1) P.A. Hertsen Moscow Oncology Research Institute – Branch of the National Medical Research Radiological Center, Ministry of Health of Russia, Moscow, Russia;
2) Peoples’ Friendship University of Russia, Moscow, Russia;
3) I.M. Sechenov First Moscow State Medical University, Ministry of Health of Russia (Sechenov University), Department of Oncology, Radiotherapy and Plastic Surgery, Moscow, Russia
Neoadjuvant polychemotherapy (NACT) is currently the cornerstone in the treatment of locally advanced breast cancer (BC) and highly chemosensitive tumors, such as triple-negative and HER/neu-positive types. The lack of clear consensus on the optimal method of breast reconstruction after NACT has resulted in a wide variety of techniques used in practice. The factors which influence the choice of surgical intervention also remain unclear.
The databases and systems, namely PubMed, Cochrane Library, eLibrary have been searched for articles published from 2012 to 2023; this review includes the results of 27 studies.
The information about the possibility of performing and expanding indications for organ-preserving treatment, performing reconstructive plastic surgery, including one-stage reconstructions using allomaterials after NACT reflects current national and global trends in the treatment of patients with breast cancer. When performing a one-stage reconstruction, various methods of reconstruction can be used: reconstruction with own tissues, reconstruction with the use of allomaterials, reconstruction with a thoracodorsal flap in combination with an endoprosthesis/tissue expander, etc. There are the following indications for performing a mastectomy with one-stage or two-stage reconstruction after NACT: the absence of a tumor response to NACT (NR); in case of a partial response, the ratio of the size of the residual tumor and the size of the breast does not allow performing an organ–preserving operation (OPO) and, therefore, achieving a satisfactory aesthetic result; if an interstitial marker is not placed in the tumor node before NACT (in CR and PR); the patient does not want an OPO, but at the same time she has a wish to maintain a satisfactory aesthetic result after surgery.
Conclusion: Performing one-stage breast reconstruction using allomaterials (DTI-reconstruction) is safe in term of oncology and it does not increase the incidence of surgical complications. The esthetic results of the operation are also comparable with similar parameters of patients who did not undergo NACT at the first stage.
Authors’ contributions: Zikiryakhodzhaev A.D., Bosieva A.R. – developing the concept and design of the study; Bosieva A.R. – collecting and processing data, writing the text; Zikiryakhodzhaev A.D. – editing the text.
Conflicts of interest: Authors declare lack of the possible conflicts of interest.
Funding: The study was conducted without sponsorship.
For citation: Zikiryakhodzhaev A.D., Bosieva A.R. Neoadjuvant chemotherapy and DTI-reconstruction in breast cancer patients.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2024; (2): 31-36 (in Russian)
https://dx.doi.org/10.18565/aig.2023.252
breast cancer
neoadjuvant chemotherapy
one-stage breast reconstruction
allomaterials
Неоадъювантная полихимиотерапия (НАПХТ) на сегодняшний день представляет собой краеугольный камень в лечении местно-распространенного рака молочной железы (РМЖ) и опухолей с высокой химиочувствительностью, таких как тройной негативный и Her/neu-позитивные типы [1–5]. Показаниями для проведения НАПХТ являются размер опухолевого узла, молекулярно-биологический тип, метастатическое поражение лимфатических узлов и степень злокачественности. НАПХТ играет ключевую роль в уменьшении размера опухоли, увеличении возможности проведения органосохраняющего хирургического лечения по сравнению с реконструктивно-пластическими операциями [6, 7] и, таким образом, уменьшении необходимости выполнения подмышечно-подлопаточно-подключичной лимфаденэктомии [2, 8, 9]. Другим преимуществом НАПХТ является оценка ответа in vivo [2, 10, 11].
