Uterine hemodynamics in the first trimester after treatment of chronic endometritis: comparison between ongoing pregnancy and early pregnancy loss
Ozerskaya I.A., Porkhovaty S.Ya., Kazaryan G.G., Ozhogina E.V.
Objective: To identify the characteristics of uterine hemodynamics in patients after treatment of chronic endometritis at 5–7 and 8–10 weeks of gestation, and to assess association between the changes in uterine hemodynamics and first trimester pregnancy status (ongoing pregnancy/pregnancy loss).
Materials and methods: The retrospective analysis included 112 pregnant women with a history of diagnosed chronic endometritis (CE). Of them 62 women previously underwent comprehensive treatment at 5–7 weeks of gestation (46 had ongoing pregnancy, 18 experienced pregnancy loss), and 48 women underwent treatment at 8–10 weeks of gestation (42 had ongoing pregnancy, 6 experienced pregnancy loss). Uterine artery Doppler ultrasound evaluation of Vmax, Vmin, Vmean, PI, RI was performed, and the arterial perfusion index (API) was calculated at the given gestational age based on pregnancy status.
Results: After treatment of CE, the most informative parameters were API and Vmean at 5–7 weeks of gestation, whereas at 8–10 weeks the differences in these parameters were predominantly associated with uterine vascular resistance indices (PI, RI) and Vmean.
Conclusion: After treatment of CE, the parameters of uterine hemodynamics in early pregnancy varied in relation to gestational age and pregnancy status in the first trimester. Comprehensive assessment of uterine artery blood flow parameters can improve the clinical benefits of uterine artery Doppler ultrasound in management of early pregnancy in women with a history of chronic endometritis.
Authors' contributions: Ozerskaya I.A. – the study concept and design, article editing; Porkhovaty S.Ya. – material collection and processing; Kazaryan G.G., Ozhogina E.V. – article writing; Ozhogina E.V. – statistical data processing.
Conflicts of interest: The authors confirm that they have no conflicts of interest to declare.
Funding: The study was conducted without any sponsorship.
Ethical Approval: The study was approved by the local Ethics Committee of the Faculty of Continuous Medical Education, Patrice Lumumba Peoples’ Friendship University of Russia.
Patient Consent for Publication: The patients have signed informed consent for participation in the study and publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available on request from the corresponding author after approval from the principal investigator.
For citation: Ozerskaya I.A., Porkhovaty S.Ya., Kazaryan G.G., Ozhogina E.V. Uterine hemodynamics in the first trimester after treatment of chronic endometritis: comparison between ongoing pregnancy and early pregnancy loss.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2026; (2): 78-85 (in Russian)
https://dx.doi.org/10.18565/aig.2026.01
Keywords
Chronic endometritis (CE) is a crucial issue in modern reproductive medicine due to its high prevalence and adverse effects on implantation and early stages of gestation [1, 2].
Despite the fact that antibacterial therapy is believed to be the initial stage in treatment of CE, this issue is controversial. Some studies have shown significant variability in the frequency of negative changes associated with CE that were found in control assessment, and the lack of unified antibacterial therapy regimens and duration of treatment [3, 4].
It is important to note, that complete morphofunctional restoration of the endometrium is not always possible after treatment, and some patients have a risk of early impaired placentation and termination of pregnancy [5–7]. There is evidence that antibacterial therapy does not always lead to minimization of the risk of adverse outcomes, at least in patient population undergoing infertility treatment by using assisted reproductive technologies, that requires a cautious interpretation of the notion of cure, exclusively as a morphological criterion [8].
One of the promising approaches to assessment of the functional state of the uterus in early pregnancy is uterine artery Doppler ultrasound. Conventionally, Doppler measurements of vascular resistance parameters – the pulsatility index (PI) and the resistance index (RI) are used in clinical practice [9, 10]. Nevertheless, the data in literature indicate that measurement of blood velocity and/or volumetric flow rate can also be informative in early pregnancy [11–14].
The issue about choosing the most clinically significant uterine hemodynamic parameters in early pregnancy in patients after treatment of CE remains disputable. The differences between hemodynamic parameters in the first trimester related to gestational age and pregnancy status are underexplored. There are few publications evaluating uterine/spiral artery blood flow in early pregnancy and the impact of treatment in the preconception period on patients with a history of CE [15, 16]. However, there are limited data comparing hemodynamic parameters of the uterine artery between 5–7 and 8–10 weeks of gestation and different pregnancy status (ongoing pregnancy or pregnancy pregnancy loss in the first trimester). It explains the relevance of our study.
