The association of polymorphic loci affecting sex hormone-binding globulin levels with the risk of isolated uterine myoma
Ponomarenko M.S., Reshetnikov E.A., Churnosova M.M., Churnosov M.I., Ponomarenko I.V.
Objective: To evaluate the role of polymorphic variants in genes associated with sex hormone-binding globulin (SHBG) levels in the development of isolated uterine myomas.
Materials and methods: The study group comprised 192 patients with isolated uterine myomas and 973 healthy women (controls). Molecular genetic analysis was performed on five gene loci associated with SHBG levels based on data from genome-wide studies: rs8023580 [T/C] NR2F2 (chromosome 15), rs780093 [C/T] GCKR (chromosome 2), rs3779195 [T/A] BAIAP2L1 (chromosome 7), rs7910927 [G/T] JMJD1C (chromosome 10), and rs4149056 [T/C] SLCO1B1 (chromosome 12). Logistic regression with the gPLINK program was used to study the association between polymorphic loci of SHBG candidate genes and the development of isolated uterine fibroids.
Results: An association was found between the rs3779195 [T/A] polymorphism in the BAIAP2L1 gene and the development of isolated uterine fibroids (OR=1.44 for the A allele in the dominant model). The rs3779195 [T/A] polymorphism of BAIAP2L1 and 20 SNPs in linkage disequilibrium with it are functionally significant. These polymorphisms are associated with the interaction of DNA in the BAIAP2L1/BRI3 gene region with 86 transcription factors and regulatory proteins, and with the transcription of 15 genes and splicing of three genes in various organs and tissues related to the pathophysiology of uterine fibroids.
Conclusion: The molecular genetic marker rs3779195 [T/A] of the BAIAP2L1 gene is associated with an increased risk of developing uterine fibroids.
Authors' contributions: Ponomarenko M.S. – conception and design of the study, data synthesis, drafting of the manuscript; Reshetnikov E.A. – drafting of the manuscript; Churnosova M.M. – literature search and analysis; Churnosov M.I. – review, final editing; Ponomarenko I.V. – statistical analysis, editing of the manuscript.
Conflicts of interest: The authors have no conflicts of interest to declare.
Funding: The study was conducted with the support of Russian Science Foundation grant No. 25-25-00034, https://rscf.ru/project/25-25-00034/.
Ethical Approval: The study was reviewed and approved by the Research Ethics Committee of the Belgorod State National Research University.
Patient Consent for Publication: All patients provided informed consent for the publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available upon request from the corresponding author after approval from the principal investigator.
For citation: Ponomarenko M.S., Reshetnikov E.A., Churnosova M.M., Churnosov M.I., Ponomarenko I.V.
The association of polymorphic loci affecting sex hormone-binding globulin levels with the risk of isolated uterine myoma.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2026; (3): 104-110 (in Russian)
https://dx.doi.org/10.18565/aig.2025.255
Keywords
Uterine fibroids are benign tumors originating from smooth muscle tissue and are the most common neoplasms in women of reproductive age [1–3]. The pathogenesis of these diseases is multifactorial and involves a complex interplay between hormonal, genetic, and environmental factors [4–6].
More than ten genome-wide association studies (GWAS) have investigated the genetic determinants of uterine fibroid development. These studies have identified several dozen risk loci associated with the disease in genes involved in sex hormone metabolism (ESR1, FSHB, etc.) [7–10]. However, currently available GWAS data explain only a small proportion (≈13%) of the heritability of uterine fibroids [8], underscoring the need for further genetic investigations.
The role of sex hormones, such as estrogen and testosterone, in uterine fibroid development is well established [11–13]. The pathophysiological effects of these hormones in the body, including uterine fibroids, largely depend on sex hormone-binding globulin (SHBG) [14, 15]. SHBG regulates the levels of free, and therefore bioactive, fractions of testosterone (80% bound by SHBG) [11] and estrogen (38% bound by SHBG) [16] in circulation. These free fractions, accounting for approximately 2% of estrogens and 1% of testosterone, exert biological activity in the female body [11, 16]. SHBG concentration is genetically determined, and numerous GWAS have addressed this [17–23]. However, the role of polymorphisms in genes that determine SHBG concentration in uterine fibroid development remains unclear.
