V.N. Gorodkov Ivanovo Research Institute of Maternity and Childhood, Ministry of Health of Russia, Ivanovo
Objective. To compare the efficacy of micronized progesterone formulations, such as prajisan and utrogestan as vaginal capsules in supporting the luteal phase in in vitro fertilization (IVF) cycles.
Subjects and methods. This was a single-center observational retrospective study. Women who had been planned to have IVF were randomized to use prajisan or utrogestan in a daily dose of 600 mg as vaginal capsules. The main parameter of achieving the goal was to estimate the number of positive pregnancy tests on day 15 after embryo transfer, as well as abdominal and transvaginal ultrasound findings.
Results. The investigation enrolled 264 patients, of whom 149 women received prajisan and 115 had utrogestan. Pragisan was as effective as the latter: the cumulative pregnancy rate was 42.95% (64) and 41.74% (48) in the prajisan and utrogestan groups, respectively. The rate of pregnancy ending in childbirth was 32.21% (48) and 33.04% (38) in these groups, respectively. Prajisan and utrogestan had a similar safety profile. No undesirable adverse reactions were recorded in the patients of both compared groups.
Conclusion. The study demonstrated that intravaginal prajisan was as effective as intravaginal utrogestan. Prajisan can successfully support the luteal phase in IVF programs.
in vitro fertilization
pregnancy rate
birth rate
vaginal micronized progesterone
В настоящее время актуальна проблема бесплодия и невынашивания беременности. Причиной бесплодия в паре могут быть нарушения мужской либо женской репродуктивной функции, а примерно у трети пар бесплодие сочетанное, то есть вызвано одновременно и мужским и женским факторами. Вспомогательные репродуктивные технологии (ВРТ) позволяют во многих случаях справиться с проблемой бесплодия. Поддержка лютеиновой фазы при экстракорпоральном оплодотворении (ЭКО) проводится с целью компенсации недостаточности прогестерона, вызванной стимуляцией яичников [1–4]. Утрожестан – широко известный микронизированный прогестерон, используемый для поддержания лютеиновой фазы в циклах ЭКО [5, 6]. В настоящее время в лечебных учреждениях используется также препарат праджисан – микронизированный вагинальный прогестерон, который также применяется в программах ЭКО.
Материал и методы исследования
Проведено одноцентровое наблюдательное ретроспективное исследование, в которое были включены 264 женщины. 149 женщин входили в группу получавших праджисан в дозировке 600 мг, 115 женщин получали утрожестан в дозировке 600 мг. В данное исследование включали женщин в возрасте от 21 до 43 лет; с индексом массы тела (ИМТ) от 17 до 37 кг/м², с документально подтвержденным бесплодием в анамнезе (невозможность зачатия на протяжении более чем 1 года, либо при наличии двусторонней окклюзии или отсутствии маточных труб), которым планировали процедуру ЭКО с переносом эмбрионов (перенос одного или двух эмбрионов). Уровень фолликулостимулирующего гормона (ФСГ) составлял ≤15 МЕ/л, при этом показатель уровня антимюллерова гормона (АМГ) составлял от ≥0,1 до ≤23 нг/мл (норма 2,1–7,3 нг/мл). Основными критериями исключения являлись: мужской фактор бесплодия, наличие нарушений со стороны сердечно-сосудистой системы, дыхательной, мочеполовой систем, желудочно-кишечного тракта и печени, гематологические и иммунологические заболевания, а также нарушения со стороны ЛОР-органов и органов зрения (область голова, уши, нос, глотка), заболевания кожи, нарушения со стороны нервной системы, нарушений психики, симптомы аллергических заболеваний, недавно перенесенные обширные хирургические вмешательства (менее 3 месяцев назад); злоупотребление в настоящее время или в недавнем прошлом какими-либо психоактивными веществами, включая алкоголизм и табакокурение; проведение химиотерапии или лучевой терапии в анамнезе; прием других препаратов прогестерона.
Главная цель исследования состояла в том, чтобы продемонстрировать как минимум сопоставимую эффективность праджисана в форме вагинальных капсул по 600 мг в сутки для поддержания лютеиновой фазы в циклах ЭКО. Основным параметром достижения цели являлась оценка количества положительных тестов на беременность на 15-й день после переноса эмбриона(ов), данных абдоминального и трансвагинального УЗИ.
Полученные результаты обрабатывались традиционными методами статистического анализа. Различия между группами определены при помощи критериев t Стьюдента и Манна–Уитни, между относительными показателями по критерию χ2. Различия считались достоверными при р<0,05.