The objective of the study was to identify the characteristics of uterine hemodynamics in patients after treatment of chronic endometritis at 5–7 and 8–10 weeks of gestation, and to assess its association with first-trimester pregnancy status (ongoing pregnancy/pregnancy loss).
Material and methods
The retrospective observational comparative study was carried out at the Medical Center “Asklepion” in 2023–2025 and included 112 pregnant women aged from 24 to 45 years with singleton pregnancy, who had a history of CE and previously underwent comprehensive treatment. Uterine artery Doppler ultrasound was performed for these patients in early pregnancy.
Inclusion criteria were the following: multiple pregnancy, uterine fibroids (any size of submucosal fibroids, Types 1 and 2, as well as interstitial and interstitial-subserousal fibroids larger than > 20 mm, Types 3, 4, 5); space-occupying lesions of the uterine appendages (ovarian cysts, sactosalpinx); age over 45 years.
All patients have signed informed consent for publication of their data.
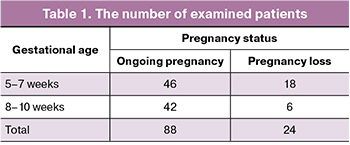
Based on the gestational age and pregnancy status in the first trimester, the patients were divided into groups: the group with ongoing pregnancy and the group with pregnancy loss before 11 weeks of gestation. The group with pregnancy loss included the cases of spontaneous abortion or missed abortion, that was confirmed by ultrasound examination and medical documentation. Separate analysis was performed both for gestational age 5–7+6 weeks and 8–10+6 weeks (further referred to as 5–7 weeks and 8–10 weeks). The number of patients in the groups is represented in Table 1.
Before starting the study, Ethics Committee’s approval was obtained from the Faculty of Continuous Medical Education, Peoples’ Friendship University of Russia named after Patrice Lumumba. All patients signed informed consent for publication of their data.
Clinically indicated transvaginal Doppler ultrasound was performed in pregnant women using Philips Affiniti 70 Ultrasound System (Philips, the Netherlands) with ultrasound transducer (frequency range: 9–3 MHz) according to the ISUOG recommendations for performance of transvaginal ultrasound [17].
To minimize the adverse effects on the embryo, Doppler ultrasound was performed strictly following the ISUOG statement on the safe use of Doppler for fetal ultrasound examination: the displayed thermal index was 0.3–0.5, exposure time was no longer that 5 minutes, and the color flow mapping was used along the lateral margins of the uterus without damaging the embryo [18].
Ultrasound measurements included uterine volume (V) and the diameter of uterine artery (UA) with an accuracy of 0.1 mm. Doppler ultrasound of the uterine arteries was used to measure peak systolic velocity (Vmax), time averaged mean blood flow velocity (Vmean), end-diastolic velocity (Vmin), pulsatility index (PI), and resistance index (RI). Based on the obtained data, the arterial perfusion index (API) was calculated, which reflected the amount of blood (%) entering 1 cm³ of the uterine body through the both uterine arteries.
Volumetric blood flow (Vvol) in each UA (cm³ per 1 cardiac cycle) was calculated by the formula:
Vvol = Vmean × S, where S represents the cross-sectional area of the uterine artery (cm²). S was calculated using the diameter (d) of each UA.
API was calculated by the formula:
API = (Vvol_RUA + Vvol_LUA / UV × 100%,
where Vvol_RUA represents volumetric blood flow in the right uterine artery (cm³), Vvol_LUA represents volumetric blood flow in the left uterine artery (cm³), and UV stands for uterine volume (cm³).
Since Vmean was used to calculate volumetric blood flow, and UV was included in the API formula, the values of API, Vmean, and UV are partially interdependent.
Statistical analysis
Statistical data processing was conducted using non-parametric statistical tests in Python module (SciPy, statsmodels). Normality of distribution of the quantitative parameters was tested by using the plots and the Shapiro–Wilk test. Due to deviation of normal distribution, the data are represented as the median and interquartile range (Me [Q1; Q3]).
The Mann–Whitney U test (two-tailed) was used in the intergroup comparison of the quantitative parameters. The differences were considered statistically significant at р<0.05.
The Benjamini–Hochberg procedure was used to control the false discovery rate (FDR) in multiple comparisons of each gestational age. The q-values were calculated. The differences were considered statistically significant at q<0.05.
The Hodges-Lehmann estimator was used to estimate the clinical significance of differences (ΔHL; the difference in the medians (ongoing pregnancy minus pregnancy loss) with 95% confidence interval calculated by bootstrap resampling (4000 replications).