This study aimed to evaluate the role of polymorphic variants of genes associated with SHBG levels in the development of isolated uterine fibroids.
Materials and methods
The study group included 192 patients with uterine fibroids, and the control group comprised 973 healthy women. Participants were recruited at the Perinatal Center (Department of Gynecology) of St. Ioasaph Belgorod Regional Clinical Hospital between 2008 and 2013. The patient group consisted of women with a confirmed diagnosis of uterine fibroids and without other hyperplastic diseases of the pelvic organs, such as genital endometriosis, adenomyosis, or endometrial hyperplasia. The control group included women without benign proliferative disorders of the reproductive system based on clinical and instrumental findings. All women enrolled in the study were of Russian ethnicity, born and residing in the Central region of Russia, and were unrelated to each other [24].
Compared with the control group, patients with uterine fibroids had a significantly higher mean age (p<0.001) and body mass index (BMI) (p<0.001). They also had a significantly higher frequency of previous pregnancies (p<0.001), induced abortions (p<0.001), and chronic endometritis (p=0.04) (Table 1). The Research Ethics Committee of the Belgorod State National Research University reviewed and approved this study.
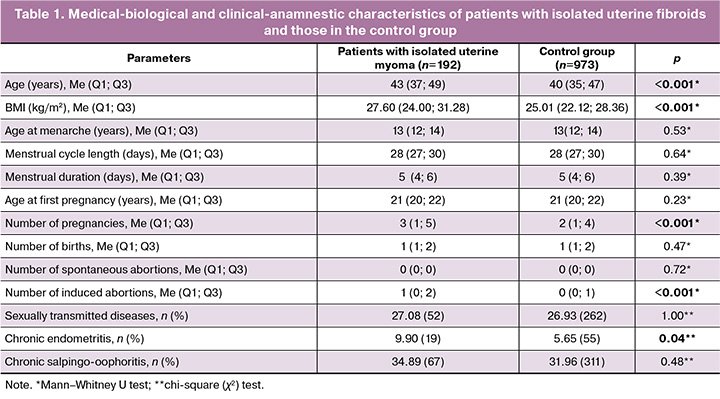
Molecular genetic analysis was performed on four gene loci associated with SHBG levels: rs8023580 [T/C] NR2F2 (15q), rs3779195 [T/A] BAIAP2L1 (7q), rs7910927 [G/T] JMJD1C (10q), and rs4149056 [T/C] SLCO1B1 (12q). SNPs were selected based on the following criteria [25, 26]: (1) association with SHBG levels according to GWAS data; (2) significant regulatory relevance (epigenetic effects and influence on gene transcription); and (3) minor allele frequency of ≥5%.
Biological material for the study (DNA samples) was obtained from the biobank of the Department of Medical and Biological Disciplines of Belgorod State National Research University (storage temperature: -80°C). Genotyping of the four SHBG-associated gene loci was performed using CFX-96 thermal cyclers with the TaqMan probes. DNA amplification kits (for each SNP) were developed by Test-Gen LLC (Ulyanovsk, Russia). The DNA sample concentrations ranged from 10 to 20 ng/μL. Genotyping quality was assessed by testing for Hardy–Weinberg equilibrium for all four SNPs in both the uterine fibroid and control groups. The agreement between the observed and expected heterozygosity (Ho/He) was evaluated.
