Structural assessment of the fetal brain in late-onset fetal growth restriction using magnetic resonance imaging
Stoliarova E.V., Kholin A.M., Syrkashev E.M., Khodzhaeva Z.S., Gus A.I.
Objective: To assess the volumes of fetal anatomical structures using magnetic resonance imaging (MRI) in pregnant women with late-onset fetal growth restriction (FGR) and small-for-gestational-age (SGA) fetuses.
Materials and methods: This prospective cohort study included 15 pregnant women with late-onset FGR and 15 pregnant women with SGA who underwent MRI. The control group consisted of nine pregnant women with appropriate-for-gestational-age fetuses. Using manual selection of the regions of interest, the volumes of the supratentorial brain and cerebellum were measured, and their ratios were calculated. Quantitative measurements of all the brain structures were converted to percentile values.
Results: The percentile values of supratentorial brain and cerebellum volumes were significantly smaller in the late-onset FGR group (p=0.004 and p<0.001, respectively). There were no statistically significant differences in the volume of the brain structures in the SGA group.
Conclusion: Fetuses with late-onset fetal growth restriction have reduced supratentorial brain and cerebellar volumes, as assessed by MRI, compared with controls. Placental insufficiency in late-onset FGR has a greater effect on brain volume than on birth weight. Further studies are needed to clarify the areas of interest in the brain and to determine their association with the severity and likelihood of reversibility of neurological deficits in newborns and infants with late-onset fetal growth restriction.
Authors' contributions: Stoliarova E.V. – conduct of the study, drafting of the manuscript; Kholin A.M. – conception of the study, editing of the manuscript; Syrkashev E.M. – conception of the study, collection of clinical material; Khodzhaeva Z.S. – supervision of the study, final editing of the manuscript; Gus A.I. – conception of the study, editing of the manuscript.
Conflicts of interest: The authors have no conflicts of interest to declare.
Funding: This article was prepared by a team of authors within the framework of research work (No. EGISU NIOKTR: 121040600408-4) in accordance with the state order of V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia,
for 2023–2024.
Ethical Approval: The study was reviewed and approved by the Research Ethics Committee of the V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia.
Patient Consent for Publication: All patients provided informed consent for the publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available upon request from the corresponding author after approval from the principal investigator.
For citation: Stoliarova E.V., Kholin A.M., Syrkashev E.M., Khodzhaeva Z.S., Gus A.I.
Structural assessment of the fetal brain in late-onset fetal growth restriction using magnetic resonance imaging.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2025; (2): 52-58 (in Russian)
https://dx.doi.org/10.18565/aig.2024.257
Keywords
Late-onset fetal growth restriction (FGR) is a significant issue in modern obstetrics and is associated with a high risk of antenatal fetal death, neonatal mortality, and morbidity [1]. Additionally, late-onset FGR has long-term health implications in childhood and adulthood, particularly in the development of postnatal neurological abnormalities, which can range from functional disorders of the central nervous system to severe mental developmental disorders [2]. Studies conducted during the neonatal period indicated low rates of psychoneurological development, while later assessments in infancy and school age revealed a decline in cognitive abilities among affected children [3].
The pathophysiological pathways and symptomatology of early and late-onset FGR differ significantly. The development of late-onset FGR (after 32 weeks of gestation) is largely linked to aging of the normal placenta. In later pregnancy, a relative increase in the fetus's respiratory needs means that insufficient placental function can lead to hypoxia, even when nutrient supply may be adequate [4]. Chronic hypoxemia and nutrient deficiency prompt adaptations in the fetal cardiovascular system, resulting in preferential shunting of circulating blood to the developing brain [5]. Initially, this mechanism was thought to be primarily neuroprotective; however, recent studies have suggested that the redistribution of blood flow detected by Doppler ultrasound is also associated with neurological deficits in neonates and subsequently in children [6–8]. Although placental insufficiency likely precedes intrauterine brain developmental impairment, its diagnosis remains extremely challenging and its effects on central nervous system development are still poorly understood.