Results
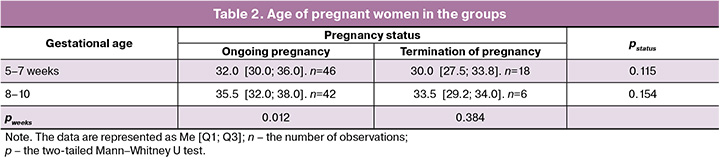
There was no statistically significant age difference between the patients in the groups with ongoing pregnancy and pregnancy loss at similar gestational age (p>0.05). At the same time, the age of women in the group with ongoing pregnancy at 8–10 weeks of gestation was higher compared with pregnancy at 5–7 weeks (p=0,012), whereas there was no age difference between the women in the group with pregnancy loss at 8–10 weeks and 5–7 weeks (p>0.05) (Table 2).
There was no statistically significant asymmetry found in blood flow in RUA and LUA (p>0.05). Therefore, the average values were included in further analysis.
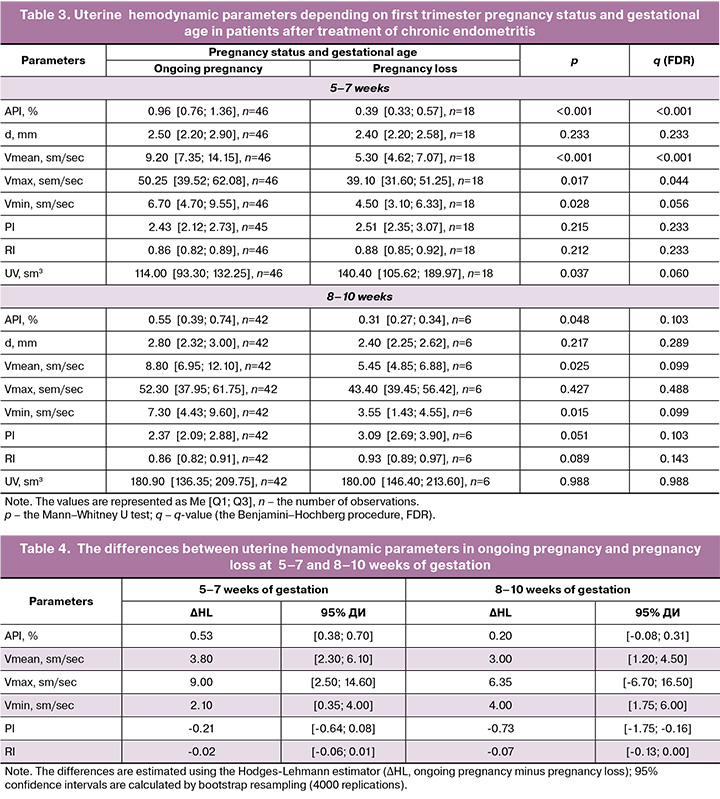
In patients after treatment of CE, marked changes in API and Vmean (q<0.001 for both parameters) were found between ongoing pregnancy and pregnancy loss at 5–7 weeks. API was higher in ongoing pregnancy (p<0,001; ΔHL=0,53). Similarly, Vmean was higher (p<0.001; ΔHL=3.80). Moderate differences were found in Vmax (p=0.017; q=0.044; ΔHL=9.00), whereas Vmin had borderline significance (p=0.028; q=0.056; ΔHL=2.10). The differences in PI and RI were not statistically significant (p>0.05). Also, the difference in uterine volume (p=0.037; q=0.060) tended to be even more marked. Reduced uterine volume was in ongoing pregnancy versus pregnancy loss.
The difference was found in blood flow velocity between ongoing pregnancy and pregnancy loss at 8–10 weeks. Marked tendency towards the difference in Vmean and Vmin was in ongoing pregnancy, and their values were higher (Vmean: p=0.025; q=0.099; ΔHL=3.00; Vmin: p=0.015; q=0.099; ΔHL=4.00). However, statistical significance was not achieved after FDR correction, that requires careful interpretation given the small size of the subgroup with termination of pregnancy (n=6). Vascular resistance parameters tended to be higher in pregnancy loss, and marked increase in was in PI (p=0.051; q=0.103; ΔHL=-0.73). There was difference in API at p=0.048. However, after FDR correction the differences were not preserved (q=0.103).