Statistical analysis
Statistical analysis of the medical-biological and clinical-anamnestic data was performed using Statistica software. The normality of the distribution of all continuous variables was initially assessed using the Shapiro–Wilk test. As none of the continuous variables followed a normal distribution, they are presented as the median (Me) and interquartile range (Q1; Q3) and were compared using the Mann–Whitney U test. Categorical variables are summarized as counts (n) and percentages (%). A comparative analysis of these variables, as well as an assessment of the differences between the observed and expected genotype distributions (for Hardy–Weinberg equilibrium), was conducted using Pearson's χ² test. Differences were considered statistically significant at p<0.05. The association between polymorphic loci of SHBG candidate genes and the development of isolated uterine fibroids was analyzed using gPLINK software (logistic regression method) [27]. The presence and direction of associations were evaluated under four genetic models (allelic, additive, dominant, and recessive) based on odds ratios (OR) and 95% confidence intervals (95% CI) calculated for each model. Covariates (age, BMI, number of pregnancies and induced abortions, and history of chronic endometritis) were included in the analyses, and a permutation test (pperm) was applied to address multiple comparisons [28]. A pperm<0.05 was considered statistically significant. For the molecular genetic marker rs3779195 BAIAP2L1 [T/A] (7q), which is associated with the development of isolated uterine fibroids, and for 20 SNPs in strong linkage disequilibrium with it (linkage strength parameters r²=0.81–1.00 and D'=0.94–1.00), the predicted functional potential was evaluated using in silico data analysis [29]. The HaploReg [accessed July 13, 2025] [30] and GTEx portal [accessed July 13, 2025] [31] tools were used for this analysis.
Results
The distribution of all four analyzed SNPs in both the isolated uterine fibroids group (pHWE≥0.093) and the control group (pHWE≥0.060) was consistent with Hardy–Weinberg equilibrium (p>0.05) (Table 2). The minor allele frequencies at the examined loci ranged from 0.199 to 0.464 in patients with isolated uterine fibroids and from 0.176 to 0.496 in the control group (Table 2).
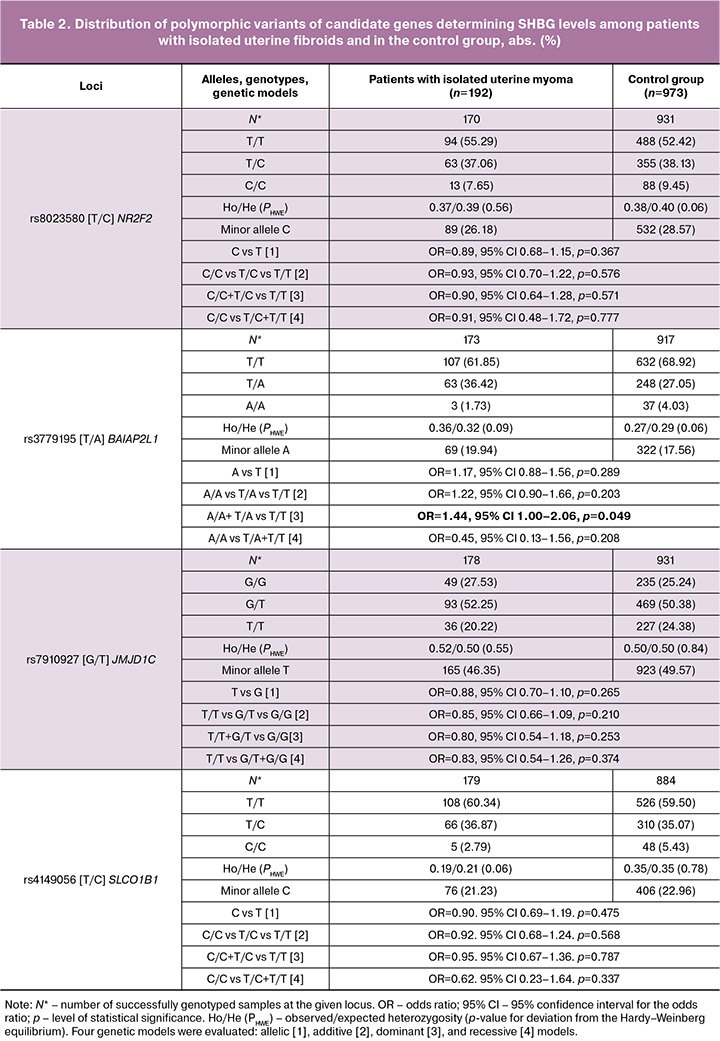
An association was identified between the polymorphic locus rs3779195 of the BAIAP2L1 gene [T/A] and the development of isolated uterine fibroids (Table 2). Under the dominant model, the A allele of rs3779195 BAIAP2L1 [T/A] was associated with an increased risk of isolated uterine fibroids (AA+TA genotypes were observed in 66 [38.15%] patients and 285 [31.08%] women in the control group; OR=1.44, 95% CI 1.00–2.06, p=0.049, pperm=0.049).
