Am. J. Obstet. Gynecol. 2022; 226(3): 366-78. https://dx.doi.org/10.1016/j.ajog.2021.11.1357.
- McCowan L.M., Figueras F., Anderson N.H. Evidence-based national guidelines for the management of suspected fetal growth restriction: comparison, consensus, and controversy. Am. J. Obstet. Gynecol. 2018; 218(2S): S855-S868. https://dx.doi.org/10.1016/j.ajog.2017.12.004.
- Gordijn S.J., Beune I.M., Thilaganathan B., Papageorghiou A., Baschat A.A., Baker P.N. et al. Consensus definition of fetal growth restriction: a Delphi procedure. Ultrasound Obstet. Gynecol. 2016; 48(3): 333-9. https://dx.doi.org/10.1002/uog.15884.
- Министерство здравоохранения Российской Федерации. Клинические рекомендации. Недостаточный рост плода, требующий предоставления медицинской помощи матери (задержка роста плода). М.; 2022. 71 с. [Ministry of Health of the Russian Federation. Clinical guidelines. Insufficient growth of the fetus, requiring the provision of medical care to the mother (fetal growth retardation). Moscow; 2022. 71 p. (in Russian)].
- Leite D.F.B., de Melo E.F. Jr., Souza R.T., Kenny L.C., Cecatti J.G. Fetal and neonatal growth restriction: new criteria, renew challenges. J. Pediatr. 2018; 203: 462-3. https://dx.doi.org/10.1016/j.jpeds.2018.07.094.
- Рюмина И.И., Байбарина Е.Н., Нароган М.В., Маркелова М.М., Орловская И.В., Зубков В.В., Дегтярев Д.Н. Использование международных стандартов роста для оценки физического развития новорожденных и недоношенных детей. Неонатология: новости, мнения, обучение. 2023; 11(2): 48-52. [Ryumina I.I., Baibarina E.N., Narogan M.V., Markelova M.M., Orlovskaya I.V., Zubkov V.V., Degtyarev D.N. Using international growth standards to assess the physical development of newborns and premature infants. Neonatology: News, Opinions, Training. 2023; 11(2): 48-52. (in Russian)]. https://dx.doi.org/10.33029/2308-2402-2023-11-2-48-52.
- Feng Y., Zheng H., Fang D., Mei S., Zhong W., Zhang G. Prediction of late-onset fetal growth restriction using a combined first- and second-trimester screening model. J. Gynecol. Obstet. Hum. Reprod. 2022; 51(2): 102273. https://dx.doi.org/10.1016/j.jogoh.2021.102273.
- Leite D.F.B., Cecatti J.G. Fetal growth restriction prediction: How to move beyond. ScientificWorldJournal. 2019; 2019: 1519048. https://dx.doi.org/10.1155/2019/1519048.
- Benson-Cooper S., Tarr G.P., Kelly J., Bergin C.J. Accuracy of ultrasound in estimating fetal weight in New Zealand. Australas J. Ultrasound Med. 2021; 24(1): 13-9. https://dx.doi.org/10.1002/ajum.12239.
- Dudley N.J., Jindal S., Varley H. An evaluation of systematic and random errors in ultrasound estimated fetal weight during serial ultrasound. Ultrasound. 2023; 31(4): 259-65. https://dx.doi.org/10.1177/1742271X221139796.
- Ridha N., Bergin C.J., Kelly J., Tarr G.P., Anderson N., Sadler L. Accuracy of ultrasound in the estimation of customised birth weight in a public hospital service. J. Med. Imaging Radiat. Oncol. 2022; 66(8): 1044-51. https://dx.doi.org/10.1111/1754-9485.13408.
- Miranda J., Rodriguez‐Lopez M., Triunfo S., Sairanen M., Kouru H., Parra‐Saavedra M. et al. Prediction of fetal growth restriction using estimated fetal weight vs a combined screening model in the third trimester. Ultrasound Obstet. Gynecol. 2017; 50(5): 603-11. https://dx.doi.org/10.1002/uog.17393.