There was a steady increase in uterine volume from 5–7 weeks to 8–10 weeks in ongoing pregnancy (114.00 cm3 versus 180.90 cm3). In cases of pregnancy loss between 5–7 weeks and 8–10 weeks the difference in uterine volume was not statistically significant. This could be due to a limited number of observations (comparison of gestational age was done separately). Reduction in API was observed from 5–7 weeks to 8–10 weeks in ongoing pregnancy, that reflected mathematical dependence of API on Vmean and UV.
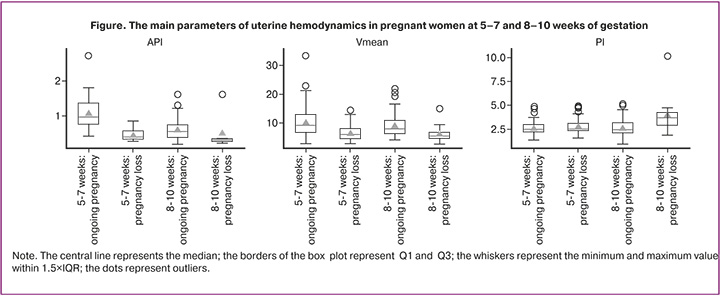
Descriptive statistics of the uterine hemodynamic parameters and the results of intergroup comparisons (p and q (FDR)) are represented in Table 3. Clinically interpretable features and marked intergroup differences in effect size of the analyzed parameters (ΔHL with 95% CI) are represented in Table 4. Comparison of the main parameters (API, Vmean, PI) is shown in the box plot.
Discussion
Evaluation of blood flow in uterine arteries and their branches has been used in obstetrics since the end of the 20th century, and is widely used in monitoring physiological changes of pregnancy and predicting complications including fetal growth restriction and preeclampsia [9, 19, 20].
The results of this study confirm that in patients after treatment of CE, uterine hemodynamic parameters in early pregnancy are different depending on first trimester pregnancy status (ongoing pregnancy/termination of pregnancy). This is consistent with the assumption that CE is associated with impairment of morphofunctional characteristics of the endometrium, changes in the local inflammatory and immune response, and as a consequence, with impaired implantation and early placentation, that can increase the risk of early reproductive losses even after treatment [21, 22].
The age of women in the groups with different pregnancy status within similar gestational age was comparable, that reduces the risk of confounding in the interpretation of the identified differences. The age difference between the subgroups with 5–7 weeks and 8–10 weeks of ongoing pregnancy apparently reflects a special feature of formation of the sample in the retrospective study. However, this factor should be taken into account in gestational age-dependent comparisons.
Marked intergroup differences in the values of API and Vmean were found at 5–7 weeks of gestation. When interpreting these results, it is important to take into account that Vmean is included in the formula for calculation of volumetric blood flow and, therefore, is a component of API. Thus, the values of API and Vmean are partially interdependent and should not be interpreted as completely independent predictors. However, API is an integral feature of perfusion. In addition to the velocity component, it takes into account the cross-sectional area of the uterine artery and is normalized with uterine volume. This makes it possible to interpret API as an indicator of total blood flow entering 1 сm3 of uterine volume. Therefore, measurements of API and Vmean can be simultaneously informative in early pregnancy, and can reflect both reduction of blood flow velocity and less favorable ratio between total uterine artery blood flow and uterine volume. The published review paper (2024) reported that endometrial vascular changes in women with infertility may indicate association of CE with vascular pathology [23]. Further it is expedient that contribution of the API components (blood flow velocity, arterial diameter and uterine volume) should be evaluated separately.
Special consideration should be given to the dynamics of uterine volume, which consistently increases with advancing gestational age. In our study, uterine volume increased from 5–7 weeks to 8–10 weeks of ongoing pregnancy. Similar trend without reaching statistical significance was observed in the group with pregnancy loss most likely due to the small size of the subgroup with 8–10 weeks of gestation. Since uterine volume is included in the API formula as the denominator, reduction in the values of API or absence of increase in API with advancing gestationalweek may reflect not only changes in uterine blood flow but also the effect of normalization with increased uterine volume, when an increase in total UA blood flow volume does not outpace an increase in uterine volume. This enhances the clinical interpretability of API as an integral indicator of the relationship between uterine blood flow and uterine volume in comparison with different gestational age.