In silico analysis (using HaploReg and GTEx Portal) indicated that the rs3779195 [T/A] polymorphism in BAIAP2L1, along with 20 SNPs in strong linkage disequilibrium, was associated with interactions between this genomic region and 68 transcription factors (at 17 loci) and 18 regulatory proteins (at 4 loci). They are also linked to transcriptional regulation (eQTL) of 15 genes and alternative splicing (sQTL) of 3 genes (BRI3, TECPR1, BAIAP2L1) in organs/tissues directly related to the pathophysiology of uterine fibroids: ovaries (eQTL – RP11-307C18.1), adipose tissue (eQTL – BRI3, RP11-307C18.1; sQTL – BRI3), brain (eQTL – BHLHA15, RP11-307C18.1; sQTL – TECPR1, BRI3), adrenal glands (eQTL – RP11-307C18.1), thyroid gland (eQTL – BAIAP2L1, RP11-307C18.1, TECPR1, BHLHA15, LMTK2; sQTL – BRI3), blood (eQTL – TECPR1, RP11-307C18.1), and skeletal muscle (eQTL – BRI3, RP11-307C18.1, ASNS, BAIAP2L1; sQTL – BRI3), among others.
The polymorphic locus rs3779195 [T/A] in BAIAP2L1 and several SNPs in linkage disequilibrium with it are predicted to have important functional significance in the liver, which is the primary site of SHBG synthesis. These SNPs are located within DNA regions characterized by various epigenetic histone modifications (acetylation/methylation) marking promoters (three SNPs), enhancers (ten SNPs), active promoters (six SNPs), and active enhancers (eight SNPs). Furthermore, in the liver, rs3779195 [T/A] BAIAP2L1 and 17 linked SNPs were associated with the expression of two genes: BRI3 and RP11-307C18.1. Notably, the A allele of rs3779195 [T/A] BAIAP2L1, which is associated with an increased risk of isolated uterine fibroids (OR = 1.44), is linked to the elevated expression of BRI3 (NES = 0.87) and reduced transcription of RP11-307C18.1 (NES=-0.54) in the liver.
The protein product of the BAIAP2L1 gene (brain-specific angiogenesis inhibitor 1–associated protein 2-like protein 1, also known as insulin receptor tyrosine kinase substrate [IRTKS]) participates in several biologically important processes, including cell morphogenesis and migration (participation in plasma membrane protrusion formation and actin assembly), as well as proliferation and apoptosis (via activation of EGFR–ERK, PI3K/AKT, and other signaling pathways) [32–34]. These processes play a critical role in oncogenesis [33, 34], including malignant neoplasms of the female reproductive system (e.g., ovarian cancer) [32]. Notably, positive genetic correlations have been reported between uterine fibroids and ovarian cancer [35], suggesting that BAIAP2L1 may be one of the genes underlying these correlations.
Interestingly, several SNPs in linkage disequilibrium with rs3779195 [T/A] BAIAP2L1 are associated with SHBG concentrations and sex hormone levels according to GWAS data: SHBG – rs112758337 (linkage parameters D'=1.00; r²=0.96), rs1688606 (D'=1.00; r²=0.96), and rs4268041 (D'=1.00; r²=0.99) [19]; total testosterone – rs1635612 (D'=1.00; r²=0.96) [19] and rs35903783 (D'=1.00; r²=0.41) [22]. Thus, the genomic region encompassing SNP rs3779195 [T/A] BAIAP2L1, owing to its predicted pronounced functional significance (epigenetic and eQTL effects) in the liver (the principal organ of SHBG synthesis), likely plays an important role in the regulation of SHBG and testosterone levels in the body, thereby influencing the risk of uterine fibroid development.