Кроме того, существует большая вариабельность ответа РМЖ на НАПХТ? и, соответственно, точная оценка ответа на лечение имеет решающее значение при определении объема лечения и необходимости выполнения дальнейших хирургических вмешательств. Тем не менее установление полного патоморфологического ответа (pCR), ассоциированного с прогнозом заболевания, позволяет онкологам обеспечить персонализированный подход в определении оптимального объема хирургического лечения [12, 13].
Методы визуализации (ультразвуковое исследование молочных желез, рентгеномаммография, магнитно-резонансная томография) играют важную роль в оценке ответа опухоли на НАПХТ и принятии решения врачом-онкологом относительно объема оперативного лечения.
Несмотря на то что НАПХТ позволяет повысить частоту выполнения органосохраняющих операций, остаются больные, у которых данный объем хирургического лечения не является оптимальным после НАПХТ. В таких случаях одномоментная реконструкция молочной железы по-прежнему остается вариантом, обеспечивающим лучшее качество жизни, высокую удовлетворенность пациента, хороший косметологический результат и психоэмоциональное благополучие.
Показаниями к выполнению мастэктомии с одномоментной одноэтапной или двухэтапной реконструкцией после НАПХТ являются:
- отсутствие ответа опухоли на НАПХТ (NR);
- при частичном ответе – соотношение размера резидуальной опухоли и размера молочной железы, не позволяющие выполнить органосохраняющую операцию (ОСО) и, соответственно, достичь удовлетворительного эстетического результата;
- если до НАПХТ не была установлена внутритканевая метка в опухолевый узел (при CR и PR);
- нежелание пациентки выполнить ОСО, но при этом сохранить удовлетворительный эстетический результат после операции.
Отсутствие четкого консенсуса относительно оптимального способа реконструкции молочной железы после НАПХТ привело к большому разнообразию используемых методик [14–16]. Также остаются неясными факторы, влияющие на выбор оперативного вмешательства.
При выполнении одномоментной реконструкции могут быть использованы различные способы: собственными тканями, с использованием алломатериалов, реконструкция торакодорзальным лоскутом в комбинации с эндопротезом/тканевым экспандером и т.д.
В ряде исследований продемонстрированы эквивалентные онкологические результаты у пациентов, которым выполнена DTI-реконструкция (Direct-to-Implant, одномоментная реконструкция молочной железы с использованием силиконового эндопротеза) и мастэктомия без реконструкции после НАПХТ. Так, Monrigal E. в своем исследовании отметил в 46 (21,9%) случаях развитие ранних осложнений после DTI-реконструкций: 20 липонекрозов, 9 инфицирований послеоперационных ран, 6 гематом, которые потребовали ре-операций в 23 случаях. Большинство липонекрозов отмечено при реконструкции TRAM-лоскутом (p=0,000004). У 55 пациентов отмечены поздние осложнения (26,2%), такие как капсулярная контрактура, инфицирование, протрузии, что в 23,6% (14) случаев потребовало выполнения реопераций [17]. Donker M. et al. на основании проведенного исследования установили, что нет статистически значимой разницы в показателях хирургических осложнений среди больных, которым выполнена DTI-реконструкция с/без проведением НАПХТ (p≥0,05) (табл. 1) [18].