Comparison with data in literature showed heterogeneous data on the role of first trimester uterine arteries Doppler ultrasound in reproductive losses. Some studies demonstrate association of UA parameters with early adverse outcomes, while others indicate limited prognostic applicability and contest-depended outcomes (population, gestational age, clinical situation, standardization of methodology) [24, 25]. The study (1998) showed that marked asymmetry and abnormal spectrum of UA blood flow in the first trimester can be associated with subsequent pregnancy loss [26]. Thereafter, impaired uterine hemodynamics in patients with missed miscarriage and/or spontaneous abortion in the first trimester was confirmed by other authors [27–30]. Therefore, it is prudent to consider the results obtained by us not as a universal screening tool, but as a clinically oriented set of markers to be used in patients after treatment of CE during first trimester pregnancy evaluation at certain weeks of gestation.
The reproducibility of Doppler ultrasound measurements in the first trimester depends on measurement protocol, the examiner’s ultrasound experience and patient factors (including body mass index), that requires standardization of methodology, and partially explains the interobserver variability [31].
The group with pregnancy loss in this study included both spontaneous abortion and missed miscarriage. Combination of these types of abortions is clinically justified in analyzing early pregnancy adverse outcomes. However, it increases heterogeneity and can mask the differences between certain parameters. Similar methodological compromises have been described in other studies on early reproductive losses [27, 29].
The limitations of our study (retrospective design and small sample sizes, particularly in the group with termination of pregnancy at 8–10 weeks) reduce statistical power and increase the risk of chance findings. Effect size estimation and robust approach (confidence intervals calculation by bootstrap resampling for the main parameters and small p-value estimation for permutation tests) were used to improve the reliability of interpretation, that is consistent with current recommendations for limited samples and multiple comparisons. [32].
Two-stage treatment strategy for endometrial cancer is used in Russia. The first stage is aimed to eliminate the inflammatory process, while the second stage is restoration of the morphofunctional potential of the endometrium, including local hemodynamics and microcirculation [33–35]. This approach is supported by foreign researchers [36]. The data in our study are consistent with this approach: higher values of API and Vmean in ongoing pregnancy (mostly marked at 5–7 weeks) indicate that adequate uterine microcirculation is a favorable prognostic factor in early pregnancy after treatment of CE. Therefore, the obtained data show that the clinical and pathogenetic model demonstrates the importance of not only eradication of infection, but also the subsequent restoration of perfusion and microcirculation as a component of the functional readiness of the endometrium for pregnancy.
In practical terms, the obtained results support gestational age-specific interpretation. At 5–7 weeks of ongoing pregnancy, API and correlated Vmean are the most informative parameters of uterine arterial perfusion compared with the group with pregnancy loss, whereas at 8–10 weeks, PI and RI are the most informative parameters reflecting resistance associated with spiral artery remodeling. Verification of these findings in prospective studies with a larger sample size and a standardized protocol for Doppler ultrasound will help determine more accurately the threshold values and clinical algorithms for monitoring the patients after treatment of CE. Given that this is a pilot study, further studies are necessary to confirm the reproducibility of the identified associations.
Conclusion
After treatment of CE, uterine hemodynamic parameters in patients in early pregnancy are different depending on gestational age and pregnancy status in the first trimester. The most informative parameters at 5–7 weeks of pregnancy are API and Vmean, whereas PI and RI at 8–10 weeks reflecting vascular resistance. A comprehensive evaluation of these parameters can enhance the clinical informativeness of Doppler ultrasound in management of patients in early pregnancy after treatment of CE.
References
- Александрова Т.Н., Юрьев С.Ю. Хронический эндометрит как звено репродуктивных потерь: новые аспекты патогенеза (обзор литературы). Мать и дитя в Кузбассе. 2020; 4(83): 70-4. [Aleksandrova T.N., Yuriev S.Yu. Chronic endometritis as a part of reproductive losses: new aspects of pathogenesis (review of literature). Mother and Child in Kuzbass. 2020; 4(83): 70-4 (in Russian)]. https://dx.doi.org/10.24411/2686-7338 - 2020-10051
- HogenEsch E., Hojjati R., Komorowski A., Maniar K., Pavone M.E., Bakkensen J. et al. Chronic endometritis: screening, treatment, and pregnancy outcomes in an academic fertility center. J. Assist. Reprod. Genet. 2023; 40(10): 2463-71. https://dx.doi.org/10.1007/s10815-023-02902-z
- Liu J., Liu Z.A., Liu Y., Cheng L., Yan L. Impact of antibiotic treatment for chronic endometritis on pregnancy outcomes in women with reproductive failures (RIF and RPL): a systematic review and meta-analysis. Front. Med. (Lausanne). 2022; 9: 980511. https://dx.doi.org/10.3389/fmed.2022.980511
- Дубровина С.О., Божинская Д.М., Гимбут В.С., Богунова Д.Ю., Циркунова Н.С., Циркунова К.Г. Хронический эндометрит. Акушерство и гинекология. 2024; 6: 147-52. [Dubrovina S.O., Bozhinskaya D.M., Gimbut V.S., Bogunova D.Yu., Tsirkunova N.S., Tsirkunova K.G. Chronic endometritis. Obstetrics and Gynecology. 2024; (6): 147-52 (in Russian)]. https://dx.doi.org/10.18565/aig.2024.137
- Толибова Г.Х., Траль Т.Г., Коган И.Ю., Олина А.А. Эндометрий. Атлас. М.: StatusPraesens; 2022. 184 с. [Tolibova G.Kh., Tral T.G., Kogan I.Yu., Olina A.A. Endometrium. Atlas. Moscow: StatusPraesens; 2022. 184 p. (in Russian)].