Neurological impairment following intrauterine growth restriction in late pregnancy is linked to specific structural changes in the fetal brain, as detected by magnetic resonance imaging (MRI) [2]. Recent studies have reported significant differences in gyrification, brain and cerebellum volumes, and the degree of cortical asymmetry between small for gestational age (SGA) fetuses and those with late-onset growth restriction [9–12]. Therefore, studying the structural features of the fetal central nervous system in pregnant women with FGR enhances our understanding of the pathogenesis of this condition and broadens our ability to identify new markers for timely detection of fetuses at risk of developing neurological deficits.
This study aimed to assess the volumes of fetal anatomical structures using MRI in pregnant women with late-onset FGR and SGA.
Materials and methods
Prospective recruitment for the study was conducted at the obstetric clinic of the V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 30 patients were examined and divided into two groups. Group I included 15 pregnant women with late-onset FGR, while group II comprised 15 pregnant women with SGA. The control group included nine patients with uncomplicated pregnancies and fetuses appropriate for gestational age (AGA), who had previously undergone MRI due to suspected anatomical features of the pelvis or fetal developmental abnormalities according to ultrasound examination (US), which were not confirmed after birth. Clinical and anamnestic characteristics, data on the course of pregnancy, childbirth, postpartum period, neonatal outcomes, and results from MRI, ultrasound, and Doppler examinations were studied.
The inclusion criteria for the study were singleton pregnancy, gestational age > 32 weeks, and no contraindications for MRI. For the FGR group, the inclusion criteria were an estimated fetal weight (EFW) <10th percentile in combination with impaired blood flow according to Doppler ultrasound, or <3rd percentile with normal Doppler blood flow parameters. The inclusion criteria for the SGA group were EFW <10th percentile and normal blood flow parameters. In the control group, EFW was in the range from 25th to 90th percentiles. The diagnoses of late-onset FGR and SGA were established in accordance with current clinical guidelines approved by the Ministry of Health of the Russian Federation [13].
Exclusion criteria were congenital malformations, chromosomal abnormalities of the fetus, and poor-quality MRI images.
All patients underwent fetal MRI, the only indication for which in groups I and II was suspected to be FGR/SGA. The volume of the supratentorial brain and cerebellum was measured on T2-weighted images (T2WI) by manually delineating the regions of interest and summing the selected regions using the OsiriX MD software for macOS.
The anatomical boundaries of the supratentorial brain were defined by the parenchyma of the frontal, parietal, occipital, and temporal lobes. The measurements did not include brainstem or lateral ventricular cavities. The outer borders of the cerebellum were defined by its hemispheres, including the vermis, whereas the brainstem and fourth ventricle were excluded (Fig. 1).
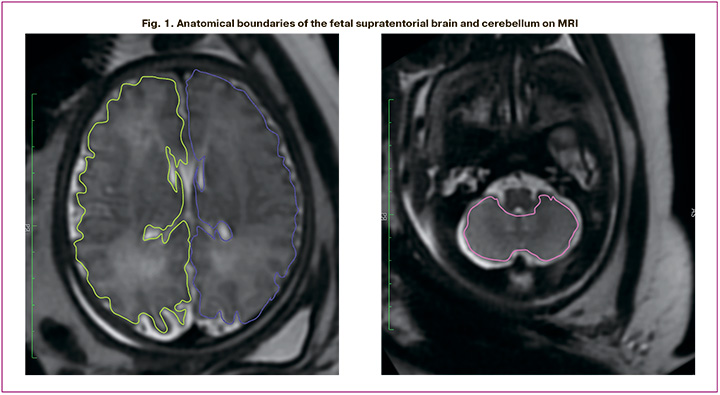
When comparing the study groups, absolute/quantitative measurements of the obtained volumes were transformed into percentiles to normalize the data and eliminate differences in the measurements of fetal brain structures due to varying gestational ages at the time of the MRI. Measurements for each fetal brain structure were transformed according to the growth curves published by Kyriakopoulou V. et al. in 2017, which were derived from the examination of 127 healthy fetuses with body weights corresponding to gestational age [14].
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics 29.0.2.0 (20) software package for macOS. Continuous variables were expressed as mean±standard deviation (SD). Continuous variables were compared between the groups using an independent t-test. Categorical variables were compared using the χ2 test or Fisher's exact test. Statistical significance was set at p<0.05.