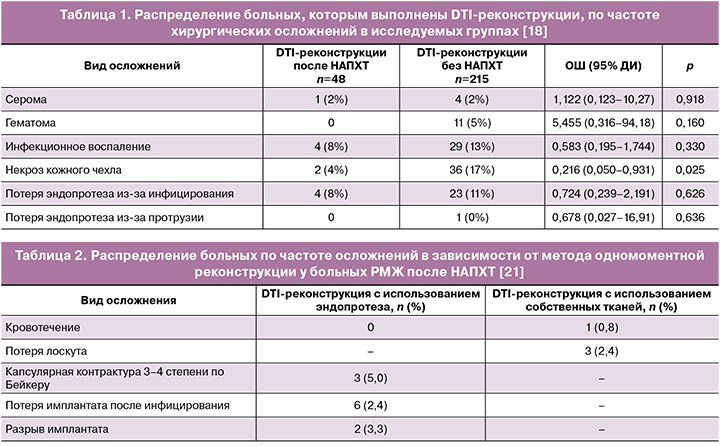
Chi W. в 2023 г. представил результаты исследования, согласно которому возраст пациента и стадия заболевания являются основополагающими при планировании одномоментной реконструкции после НАПХТ. Всего в исследование включены 1651 больных, которые получили НАПХТ, из которых 247 (15,0%) выполнены DTI-реконструкции, а 1404 – радикальные мастэктомии. Пациенты в группе DTI-реконструкции были моложе (P<0,001), имели более низкий индекс массы тела (P<0,001) и меньшую распространенность РМЖ (P=0,003) в сравнении с обычными мастэктомиями. Пациенты в группе одномоментной реконструкции с использованием собственных тканей были старше (P<0,001) и имели более высокий индекс массы тела (P=0,007), больший размер опухоли (P=0,024). Клинически значимые осложнения, которые требовали повторного хирургического вмешательства, чаще встречались в группе одномоментных реконструкций силиконовым эндопротезом в сравнении с использованием собственных тканей (P=0,039). Авторами отмечено, что наиболее оптимальным способом одномоментной реконструкции является использование собственных тканей [19].
Авторами многих исследований отмечено, что существенное влияние на принятие решения относительно способа выполнения одномоментной реконструкции оказывает лучевая терапия. Показано, что при DTI-реконструкции выше частота капсулярной контрактуры и хирургических осложнений. Jagsi R. et al. (2018) проведено крупное проспективное исследование с включением 2247 пациентов, на основании которого авторами отмечена большая удовлетворенность пациентов эстетическими результатами операции после одномоментной реконструкции собственными тканями [20].
Безопасность выполнения DTI-реконструкции после НАПХТ доказана авторами ряда исследований. Beugels J. et al. проанализировали частоту осложнений после одномоментных реконструкций DIEP-лоскутом у больных, которым на первом этапе проведена НАПХТ. Установлено, что частота хирургических осложнений составила 10,4% со стороны реципиентного участка и 22,2% – для донорского участка. Авторами не отмечена статистически значимая разница в частоте осложнений между больными, которым проведена на первом этапе НАПХТ, и пациентами, которым она не проводилась (табл. 2) [21]. Ishiba T. et al. проанализировали результаты лечения 909 пациентов, перенесших одномоментные реконструкции с использованием эндопротеза и собственными тканями, из которых 43 получили на первом этапе НАПХТ. Установлено, что частота осложнений составила 7% при одномоментных реконструкциях после НАПХТ против 3,6%, которым не проводилась предоперационная лекарственная терапия (р=0,25). Авторы отмечают безопасность DTI-реконструкции после НАПХТ [22].
Naoum G.E. et al. проанализировали данные 300 больных, которым проведена НАПХТ с последующим выполнением реконструктивно-пластических операций и проведением адъювантной лучевой терапии. Авторами отмечено, что использование тканевых экспандеров/имплантатов было ассоциировано с повышенным риском развития инфекционных осложнений в сравнении с реконструкцией собственными тканями (ОШ 8,1; 95% ДИ 1,7–38,8; P=0,009), что позволяет предположить, что частота осложнений могла быть выше из-за необходимости выполнения повторных операций при замене экспандера на эндопротез [23].