- Yan X., Jiao J., Wang X. The pathogenesis, diagnosis, and treatment of chronic endometritis: a comprehensive review. Front. Endocrinol. 2025; 16: 1603570. https://dx.doi.org/10.3389/fendo.2025.1603570
- Kuroda K., Horikawa T., Moriyama A., Nakao K., Juen H., Takamizawa S. et al. Impact of chronic endometritis on endometrial receptivity analysis results and pregnancy outcomes. Immun. Inflamm. Dis. 2020; 8(4): 650-8. https://dx.doi.org/10.1002/iid3.354
- Duan H., Li X., Hao Y., Shi J., Cai H. Risk of spontaneous abortion after antibiotic therapy for chronic endometritis before in vitro fertilization and intracytoplasmic sperm injection stimulation. Fertil. Steril. 2022; 118(2): 337-46. https://dx.doi.org/10.1016/j.fertnstert.2022.04.026
- Sotiriadis A., Hernandez-Andrade E., da Silva Costa F., Ghi T., Glanc P., Khalil A. et al. ISUOG practice guidelines: role of ultrasound in screening for and follow-up of pre-eclampsia. Ultrasound Obstet. Gynecol. 2019; 53(1): 7-22. https://dx.doi.org/10.1002/uog.20105
- Oancea M., Grigore M., Ciortea R., Diculescu D., Bodean D., Bucuri C. et al. Uterine artery doppler ultrasonography for first trimester prediction of preeclampsia in individuals at risk from low-resource settings. Medicina (Kaunas). 2020; 56(9): 428. https://dx.doi.org/10.3390/medicina56090428
- Leible S., Canals A., Sepulveda W. Physiological asymmetry in uterine artery blood flow during the first trimester of pregnancy: new insights from a quantitative assessment using Pulsed-wave color Doppler and M-mode color power angio imaging. Placenta. 2022; 121: 109-15. https://dx.doi.org/10.1016/j.placenta.2022.03.006
- Rigano S., Ferrazzi E., Boito S., Pennati G., Padoan A., Galan H. Blood flow volume of uterine arteries in human pregnancies determined using 3D and bi-dimensional imaging, angio-Doppler, and fluid-dynamic modeling. Placenta. 2010; 31(1): 37-43. https://dx.doi.org/10.1016/j.placenta.2009.10.010
- McKelvey A., Pateman K., Balchin I., Peebles D.M., Rodeck C.H., David A.L. Total uterine artery blood volume flow rate in nulliparous women is associated with birth weight and gestational age at delivery. Ultrasound Obstet. Gynecol. 2017; 49(1): 54-60. https://dx.doi.org/10.1002/uog.15917
- Mäkikallio K., Tekay A., Jouppila P. Uteroplacental hemodynamics during early human pregnancy: a longitudinal study. Gynecol. Obstet. Invest. 2004; 58(1): 49-54. https://dx.doi.org/10.1159/000077914
- Likhachov V., Taranovska О. Changes in the intensity of uterine circulation in pregnant women with a history of chronic endometritis. Neonatology, surgery and perinatal medicine. 2023; 13(2(48): 78-84. https://dx.doi.org/10.24061/2413-4260.XIII.2.48.2023.11
- Кохно Н.И., Самойлова Т.Е., Докудаева Ш.А. Прегравидарные прогностические критерии децидуита. Гинекология. 2019; 21(2): 58-65. [Kokhno N.I., Samoylova T.E., Dokudaeva Sh.A. Pregravid prognostic criteria for deciduitis. Gynecology. 2019; 21(2): 58-65 (in Russian)]. https://dx.doi.org/10.26442/20795696.2019.2.190221
- Bhide A., Acharya G., Baschat A., Bilardo C.M., Brezinka C., Cafici D. et al. ISUOG Practice Guidelines (updated): use of Doppler velocimetry in obstetrics. Ultrasound Obstet. Gynecol. 2021; 58(2): 331-9. https://dx.doi.org/10.1002/uog.23698