Results
Clinical characteristics of the study population
The study included 39 patients: 15 pregnant women in the late-onset FGR group, 15 in the SGA group, and 9 in the control group. The clinical and anamnestic characteristics of the pregnant women, along with the obstetric and perinatal outcomes, are presented in Table 1. In the control group, there were no cases of hypertensive disorders among the pregnant women; all children were born on time, and the average weight of the newborns was 3313.56±228.89 g.
Statistically significant differences were found in maternal preeclampsia, gestational age at delivery, and newborn body weight between the FGR, SGA, and control groups. In the FGR group, the mean newborn body weight was 2129.0±617.30 g, whereas in the SGA group, it was 2583.64±407.31 g (p<0.001).
Analysis of fetal brain structure volumes
The mean volumes of the supratentorial brain and cerebellum, and their ratio, along with the standard deviation and significance level, are presented in Table 2. The mean volume of brain structures was smaller in the FGR group than in the control group, but the results were not statistically significant. However, the mean volume of the supratentorial brain and cerebellum in the SGA group was greater than that in the control group when comparing absolute values. These results may be associated with the gestational age distribution at the time of MRI (Fig. 2). The mean gestational age at the time of MRI was 35.38±3.42 weeks in the FGR group, 35.48±2.45 weeks in the control group, and 37.36±2.13 weeks in the SGA group. No statistically significant differences were observed between cerebellum/supratentorial brain ratios. The mean percentile values of the brain structures, including supratentorial brain volume, cerebellum volume, and their ratio, as well as the standard deviation and significance level, are presented in Table 2 and Figures 3 and 4. When assessing the supratentorial brain and cerebellum, the mean percentile in the FGR group was significantly lower than that in the control group. No statistically significant differences were found between the SGA and control groups.
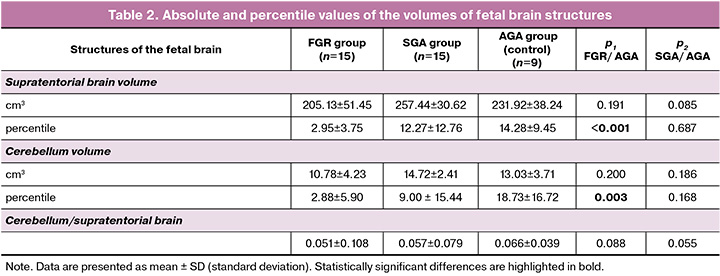
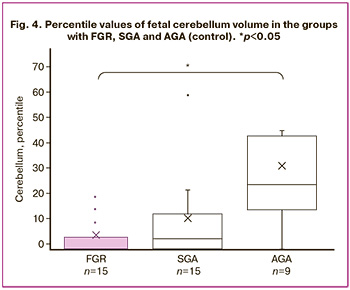
Comparison of brain volumes and birth weight in the FGR and SGA groups
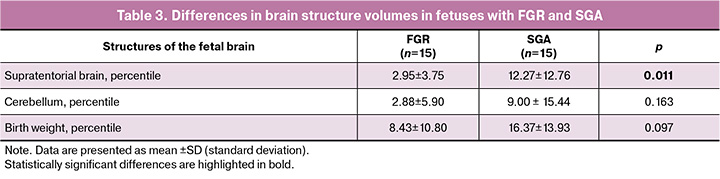
The volumes of brain structures and birth weights in the FGR and SGA groups, along with the significance level, are presented in Table 3. Birth weight was comparable between the groups; however, the supratentorial brain volume was significantly different between the FGR and SGA groups (p=0.011).
Discussion
In our study, we found that, when comparing the absolute volume measurements of the supratentorial brain and cerebellum, the FGR group exhibited lower values; however, these differences were not statistically significant. To eliminate the influence of gestational age, we transformed the absolute volumes of the brain structures into relative units expressed in percentiles based on previously published nomograms. This approach enabled us to identify a significant decrease in the volumes of the supratentorial brain and cerebellum in the FGR group compared to those in the control group, supporting the hypothesis that late-onset FGR affects fetal brain volume.