 Результаты исследования и обсуждение
Результаты исследования и обсуждение
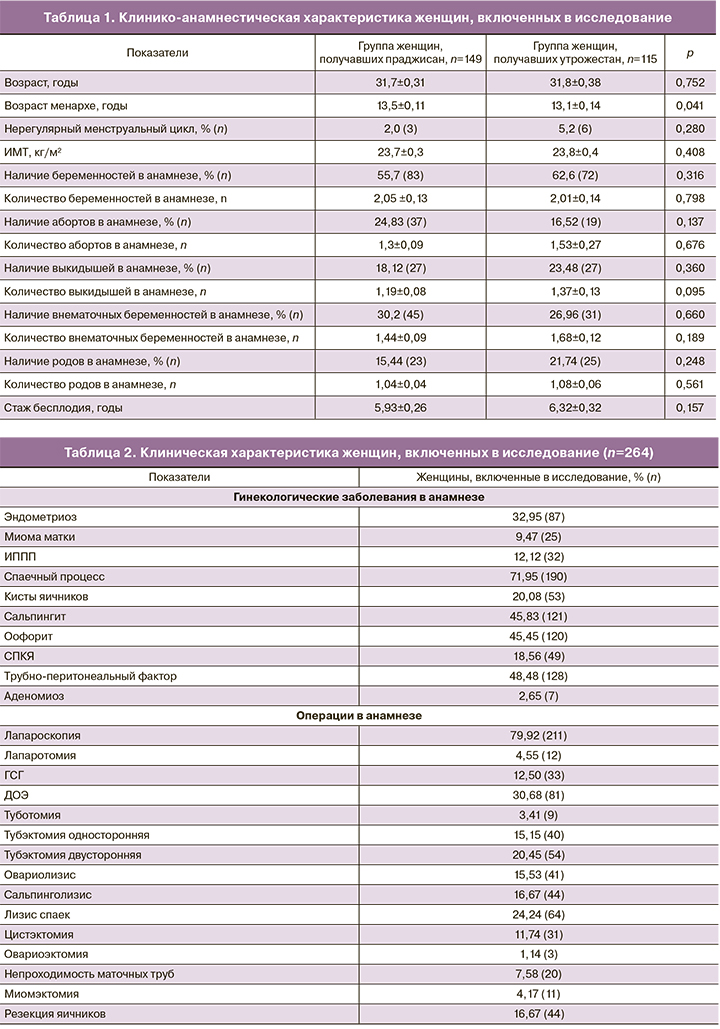
В общей сложности было скринировано 264 женщины, из которых 149 женщин входили в группу получавших праджисан в дозировке 600 мг, 115 женщин получали утрожестан в дозировке 600 мг. Исходные характеристики были схожими в обеих группах (табл. 1, 2).
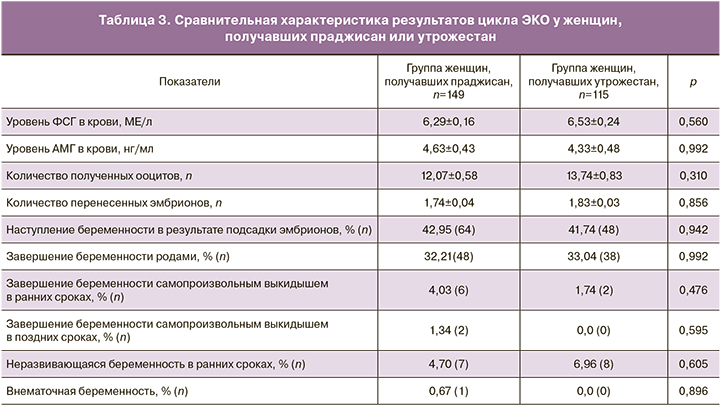
Главная цель данного исследования была достигнута: праджисан продемонстрировал сопоставимую с утрожестаном эффективность в поддержании лютеиновой фазы в циклах ЭКО, наступления беременности после переноса эмбриона(ов). Совокупная частота наступления беременности составила 42,95% (64) и 41,74% (48) в группах праджисана и утрожестана соответственно (табл. 3). Количество перенесенных эмбрионов в обеих группах было одинаковым. Частота завершения беременности родами составила 32,21% (48) и 33,04% (38) для групп праджисана и утрожестана соответственно (рисунок). Частота ненаступления беременности была одинаковой, и составила 57,05% (85) и 58,26% (67) для групп праджисана и утрожестана соответственно.
Нежелательных побочных реакций у пациенток обеих сравниваемых групп не зарегистрировано.
Результаты, полученные в ходе исследования, предоставляют убедительную базу в пользу применения праджисана для поддержки лютеиновой фазы в рамках программ ВРТ. Дозировка праджисана в этом исследовании составила 600 мг в сутки интравагинально, была выбрана на основании существующих рекомендаций и данных ранее проведенных клинических исследований [7–10]. Проведена сравнительная оценка средней стоимости данных препаратов, в ходе которой было установлено, что праджисан является более доступным (предельная отпускная цена производителя по данным ГРЛС (2018 г.) составляет от 218,05 до 314,79 рублей), в отличие от утрожестана (предельная отпускная цена производителя – от 341,42 до 364,40 рублей).
Заключение
Таким образом, исследование продемонстрировало, что интравагинальное применение праджисана не менее эффективно и безопасно по сравнению с интравагинальным применением утрожестана в отношении достижения главной цели исследования – частоты наступления беременности и завершения ее родами при более низкой стоимости лечения.