Sabitovic A. et al. провели систематический обзор метаанализа, посвященного оценке результатов одномоментных реконструкций молочной железы у пациенток, которые ранее получили НАПХТ, в сравнении с контрольной группой [24]. В метаанализ были включены 8 исследований с участием 51 731 пациентки, из которых в 5161 случае проведена НАПХТ. Авторами не отмечено статистически значимого влияния НАПХТ на частоту осложнений после DTI-реконструкций (ОШ=1,35; 95% ДИ 0,96–1,91; p=0,09), в том числе на частоту некроза кожного лоскута (ОШ=1,39; 95% ДИ 0,61–3,17; p=0,44) или частоту повторных операций (ОШ=1,09; 95% ДИ 0,87–1,37; р=0,45). При подгрупповом анализе при выполнении одномоментной реконструкции молочной железы с использованием силиконового эндопротеза после НАПХТ отмечена меньшая частота осложнений и неудачных эстетических результатов по сравнению с двухэтапной реконструкцией тканевым экспандером/имплантатом (ОШ=0,43, 95% ДИ 0,26–0,71, р=0,0011) [24].
Yang J.-R. et al. в 2021 г. представили результаты исследования с включением 269 пациентов. Средний срок наблюдения составил 46,3 месяца. Из 269 в 46 случаях проведена НАПХТ; чаще всего выполнялись одномоментные двухэтапные реконструкции с использованием тканевого экспандера/имплантата в сравнении с одномоментной одноэтапной реконструкцией силиконовым эндопротезом непосредственно с использованием имплантата (p<0,001). Частота осложнений, а также удовлетворенность эстетическими результатами в группе НАПХТ и без проведения НАПХТ были сопоставимы (р<0,001) (табл. 3–5) [25].
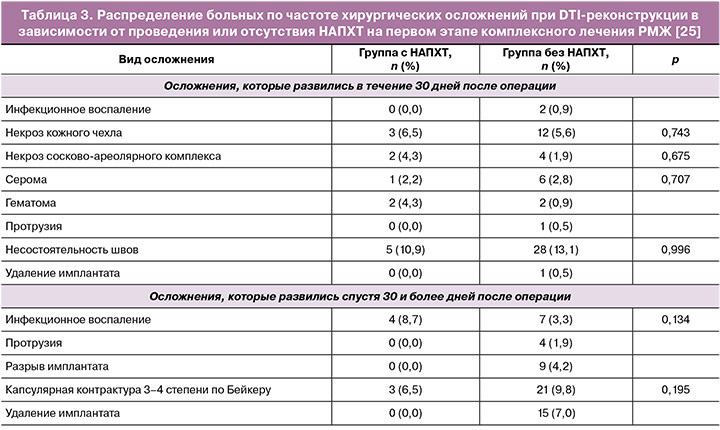
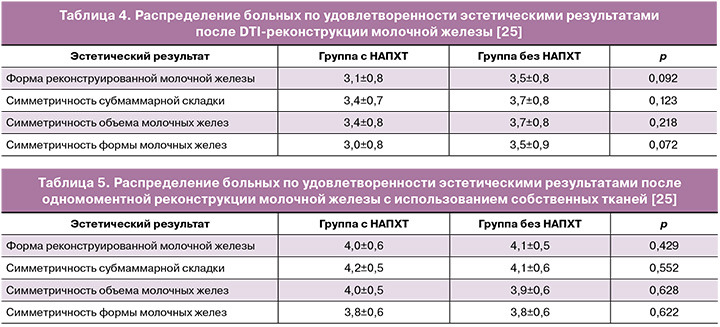
Авторами также не отмечено статистически значимой разницы в проведении/отсутствии НАПХТ и ее влиянии на удовлетворенность эстетическими результатами при одномоментной реконструкции силиконовыми эндопротезами и собственными тканями (табл. 3, 4) [25].
Bowen M.E. et al. представили результаты крупного когортного исследования, на основании которого авторы отметили, что НАПХТ не повышает риск развития хирургических осложнений [26]. Аналогичным образом, Beugels J. et al. сообщили о сопоставимой частоте послеоперационных осложнений у пациентов, получавших и не получавших НАПХТ с последующим выполнением DTI-реконструкции с использованием DIEP лоскута [27].
Заключение
Выполнение одномоментной реконструкции молочной железы с использованием алломатериалов (DTI-реконструкция) является безопасным с онкологической точки зрения, не повышает частоту развития хирургических осложнений. Эстетические результаты операции также сопоставимы с аналогичными показателями у пациентов, которым не проводилась НАПХТ на первом этапе.