The supratentorial region is located above the tentorium cerebelli and contains the cerebrum, whereas the infratentorial region, situated below the tentorium cerebelli, houses the cerebellum. Research has established a clear separation of cognitive functionality within and around the tentorium cerebelli [9]. The brain volumes obtained were consistent with the findings reported in previous studies. Several authors have noted changes in the brain structure among infants who experienced FGR in utero, including increased vulnerability of the cerebellum [15–17]. In our findings, we observed a more pronounced decrease in cerebellar volume than in the supratentorial brain when examining percentile values.
In a study by Limperopoulos C. (2016), it was emphasized that the developing cerebellum plays a crucial role in movement, cognition, and social-behavioral adaptation [18]. Additionally, Sanz-Cortes M. et al. (2014) assessed cerebellar volume via MRI in patients with SGA at 37 weeks of gestation and found larger volumes in low-birth-weight fetuses than in those corresponding to gestational age [19]. Notably, in our study, we did not find a significant decrease in brain structures in low-birth-weight fetuses. SGA is generally believed to constitute a group of constitutionally small but healthy fetuses that are not at risk for future neurological deficits [1]. A fundamental difference in the antenatal diagnosis of SGA is the absence of blood flow disorders, as indicated by ultrasound Doppler examination; however, differentiating SGA from FGR remains challenging [20].
Particularly interesting are the findings of our comparison of brain volumes and birth weights between the FGR and SGA groups. Although birth weights (in grams and percentiles) were comparable between the two groups, the supratentorial brain volume was significantly smaller in the FGR group. This suggests that placental insufficiency in late pregnancy may have a more profound impact on brain structure development than on birth weight. Unlike indicators of fetal hypoxemia, fetal size is not a reliable marker of unfavorable perinatal outcomes at full term. This is particularly relevant, as the fetus may not be classified as small based on traditional cutoff values, making the diagnosis of placental insufficiency more challenging in late pregnancy. Fetal Doppler ultrasound examinations, regardless of whether the cerebroplacental ratio or umbilical-cerebral ratio is measured, have demonstrated a stronger association with perinatal fetal or newborn death than with fetal size [21].
The quest for sensitive markers that can identify conditions indicative of suboptimal intrauterine development remains ongoing, as both FGR and SGA infants face risks of neurodevelopmental impairments compared to those born at appropriate gestational ages in the postnatal period [22, 23]. Cognitive and behavioral impairments persist into childhood, adolescence, and adulthood in both FGR and SGA infants [24].
The results of our study should be interpreted with caution in light of these limitations. The small sample size was due to the difficulty in recruiting healthy pregnant women to undergo MRI. The measurements were performed through manual delineation of the brain structures, utilizing the methodology previously described for healthy fetuses. Additionally, because of the accuracy required to identify anatomical boundaries, some images were unsuitable for processing, primarily because of motion artifacts.
Further studies are necessary, including detailed examination of the most vulnerable brain structures. Moreover, it will be crucial to assess the neurological status of both newborns and children. In the future, this will enable the development of biomarkers to predict suboptimal neurodevelopment in fetuses with growth restriction.
Conclusion
Fetuses with late-onset growth restriction exhibited reduced volumes of the supratentorial brain and cerebellum, as assessed by MRI, compared to the control group, while the changes in SGA fetuses were not statistically significant. Our results confirmed the adverse effects of placental insufficiency on the structural development of the brain, particularly pronounced in the prenatally diagnosed phenomenon of cerebral blood flow redistribution. Further studies are needed to establish the relationship between the structural and functional features of the central nervous system and neurological outcomes of newborns and children. A promising approach involves searching for sensitive markers at the antenatal stage that characterize the severity and reversibility of neurological deficits in newborns and infants with late-onset FGR.
References
- Peasley R., Rangel L.A.A., Casagrandi D., Donadono V., Willinger M., Conti G. et al. Management of late-onset fetal growth restriction: pragmatic approach. Ultrasound Obstet. Gynecol. 2023; 62(1): 106-14. https://dx.doi.org/10.1002/uog.26190.