- Salvesen K., Abramowicz J., Ter Haar G., Miloro P., Sinkovskaya E., Dall'Asta A. et al. ISUOG statement on the safe use of Doppler for fetal ultrasound examination in the first 13 + 6 weeks of pregnancy (updated). Ultrasound Obstet. Gynecol. 2021; 57(6): 1020. https://dx.doi.org/10.1002/uog.23610
- Velauthar L., Plana M.N., Kalidindi M., Zamora J., Thilaganathan B., Illanes S.E. et al. First-trimester uterine artery Doppler and adverse pregnancy outcome: a meta-analysis involving 55,974 women. Ultrasound Obstet. Gynecol. 2014; 43(5): 500-7. https://dx.doi.org/10.1002/uog.13275
- Лазарева Г.А., Чебышева Е.Л. Прогнозирование перинатального исхода при преэклампсии на основании доплерометрических показателей. Доктор.Ру. 2022; 21(5): 13-7. [Lazareva G.A., Chebysheva E.L. Prediction of perinatal outcome in preeclampsia based on Doppler parameters. Doctor.Ru. 2022; 21(5): 13-7 (in Russian)]. https://dx.doi.org/10.31550/1727-2378-2022-21-5-13-17
- Доброхотова Ю.Э., Кузнецов П.А., Джохадзе Л.С. Привычное невынашивание. Актуальное сегодня (Протокол ESHRE 2023 г., Национальные клинические рекомендации «Привычный выкидыш» 2021 г., материалы Всемирного конгресса ESHRE 2023 г.). РМЖ. Мать и дитя. 2023; 6(3): 219-25. [Dobrokhotova Yu.E., Kuznetsov P.A., Dzhokhadze L.S. Habitual miscarriage. Relevant today (ESHRE Protocol 2023, National Clinical Guidelines "Habitual Miscarriage" 2021, Proceedings of the ESHRE World Congress 2023). RMJ. Mother and Child. 2023; 6(3): 219-25 (in Russian)]. https://dx.doi.org/10.32364/2618-8430-2023-6-3-1
- Kimura F., Takebayashi A., Ishida M., Nakamura A., Kitazawa J., Morimune A. et al. Review: chronic endometritis and its effect on reproduction. J. Obstet. Gynaecol. Res. 2019; 45(5): 951-60. https://dx.doi.org/10.1111/jog.13937
- Ticconi C., Inversetti A., Marraffa S., Campagnolo L., Arthur J., Zambella E. et al. Chronic endometritis and recurrent reproductive failure: a systematic review and meta-analysis. Front. Immunol. 2024; 15: 1427454. https://dx.doi.org/10.3389/fimmu.2024.1427454
- Aygün E.G., Kahraman E. First-trimester uterine artery Doppler indices and pregnancy outcomes in naturally conceived and frozen-thawed embryo transfer cycles. Diagnostics (Basel). 2025; 15(17): 2223. https://dx.doi.org/10.3390/diagnostics15172223
- Pellizzari P., Pozzan C., Marchiori S., Zen T, Gangemi M. Assessment of uterine artery blood flow in normal first-trimester pregnancies and in those complicated by uterine bleeding. Ultrasound Obstet. Gynecol. 2002; 19(4): 366-70. https://dx.doi.org/10.1046/j.1469-0705.2002.00667.x
- Leible S., Cumsille F., Walton R., Muñoz H., Jankelevich J., Sepulveda W. Discordant uterine artery velocity waveforms as a predictor of subsequent miscarriage in early viable pregnancies. AJOG. 1998; 179(6): 1587-93. https://dx.doi.org/10.1016/S0002-9378(98)70030-4
- Cavoretto P.I., Salmeri N., Candiani M., Farina A. Reference ranges of uterine artery pulsatility index from first to third trimester based on serial Doppler measurements: longitudinal cohort study. Ultrasound Obstet. Gynecol. 2023; 61(4): 474-80. https://dx.doi.org/10.1002/uog.26092
- Guedes-Martins L., Saraiva J.P., Gaio A.R., Reynolds A., Macedo F., Almeida H. Uterine artery Doppler in the management of early pregnancy loss: a prospective, longitudinal study. BMC Pregnancy Childbirth. 2015; 15: 28. https://dx.doi.org/10.1186/s12884-015-0464-9