Two previously published GWAS demonstrated an association between the A allele of rs3779195 [T/A] BAIAP2L1 and low SHBG concentrations [18, 19]. In the present study, we showed that the A allele of the rs3779195 [T/A] BAIAP2L1 polymorphism, previously linked to lower SHBG concentrations in GWAS, is involved in the development of isolated uterine fibroids and represents a risk factor for their occurrence (OR=1.44). A potential mechanism underlying the association between SHBG (and, accordingly, the rs3779195 [T/A] BAIAP2L1 polymorphism associated with SHBG levels) and the formation of isolated uterine fibroids may be related to its role in estrogen transport. Published data indicate that approximately 38% of circulating estrogens are bound to SHBG, whereas only approximately 2% circulate in a free form. These free estrogens exert biological activity in women [36]. Thus, SHBG regulates the concentration of biologically active estrogens in the blood by modulating their bioavailability; lower SHBG levels result in higher concentrations of active estrogens and more pronounced phenotypic effects [37, 38]. Estradiol, a well-established driver of tumor growth, stimulates the proliferation of fibroid cells [12].
Conclusion
The findings of this study suggest an association between the rs3779195 [T/A] BAIAP2L1 polymorphism and the development of isolated uterine fibroids. The A allele of rs3779195 [T/A] BAIAP2L1 was associated with an increased risk of isolated uterine fibroids (OR=1.44). The rs3779195 [T/A] BAIAP2L1 polymorphism and 20 single nucleotide polymorphisms (SNPs) in linkage disequilibrium with it are located in genomic regions that are functionally significant in various organs and tissues related to the pathophysiology of uterine fibroids.
References
- Bulun S.E., Yin P., Wei J., Zuberi A., Iizuka T., Suzuki T. et al. Uterine fibroids. Physiol. Rev. 2025; 105(4): 1947-88. https://dx.doi.org/10.1152/physrev.00010.2024
- Yang Q., Ciebiera M., Bariani M.V., Ali M., Elkafas H., Boyer T.G. et al. Comprehensive review of uterine fibroids: developmental origin, pathogenesis, and treatment. Endocr. Rev. 2022; 43(4): 678-719. https://dx.doi.org/10.1210/endrev/bnab039
- Пономаренко М.С., Решетников Е.А., Пономаренко И.В., Чурносов М.И. Факторы риска развития миомы матки. Акушерство и гинекология. 2024; 3: 20-7. [Ponomarenko M.S., Reshetnikov E.A., Ponomarenko I.V., Churnosov M.I. Risk factors for uterine fibroids. Obstetrics and Gynecology. 2024; (3): 20-7 (in Russian)]. https://dx.doi.org/10.18565/aig.2023.275
- Koltsova A.S., Efimova O.A., Pendina A.A. A view on uterine leiomyoma genesis through the prism of genetic, epigenetic and cellular heterogeneity. Int. J. Mol. Sci. 2023; 24(6): 5752. https://dx.doi.org/10.3390/ijms24065752
- Пономаренко М.С., Решетников Е.А., Пономаренко И.В., Чурносов М.И. Этиопатогенетические механизмы развития миомы матки. Акушерство и гинекология. 2024; 1: 34-41 [Ponomarenko M.S., Reshetnikov E.A., Ponomarenko I.V., Churnosov M.I. Etiopathogenetic mechanisms of uterine fibroids development. Obstetrics and Gynecology. 2024; (1): 34-41 (in Russian)] https://dx.doi.org/10.18565/ig.2023.241
- Пономаренко И.В., Чурносов М.И. Современные представления об этиопатогенезе и факторах риска лейомиомы матки. Акушерство и гинекология. 2018; 8: 27-32. [Ponomarenko I.V., Churnosov M.I. Current views on the etiopathogenesis and risk factors of uterine leiomyoma. Obstetrics and Gynecology. 2018; (8): 27-32 (in Russian)] https://dx.doi.org/10.18565/aig.2018.8.27-32