- Бересток Т.С., Зикиряходжаев А.Д., Ермощенкова М.В., Аблицова Н.В., Онофрийчук И.М., Власова М.Ю., Трошенков Е.А. Осложнения после одномоментной одноэтапной и двухэтапной реконструкции имплантатами у больных раком молочной железы при проведении комбинированного/комплексного лечения. Онкология. Журнал им. П.А. Герцена. 2023; 12(3): 54-61. [Berestok T.S., Zikiryakhodzhaev A.D., Ermoshchenkova M.V., Ablitsova N.V., Onofriychuk I.M., Vlasova M.Yu., Troshenkov E.A. Complications after simultaneous one-stage and two-stage reconstruction with implants in patients with breast cancer during combined/combination treatment. P.A. Herzen Journal of Oncology. 2023; 12(3): 54-61. (in Russian)]. https://dx.doi.org/10.17116/onkolog20231203154.
- Зикиряходжаев А.Д., Фролова М.А., Рассказова Е.А., Глазкова Е.В. Лечение тройного негативного подтипа рака молочной железы. Опухоли женской репродуктивной системы. 2017; 13(2): 20-6. [Zikiryakhodzhaev A.D., Frolova M.A., Rasskazova E.A., Glazkova E.V. Treatment of triple-negative breast cancer. Tumors of Female Reproductive System. 2017; 13(2): 20-6. (in Russian)]. https://dx.doi.org/10.17650/1994-4098-2017-13-2-20-26.
- Токаев В.К., Зикиряходжаев А.Д., Трошенков Е.А., Рассказова Е.А., Поляк М.А., Никитина Е.А., Малик Д.С. Реконструктивно-пластические операции у больных раком молочной железы со стадиями T1-3N1-3M0. Современная наука: актуальные проблемы теории и практики. Серия: Естественные и технические науки. 2023; 3: 242-8. [Tokaev V.K., Zikiryahodzhaev A.D., Troshenkov E.A., Rasskazova E.A., Polyak M.A., Nikitina E.A., Malik DS. Reconstructive plastic surgery in patients with breast cancer stages T1-3N1-3M0. Modern Science: Actual Problems of Theory and Practice. Series "Natural and Technical Sciences". 2023; (3): 242-8. (in Russian)]. https://dx.doi.org/10.37882/2223–2966.2023.03.36.
- Cardoso F., Kyriakides S., Ohno S., Penault-Llorca F., Poortmans P., Rubio I.T. et al. Early breast cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019; 30(8): 1194-220. https://dx.doi.org/10.1093/annonc/mdz173.
- Burstein H.J., Curigliano G., Loibl S., Dubsky P., Gnant M., Poortmans P. et al. Estimating the benefits of therapy for early-stage breast cancer: The St. Gallen International Consensus Guidelines for the primary therapy of early breast cancer 2019. Ann. Oncol. 2019; 30(10): 1541-57. https://dx.doi.org/10.1093/annonc/mdz235.
- Puig C.A., Hoskin T.L., Day C.N., Habermann E.B., Boughey J.C. National trends in the use of neoadjuvant chemotherapy for hormone receptor-negative breast cancer: A National Cancer Data Base Study. Ann. Surg. Oncol. 2017; 24(5): 1242-50. https://dx.doi.org/10.1245/s10434-016-5733-y.
- Killelea B.K., Yang V.Q., Mougalian S., Horowitz N.R., Pusztai L., Chagpar A.B., Lannin D.R. Neoadjuvant chemotherapy for breast cancer increases the rate of breast conservation: results from the National Cancer Database. J. Am. Coll. Surg. 2015; 220(6): 1063-9. https://dx.doi.org/10.1016/j.jamcollsurg.2015.02.011.