- Zhong Y., Wang N., Lu S., Lu Y., Pan X., Zhou Y. Doppler evaluation of uterine blood flow in patients with unexplained recurrent pregnancy loss. Int. J. Womens Health. 2024; 16: 1803-14. https://dx.doi.org/10.2147/IJWH.S477828
- Bachnas M.A., Budihastuti U.R., Melinawati E., Anggraini N.W.P., Ridwan R., Astetri L. et al. First-trimester Doppler ultrasound for predicting successful management of pregnancy with recurrent pregnancy losses due to antiphospholipid syndrome and thrombophilia: a cohort study. J. Hum. Reprod. Sci. 2024; 17(4): 261-8. https://dx.doi.org/10.4103/jhrs.jhrs_137_24
- Prins L.I., Smith E., Naaktgeboren Ch.M., Khan M.G., Bilardo C.M., Griffioen W. et al. Inter- and intra-observer reliability in first-trimester uterine artery Doppler measurements. Eur. J. Obstet. Gynecol. Reprod. Biol. 2025; 311: 114058. https://dx.doi.org/10.1016/j.ejogrb.2025.114058
- Benjamini Y., Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society: Series B (Methodological). 1995; 57(1): 289-300. https://dx.doi.org/10.1111/j.2517-6161.1995.tb02031.x
- Сухих Г.Т., Шуршалина А.В. Хронический эндометрит: руководство. М.: ГЭОТАР-Медиа; 2013. 64 с. [Sukhikh G.T., Shurshalina A.V. Chronic endometritis: a guide. Moscow: GEOTAR-Media; 2013. 64 p. (in Russian)].
- Суханов А.А., Дикке Г.Б., Кукарская И.И. Эпидемиология женского бесплодия и опыт восстановления репродуктивной функции у пациенток с хроническим эндометритом в Тюменском регионе. Проблемы репродукции. 2023; 29(3): 98-107. [Sukhanov A.A., Dikke G.B., Kukarskaya I.I. Epidemiology of female infertility and the experience of recovery of reproductive function in patients with chronic endometritis in the tyumen region. Russian Journal of Human Reproduction. 2023; 29(3): 98-107 (in Russian)]. https://dx.doi.org/10.17116/repro20232903198
- Гомболевская Н.А., Марченко Л.А., Муравьева В.В. Современные методы лечения пациенток с хроническим эндометритом. Акушерство и гинекология. 2015; 12: 39-45. [Gombolevskaya N.A., Marchenko L.A., Muravyova V.V. Modern methods of treatment of patients with chronic endometritis. Obstetrics and Gynecology. 2015; (12): 39-45 (in Russian)].
- Chen X., Chen M., Liu M., Qi L., Liu Z., Chen C. et al. Intrauterine infusion of autologous platelet-rich plasma modulates endometrial immune status and improves pregnancy outcomes in patients with persistent chronic endometritis. Front. Immunol. 2025; 16: 1528522. https://dx.doi.org/10.3389/fimmu.2025.1528522
Received 12.01.2026
Accepted 29.01.2026
About the Authors
Irina A. Ozerskaya, Dr. Med. Sci., Professor, Professor at the Department of Ultrasound Diagnostics of the Faculty of Continuing Medical Education of the Medical Institute, Peoples’ Friendship University of Russia named after Patrice Lumumba (RUDN University), 117198, Russia, Moscow, Miklukho-Maklay str., 6, +7(926)606-09-05,ozerskaya_usd@mail.ru, https://orcid.org/0000-0001-8929-6001
Sergey Ya. Porkhovaty, PhD, Ultrasound diagnostics doctor, LLC “Asklepion”, 125222, Russia, Moscow, Dubravnaya str., 41 bldg. 2, https://orcid.org/0009-0002-7263-1568
Gayane G. Kazaryan, PhD, Head of the Ultrasound Department, LLC “Medskan”, 119421, Russia, Moscow, Obrucheva str., 21A, https://orcid.org/0000-0002-1198-8187
Ekaterina V. Ozhogina, PhD, Leading Reproductive Specialist, Nova Clinic Center for Reproduction and Genetics, 119415, Russia, Moscow, Lobachevsky str., 20,
https://orcid.org/0009-0007-5205-2901
Corresponding author: Irina A. Ozerskaya, ozerskaya_usd@mail.ru