- Välimäki N., Kuisma H., Pasanen A., Heikinheimo O., Sjöberg J., Bützow R. et al. Genetic predisposition to uterine leiomyoma is determined by loci for genitourinary development and genome stability. Elife. 2018; 7: e37110. https://dx.doi.org/10.7554/eLife.37110
- Rafnar T., Gunnarsson B., Stefansson O.A., Sulem P., Ingason A., Frigge M.L. et al. Variants associating with uterine leiomyoma highlight genetic background shared by various cancers and hormone-related traits. Nat. Commun. 2018; 9(1): 3636. https://dx.doi.org/10.1038/s41467-018-05428-6
- Пономаренко М.С., Решетников Е.А., Пономаренко И.В., Чурносов М.И. Молекулярно-генетические факторы формирования миомы матки. Медицинский Совет. 2025; 4: 21-5. [Ponomarenko M.S., Reshetnikov E.A., Ponomarenko I.V., Churnosov M.I. Molecular genetic factors of uterine fibroids formation. Medical Council. 2025; 4: 21-5 (in Russian)]. https://dx.doi.org/10.21518/ms2025-051
- Ponomarenko M.S., Reshetnikov E.A., Churnosova M.M., Reshetnikova Y.N., Churnosov V.I., Ponomarenko I.V. Comorbidity and syntropy of benign proliferative diseases of the female reproductive system: non-genetic, genetic, and epigenetic factors (review). Research Results in Biomedicine. 2023;9(4): 544-56. https://dx.doi.org/10.18413/2658- 6533-2023-9-4-0-9
- Lv M., Yu J., Huang Y., Ma J., Xiang J., Wang Y. et al. Androgen signaling in uterine diseases: new insights and new targets. Biomolecules. 2022; 12(11): 1624. https://dx.doi.org/10.3390/biom12111624
- Wong J.Y., Gold E.B., Johnson W.O., Lee J.S. Circulating sex hormones and risk of uterine fibroids: study of women's health across the nation (SWAN). J. Clin. Endocrinol. Metab. 2016; 101(1): 123-30. https://dx.doi.org/10.1210/jc.2015-2935
- Alsudairi H.N., Alrasheed A.T., Dvornyk V. Estrogens and uterine fibroids: an integrated view. Research Results in Biomedicine. 2021; 7(2): 156-63. https://dx.doi.org/10.18413/2658-6533-2021-7-2-0-6
- Goldman A.L., Bhasin S., Wu F.C.W., Krishna M., Matsumoto A.M., Jasuja R. A reappraisal of testosterone's binding in circulation: physiological and clinical implications. Endocr. Rev. 2017; 38(4): 302-24. https://dx.doi.org/10.1210/er.2017-00025
- Чурносов В.И. Ассоциации полиморфных локусов генов-кандидатов с уровнем половых гормонов у больных гиперплазией эндометрия. Научные результаты биомедицинских исследований. 2025; 11(2): 243-62. [Churnosov V.I. Associations of polymorphic loci of candidate genes with the level of sex hormones in patients with endometrial hyperplasia. Research Results in Biomedicine. 2025; 11(2): 243-62 (in Russian)] https://dx.doi.org/10.18413/2658-6533-2025-11-2-0-3
- Balogh A., Karpati E., Schneider A.E., Hetey S., Szilagyi A., Juhasz K. et al. Sex hormone-binding globulin provides a novel entry pathway for estradiol and influences subsequent signaling in lymphocytes via membrane receptor. Sci. Rep. 2019; 9(1): 4. https://dx.doi.org/10.1038/s41598-018-36882-3
- Prescott J., Thompson D.J., Kraft P., Chanock S.J., Audley T., Brown J. et al. Genome-wide association study of circulating estradiol, testosterone, and sex hormone-binding globulin in postmenopausal women. PLoS One. 2012; 7(6): e37815. https://dx.doi.org/10.1371/journal.pone.0037815
- Coviello A.D., Haring R., Wellons M., Vaidya D., Lehtimäki T., Keildson S. et al. A genome-wide association meta-analysis of circulating sex hormone-binding globulin reveals multiple loci implicated in sex steroid hormone regulation. PLoS Genet. 2012; 8(7): e1002805. https://dx.doi.org/10.1371/journal.pgen.1002805