- Dudink I., Hüppi P.S., Sizonenko S.V., Castillo-Melendez M., Sutherland A.E., Allison B.J. et al. Altered trajectory of neurodevelopment associated with fetal growth restriction. Exp. Neurol. 2022; 347: 113885. https://dx.doi.org/10.1016/j.expneurol.2021.113885.
- Malhotra A., Allison B.J., Castillo-Melendez M., Jenkin G., Polglase G.R., Miller S.L. Neonatal morbidities of fetal growth restriction: pathophysiology and impact. Front. Endocrinol. (Lausanne). 2019; 10: 55. https://dx.doi.org/10.3389/fendo.2019.00055.
- Kamphof H.D., Posthuma S., Gordijn S.J., Ganzevoort W. Fetal growth restriction: mechanisms, epidemiology, and management. Matern. Fetal Med. 2022; 4(3): 186-96. https://dx.doi.org/10.1097/FM9.0000000000000161.
- Araujo Júnior E., Zamarian A.C., Caetano A.C., Peixoto A.B., Nardozza L.M. Physiopathology of late-onset fetal growth restriction. Minerva Obstet. Gynecol. 2021; 73(4): 392-408. https://dx.doi.org/10.23736/S2724-606X.21.04771-7.
- Misan N., Michalak S., Kapska K., Osztynowicz K., Ropacka-Lesiak M. Blood-brain barrier disintegration in growth-restricted fetuses with brain sparing effect. Int. J. Mol. Sci. 2022; 23(20): 12349. https://dx.doi.org/10.3390/ijms232012349.
- Benítez-Marín M.J., Marín-Clavijo J., Blanco-Elena J.A., Jiménez-López J., González-Mesa E. brain sparing effect on neurodevelopment in children with intrauterine growth restriction: a systematic review. Children (Basel). 2021; 8(9): 745. https://dx.doi.org/10.3390/children8090745.
- Figueras F., Cruz-Martinez R., Sanz-Cortes M., Arranz A., Illa M., Botet F. et al. Neurobehavioral outcomes in preterm, growth-restricted infants with and without prenatal advanced signs of brain-sparing. Ultrasound Obstet. Gynecol. 2011; 38(3): 288-94. https://dx.doi.org/10.1002/uog.9041.
- Polat A., Barlow S., Ber R., Achiron R., Katorza E. Volumetric MRI study of the intrauterine growth restriction fetal brain. Eur. Radiol. 2017; 27(5): 2110-8. https://dx.doi.org/10.1007/s00330-016-4502-4.
- Bruno C.J., Bengani S., Gomes W.A., Brewer M., Vega M., Xie X. et al. MRI differences associated with intrauterine growth restriction in preterm infants. Neonatology. 2017; 111(4): 317-23. https://dx.doi.org/10.1159/000453576.
- Zheng W., Yan G., Jiang Y., Bao Z., Li K., Deng M. et al. Diffusion-Weighted MRI of the fetal brain in fetal growth restriction with maternal preeclampsia or gestational hypertension. J. Magn. Reson. Imaging. 2024; 59(4): 1384-93. https://dx.doi.org/10.1002/jmri.28861.
- Hutter J., Al-Wakeel A., Kyriakopoulou V., Matthew J., Story L., Rutherford M. Exploring the role of a time-efficient MRI assessment of the placenta and fetal brain in uncomplicated pregnancies and these complicated by placental insufficiency. Placenta. 2023; 139: 25-33. https://dx.doi.org/10.1016/j.placenta.2023.05.014.
- Министерство здравоохранения Российской Федерации. Клинические рекомендации. Недостаточный рост плода, требующий предоставления медицинской помощи матери (задержка роста плода). 2022. [Ministry of Health of the Russian Federation. Clinical guidelines. Insufficient growth of the fetus, requiring the provision of medical care to the mother (fetal growth retardation). 2022. (in Russian)].
- Kyriakopoulou V., Vatansever D., Davidson A., Patkee P., Elkommos S., Chew A. et al. Normative biometry of the fetal brain using magnetic resonance imaging. Brain Struct. Funct. 2017; 222(5): 2295-307. https://dx.doi.org/10.1007/s00429-016-1342-6.