- Haragan A., Himes K. Accuracy of ultrasound estimated fetal weight in small for gestational age and appropriate for gestational age grown periviable neonates. Am. J. Perinatol. 2018; 35(8): 703-6. https://dx.doi.org/10.1055/s-0037-1617433.
- Relph S., Vieira M.C., Copas A., Alagna A., Page L., Winsloe C. et al.; DESiGN Trial Team and DESiGN Collaborative Group. Characteristics associated with antenatally unidentified small-for-gestational-age fetuses: prospective cohort study nested within DESiGN randomized controlled trial. Ultrasound Obstet. Gynecol. 2023; 61(3): 356-66. https://dx.doi.org/10.1002/uog.26091.
- MacDonald T.M., Hui L., Robinson A.J., Dane K.M., Middleton A.L., Tong S. et al. Cerebral-placental-uterine ratio as novel predictor of late fetal growth restriction: prospective cohort study. Ultrasound Obstet. Gynecol. 2019 ; 54(3): 367-75. https://dx.doi.org/10.1002/uog.20150.
- Rizzo G., Mappa I., Bitsadze V., Słodki M., Khizroeva J., Makatsariya A. et al. Role of Doppler ultrasound at time of diagnosis of late-onset fetal growth restriction in predicting adverse perinatal outcome: prospective cohort study. Ultrasound Obstet. Gynecol. 2020; 55(6): 793-8. https://dx.doi.org/10.1002/uog.20406.
- Sharony R., Zipper O., Amichay K., Wiser A., Kidron D., Biron-Shental T. et al. The magnitude of elevated maternal serum human chorionic gonadotropin and pregnancy complications. J. Obstet. Gynaecol. 2017; 37(5): 576-9. https://dx.doi.org/10.1080/01443615.2016.1268578.
- Bakalis S., Peeva G., Gonzalez R., Poon L.C., Nicolaides K.H. Prediction of small for-gestational-age neonates: screening by biophysical and biochemical markers at 30-34 weeks. Ultrasound Obstet. Gynecol. 2015; 46(4): 446-51. https://dx.doi.org/10.1002/uog.14863.
Received 27.01.2025
Accepted 13.02.2025
About the Authors
Maria V. Volochaeva, PhD, Senior Researcher at the Department of Regional Cooperation and Integration, Physician at 1 Maternity Department, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4,+7(919)968-72-98, volochaeva.m@yandex.ru, https://orcid.org/0000-0001-8953-7952
Angelika V. Timofeeva, PhD (Bio), Head of the Laboratory of Applied Transcriptomics, Academician V.I. Kulakov National Medical Research Center for Obstetrics,
Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, avtimofeeva28@gmail.com, https://orcid.org/0000-0003-2324-9653
Ivan S. Fedorov, Researcher at the Laboratory of Applied Transcriptomics, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, is_fedorov@oparina4.ru, https://orcid.org/0000-0002-2104-5887
Natalia E. Kan, Professor, Dr. Med. Sci., Deputy Director General for Research – Director of the Institute of Obstetrics, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, kan-med@mail.ru,
Researcher ID: B-2370-2015, SPIN: 5378-8437, Authors ID: 624900, Scopus Author ID: 57008835600, https://orcid.org/0000-0001-5087-5946
Victor L. Tyutyunnik, Professor, Dr. Med. Sci., Leading Researcher at the Center for Scientific and Clinical Research, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, tioutiounnik@mail.ru,
Researcher ID: B-2364-2015, SPIN: 1963-1359, Authors ID: 213217, Scopus Author ID: 56190621500, https://orcid.org/0000-0002-5830-5099
Kristina O. Ryzhova, Resident, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia,
117997, Russia, Moscow, Ac. Oparin str., 4, cr.yanina@gmail.com, https://orcid.org/0009-0007-8318-435X
Shagane R. Gasymova, Junior Researcher at the Department of Fetal Medicine, Institute of Obstetrics; Ultrasound diagnostics doctor, Department of Ultrasound and Functional Diagnostics, obstetrician-gynecologist, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology,
Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, shagane2501@mail.ru, https://orcid.org/0009-0001-2626-6670
Corresponding author: Maria V. Volochaeva, volochaeva.m@yandex.ru