- Harrison S., Davies N.M., Howe L.D., Hughes A. Testosterone and socioeconomic position: mendelian randomization in 306,248 men and women in UK Biobank. Sci. Adv. 2021; 7(31): eabf8257. https://dx.doi.org/10.1126/sciadv.abf8257
- Haas C.B., Hsu L., Lampe J.W., Wernli K.J., Lindström S. Cross-ancestry genome-wide association studies of sex hormone concentrations in pre- and postmenopausal women. Endocrinology. 2022; 163(4): bqac020. https://dx.doi.org/10.1210/endocr/bqac020
- Ruth K.S., Campbell P.J.; Chew S., Lim E.M., Hadlow N., Stuckey B.G. et al. Genome-wide association study with 1000 genomes imputation identifies signals for nine sex hormone-related phenotypes. Eur. J. Hum. Genet. 2016; 24(2): 284-90. https://dx.doi.org/10.1038/ejhg.2015.102
- Ruth K.S., Day F.R., Tyrrell J., Thompson D.J., Wood A.R., Mahajan A. et al. Using human genetics to understand the disease impacts of testosterone in men and women. Nat. Med. 2020; 26(2): 252-8. https://dx.doi.org/10.1038/s41591-020-0751-5
- Ohlsson C., Wallaschofski H., Lunetta K.L., Stolk L., Perry J.R., Koster A. et al. Genetic determinants of serum testosterone concentrations in men. PLoS Genet. 2011; 7(10): e1002313. https://dx.doi.org/10.1371/journal.pgen.1002313
- Пономаренко И.В., Полоников А.В., Чурносов М.И. Полиморфные локусы гена LHCGR, ассоциированные с развитием миомы матки. Акушерство и гинекология. 2018; 10: 86-91. [Ponomarenko I.V., Polonikov A.V., Churnosov M.I. Polymorphic LHCGR gene loci associated with the development of uterine fibroids. Obstetrics and Gynecology. 2018; (10): 86-91 (in Russian)]. https://dx.doi.org/10.18565/aig.2018.10.86-91
- Пасенов К.Н. Особенности ассоциаций SHBG-связанных генов с раком молочной железы у женщин в зависимости от наличия наследственной отягощенности и мутаций в генах BRCA1/CHEK2. Научные результаты биомедицинских исследований. 2024; 10(1): 69-88. [Pasenov K.N. Features of associations of SHBG-related genes with breast cancer in women, depending on the presence of hereditary burden and mutations in the BRCA1/CHEK2 genes. Research Results in Biomedicine. 2024; 10(1): 69-88 (in Russian)]. https://dx.doi.org/10.18413/2658-6533-2024-10-1-0-4
- Пономарева Т.А. Генетические варианты глобулина, связывающего половые гормоны, и гормональный профиль больных генитальным эндометриозом. Научные результаты биомедицинских исследований. 2025; 11(1): 75-90. [Ponomareva TA. Genetic variants of sex hormone-binding globulin and hormonal profile in patients with genital endometriosis. Research Results in Biomedicine. 2025; 11(1): 75-90 (in Russian)]. https://dx.doi.org/10.18413/2658-6533-2025-11-1-0-4
- Purcell S., Neale B., Todd-Brown K., Thomas L., Ferreira M.A., Bender D. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 2007; 81(3): 559-75. https://dx.doi.org/10.1086/519795
- Che R., Jack J.R., Motsinger-Reif A.A., Brown C.C. An adaptive permutation approach for genome-wide association study: evaluation and recommendations for use. BioData Min. 2014; 7: 9. https://dx.doi.org/10.1186/1756-0381-7-9
- Полоников А.В., Клёсова Е.Ю., Азарова Ю.Э. Биоинформатические инструменты и интернет-ресурсы для оценки регуляторного потенциала полиморфных локусов, установленных полногеномными ассоциативными исследованиями мультифакториальных заболеваний (обзор). Научные результаты биомедицинских исследований. 2021; 7(1): 15-31. [Polonikov A.V., Klyosova E.Yu., Azarova I.E. Bioinformatic tools and internet resources for functional annotation of polymorphic loci detected by genome wide association studies of multifactorial diseases (review). Research Results in Biomedicine. 2021; 7(1): 15-31 (in Russian)] https://dx.doi.org/10.18413/2658-6533-2020-7-1-0-2