1. Иакашвили С.Н., Самчук П.М. Ультразвуковая и лабораторная диагностика плацентарной недостаточности у беременных после экстракорпорального оплодотворения в зависимости от формы бесплодия. Российский вестник акушера-гинеколога. 2017; 17(6): 44-8. [Iakashvili S.N., Samchuk P.M. Ultrasonic and laboratory diagnostics of placental insufficiency in pregnant women after extracorporeal fertilization, depending on the form of infertility. Rossiyskiy vestnik akushera-ginekologa. 2017; 17 (6): 44-8. (in Russian)]
2. Michnova L., Dostal J., Kudela M., Hamal P., Langova K. Vaginal use of micronized progesterone for luteal support.A randomized study comparing Utrogestan and Crinone 8. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech. Repub. 2017; 161(1): 86-91.
3. Palomba S., Santagni S., La Sala G.B. Progesteron administration for luteaphase deficiency in human reproduction: an old or new issue? J. Ovarian Res. 2015; 8: 77.
4. Practice Committee of American Society for Reproductive Medicine in collaboration with Society for Reproductive Endocrinology and Infertility.
5. Progesterone supplementation during the luteal phase and in early pregnancyin the treatment of infertility: an educational bulletin. Fertil. Steril. 2008; 90(5, Suppl.): S150-3.
6. Галлямова Е.М., Перминова С.Г., Дуринян Э.Р., Митюрина Е.В. Современные подходы к ведению лютеиновой фазы в программе экстракорпорального оплодотворения. Акушерство и гинекология. 2014; 1: 15-22. [Gallyamova E.M., Perminova S.G., Durinyan E.R., Mityurina E.V. Current approaches to luteal phase management in an in-vitro fertilization program. Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2014; (1): 15-22. (in Russian)]
7. Zhu X., Ye H., Fu Y. Use of Utrogestan during controlled ovarian hyperstimulation in normally ovulating women undergoing in vitro fertilization or intracytoplasmic sperm injection treatments in combination with a “freeze all” strategy: a randomized controlled dose-finding study of 100 mg versus 200 mg. Fertil. Steril. 2017; 107(2): 379-86.
8. Савельева Г.М., Сухих Г.Т., Серов В.Н., Радзинский В.Е., Манухин И.Б., ред. Гинекология. Национальное руководство. 2-е изд. М.: ГЭОТАР-Медиа; 2017. 1008с. [Savelyeva G.M., Sukhikh G.T., Serov V.N., Radzinsky V.E., Manukhin I.B., ed. Gynecology. National guideline. 2nd ed. Moscow: GEOTAR-Media; 2017. 1008p. (in Russian)]
9. Tournaye H., Sukhikh G.T., Kahler E., Griesinger G. A Phase III randomized controlled trial comparing the efficacy, safety and tolerability of oral dydrogesterone versus micronized vaginal progesterone for luteal support in in vitro fertilization. Hum. Reprod. 2017; 32(5): 1019-27.
10. Child T., Leonard S.A., Evans J.S., Lass A. Systematic review of the clinical efficacy of vaginal progesterone for luteal phase support in assisted reproductive technology cycles. Reprod. Biomed. Online. 2018; Feb. 22.
11. Gün İ., Özdamar Ö., Şahin S., Çetingöz E., Sofuoğlu K. Progesterone vaginal capsule versus vaginal gel for luteal support in normoresponder women undergoing long agonist IVF/ICSI cycles. Ginekol. Pol. 2016;87(5): 372-7.
Received 12.04.2018
Accepted 28.04.2018
Malyshkina Anna Ivanovna, MD, director of V.N. Gorodkov Ivanovo Research Institute of Maternity and Childhood, Ministry of Health of Russia; chief obstetrician-gynecologist of the Central Federal District. 153045, Russia, Ivanovo, Pobedy str. 20. Tel.: +74932336263. E-mail:
ivniimid@inbox.ru
Nazarov Sergey Borisovich, MD, professor, deputy director of V.N. Gorodkov Ivanovo Research Institute of Maternity and Childhood, Ministry of Health of Russia.
153045, Russia, Ivanovo, Pobedy str. 20. Tel.: +74932338220. E-mail:
ivniimid@inbox.ru
Svetlana S. Semenenko, Candidate of Medical Science, Head of the Department of V.N. Gorodkov Ivanovo Research Institute of Maternity and Childhood,
Ministry of Health of Russia. 153045, Russia, Ivanovo, Pobedy str. 20. Tel.: +74932338220. E-mail:
ivniimid@inbox.ru
Kozelkova Elena Vladimirovna, clinical resident of 2nd year of training in the specialty “Obstetrics and Gynecology”, V.N. Gorodkov Ivanovo Research Institute
of Maternity and Childhood, Ministry of Health of Russia. 153045, Russia, Ivanovo, Pobedy str. 20. Tel.: +74932338220. E-mail:
ivniimid@inbox.ru
For citations: Malyshkina A.I., Nazarov S.B., Semenenko S.S., Kozelkova E.V.
Comparative evaluation of the efficacy and safety of micronized progesterone
formulations in assisted reproductive technology programs.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2018; (5): 122-6. (in Russian)
https://dx.doi.org/10.18565/aig.2018.5.122-126


 Результаты исследования и обсуждение
Результаты исследования и обсуждение