- Husen S.C., Koning I.V., Go A.T.J.I., van Graafeiland A.W., Willemsen S.P., Groenenberg I.A.L. et al. Three-dimensional ultrasound imaging of fetal brain fissures in the growth restricted fetus. PLoS One. 2019; 14(5): e0217538. https://dx.doi.org/10.1371/journal.pone.0217538.
- Andescavage N., duPlessis A., Metzler M., Bulas D., Vezina G., Jacobs M. et al. In vivo assessment of placental and brain volumes in growth-restricted fetuses with and without fetal Doppler changes using quantitative 3D MRI. J. Perinatol. 2017; 37(12): 1278-84. https://dx.doi.org/10.1038/jp.2017.129.
- Egaña-Ugrinovic G., Sanz-Cortes M., Figueras F., Bargalló N., Gratacós E. Differences in cortical development assessed by fetal MRI in late-onset intrauterine growth restriction. Am. J. Obstet. Gynecol. 2013; 209(2): 126.e1-8. https://dx.doi.org/10.1016/j.ajog.2013.04.008.
- Limperopoulos C. The vulnerable immature cerebellum. Semin. Fetal. Neonatal. Med. 2016; 21(5): 293-4. https://dx.doi.org/10.1016/J.SINY.2016.07.002.
- Sanz-Cortes M., Egaña-Ugrinovic G., Zupan R., Figueras F., Gratacos E. Brainstem and cerebellar differences and their association with neurobehavior in term small-for-gestational-age fetuses assessed by fetal MRI. Am. J. Obstet. Gynecol. 2014; 210(5): 452.e1-8. https://dx.doi.org/10.1016/j.ajog.2013.12.008.
- Martinez J., Boada D., Figueras F., Meler E. How to define late fetal growth restriction. Minerva Obstet. Gynecol. 2021; 73(4): 409-14. https://dx.doi.org/10.23736/S2724-606X.21.04775-4.
- Thilaganathan B. Ultrasound fetal weight estimation at term may do more harm than good. Ultrasound Obstet. Gynecol. 2018; 52(1): 5-8. https://dx.doi.org/10.1002/uog.19110.
- Andescavage N., Bullen T., Liggett M., Barnett S.D., Kapse A., Kapse K. et al. Impaired in vivo feto-placental development is associated with neonatal neurobehavioral outcomes. Pediatr. Res. 2023; 93(5): 1276-84. https://dx.doi.org/10.1038/s41390-022-02340-0.
- Graz M.B., Tolsa J.F., Fumeaux C.J.F. Being small for gestational age: does it matter for the neurodevelopment of premature infants? A cohort study. PLoS One. 2015; 10(5): e0125769. https://dx.doi.org/10.1371/journal.pone.0125769.
- Vollmer B., Edmonds C.J. School age neurological and cognitive outcomes of fetal growth retardation or small for gestational age birth weight. Front. Endocrinol. (Lausanne). 2019; 10: 186. https://dx.doi.org/10.3389/fendo.2019.00186.
Received 21.10.2024
Accepted 30.01.2025
About the Authors
Elizaveta V. Stoliarova, PhD student, 1st Obstetric Department of Pregnancy Pathology, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, ev_stolyarova@oparina4.ru, https://orcid.org/0009-0001-2049-3119Alexey M. Kholin, PhD, Head of the Department of Telemedicine, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, a_kholin@oparina4.ru, https://orcid.org/0000-0002-4068-9805
Egor M. Syrkashev, PhD, Senior Researcher at the Radiology Department, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow,
Ac. Oparin str., 4, e_syrkashev@oparina4.ru, https://orcid.org/0000-0003-4043-907X
Zulfiya S. Khodzhaeva, Dr. Med. Sci, Professor, Deputy Director, Institute of Obstetrics, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4, z_khodzhaeva@oparina4.ru, https://orcid.org/0000-0001-8159-3714
Aleksandr I. Gus, Dr. Med. Sci., Chief Researcher at the Department of Ultrasound and Functional Diagnostics, V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparin str., 4; Head of the Department of Ultrasound Diagnostics, Medical Institute of Patrice Lumumba Peoples’ Friendship University of Russia, a_gus@oparina4.ru, https://orcid.org/0000-0003-1377-3128