- Ward L.D., Kellis M. HaploReg v4: systematic mining of putative causal variants, cell types, regulators and target genes for human complex traits and disease. Nucleic Acids Res. 2016; 44(D1): D877-81. https://dx.doi.org/10.1093/nar/gkv1340
- GTEx Consortium. The GTEx Consortium atlas of genetic regulatory effects across human tissues. Science. 2020; 369(6509): 1318-30. https://dx.doi.org/10.1126/science.aaz1776
- Chao A., Tsai C. L., Jung S.M., Chuang W.C., Kao C., Hsu A. et al. BAI1-associated protein 2-like 1 (BAIAP2L1) is a potential biomarker in ovarian cancer. PLoS One. 2015; 10(7): e0133081. https://dx.doi.org/10.1371/journal.pone.0133081
- Song Y., Zhuang G., Li J., Zhang M. BAIAP2L2 facilitates the malignancy of prostate cancer (PCa) via VEGF and apoptosis signaling pathways. Genes Genomics. 2021; 43(4): 421-32. https://dx.doi.org/10.1007/s13258-021-01061-8
- Lu Y., Zhou X.Y., Zhou C.L., Liu J., Yong T., Fan Y. et al. Insulin receptor tyrosine kinase substrate (IRTKS) promotes the tumorigenesis of pancreatic cancer via PI3K/AKT signaling. Hum. Cell. 2022; 35(6): 1885-99. https://dx.doi.org/10.1007/s13577-022-00770-w
- Zhao C., Shang A., Wu H., Li Q., Peng L., Yue C. Causal relationship between genetically predicted uterine leio-myoma and cancer risk: a two-sample Mendelian randomization. Front. Endocrinol. (Lausanne). 2024; 15: 1429165. https://dx.doi.org/10.3389/fendo.2024.1429165
- Ponomarenko M., Reshetnikov E., Churnosova M., Aristova I., Abramova M., Novakov V. et al. Genetic variants linked with the concentration of sex hormone-binding globulin correlate with uterine fibroid risk. Life (Basel). 2025; 15(7): 1150. https://dx.doi.org/10.3390/life15071150
- Hammond G.L. Plasma steroid-binding proteins: primary gatekeepers of steroid hormone action. J. Endocrinol. 2016; 230(1): 13-25. https://dx.doi.org/10.1530/JOE-16-0070
- Xing C., Zhang J., Zhao H., He B. Effect of sex hormone-binding globulin on polycystic ovary syndrome: mechanisms, manifestations, genetics, and treatment. Int. J. Womens Health. 2022; 14: 91-105. https://dx.doi.org/10.2147/IJWH.S344542
Received 17.09.2025
Accepted 12.02.2026
About the Authors
Marina S. Ponomarenko, PhD student at the Department of Biomedical Disciplines, Belgorod State National Research University, 308015, Russia, Belgorod, Pobedy str., 85, +7(4722)30-13-83, ponomarenkomc@yandex.ru, https://orcid.org/0009-0009-0312-0829Evgeny A. Reshetnikov, Dr. Sci. (Bio), Professor at the Department of Medical and Biological Disciplines, Belgorod State National Research University, 308015, Russia, Belgorod, Pobedy str., 85, +7(4722)30-13-83, reshetnikov@bsuedu.ru, http://orcid.org/0000-0002-5429-6666
Maria M. Churnosova, student at the Medical Institute, Belgorod State National Research University, 308015, Russia, Belgorod, Pobedy str., 85, +7(4722)30-13-83, churnosovamary@gmail.com, https://orcid.org/0000-0001-6444-8806
Mikhail I. Churnosov, Dr. Med. Sci., Head of the Department of Medical and Biological Disciplines, Belgorod State National Research University, 308015, Russia, Belgorod, Pobedy str., 85, +7(4722)30-13-83, churnosov@bsuedu.ru, http://orcid.org/0000-0003-1254-6134
Irina V. Ponomarenko, Dr. Med. Sci., Professor at the Department of Medical and Biological Disciplines, Belgorod State National Research University, 308015, Russia, Belgorod, Pobedy str., 85, +7(4722)30-13-83, ponomarenko_i@bsuedu.ru, https://orcid.org/0000-0002-5652-0166
Corresponding author: Irina V. Ponomarenko, ponomarenko_i@bsuedu.ru