- Mougalian S.S., Soulos P.R., Killelea B.K., Lannin D.R., Abu-Khalaf M.M., DiGiovanna M.P. et al. Use of neoadjuvant chemotherapy for patients with stage I to III breast cancer in the United States. Cancer. 2015; 121(15): 2544-52. https://dx.doi.org/10.1002/cncr.29348.
- Franceschini G., Di Leone A., Natale M., Sanchez M.A., Masett R. Conservative surgery after neoadjuvant chemotherapy in patients with operable breast cancer. Ann. Ital. Chir. 2018; 89: 290.
- von Minckwitz G., Huang C.-S., Mano M.S., Loibl S., Mamounas E.P., Untch M. et al. Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N. Engl. J. Med. 2019; 380(7): 617-28. https://dx.doi.org/10.1056/NEJMoa1814017.
- Parmar V., Krishnamurthy A., Hawaldar R., Nadkarni M.S., Sarin R., Chinoy R. Breast conservation treatment in women with locally advanced breast cancer—experience from a single centre. Int. J. Surg. 2006; 4(2): 106-14. https://dx.doi.org/10.1016/j.ijsu.2006.01.004.
- von Minckwitz G. Preoperative therapy: what, when and for whom? Ann. Oncol. 2008l; 19(Supppl 5): v113-6. https://dx.doi.org/10.1093/annonc/mdn323.
- Hunt K.K., Yi M., Mittendorf E.A., Guerrero C., Babiera G.V., Bedrosian I., Hwang R.F. et al. Sentinel lymph node surgery after neoadjuvant chemotherapy is accurate and reduces the need for axillary dissection in breast cancer patients. Ann. Surg. 2009; 250(4): 558-66. https://dx.doi.org/10.1097/SLA.0b013e3181b8fd5e.
- Cen C., Chun J., Kaplowitz E., Axelrod D., Shapiro R., Guth A., Schnabel F. Margin assessment and re-excision rates for patients who have neoadjuvant chemotherapy and breast-conserving surgery. Ann. Surg. Oncol. 2021; 28(9): 5142-8. https://dx.doi.org/10.1245/s10434-020-09524-0.
- Fisher B., Bryant J., Wolmark N., Mamounas E., Brown A., Fisher E.R. et al. Effect of preoperative chemotherapy on the outcome of women with operable breast cancer. J. Clin. Oncol. 1998; 16(8): 2672-85. https://dx.doi.org/10.1200/JCO.1998.16.8.2672.
- Conti M., Morciano F., Bufi E., D'Angelo A., Panico C., Di Paola V. et al. Surgical planning after neoadjuvant treatment in breast cancer: a multimodality imaging-based approach focused on MRI. Cancers (Basel). 2023; 15(5): 1439. https://dx.doi.org/10.3390/cancers15051439.
- Monrigal E. Mastectomy with immediate breast reconstruction after neoadjuvant chemotherapy and radiation therapy. A new option for patients with operable invasive breast cancer. Results of a 20 years single institution study. Eur. J. Surg. Oncol. 2011; 37(10): 864-70. https://dx.doi.org/10.1016/j.ejso.2011.07.009.
- Donker M., Hage J.J., Woerdeman L.A.E., Rutgers E.J.T., Sonke G.S. Surgical complications of skin sparing mastectomy and immediate prosthetic reconstruction after neoadjuvant chemotherapy for invasive breast cancer. Eur. J. Surg. Oncol. 2012; 38(1): 25-30. https://dx.doi.org/10.1016/j.ejso.2011.09.005.
- Chi W., Zhang Q., Li L., Chen M., Xiu B., Yang B., Wu J. Immediate breast reconstruction after neoadjuvant chemotherapy: factors associated with surgical selection and complications. Ann. Plast. Surg. 2023; 91(1): 48-54. https://dx.doi.org/10.1097/SAP.0000000000003574.
- Jagsi R., Momoh A.O., Qi J., Hamill J.B., Billig J., Kim H.M. et al. Impact of radiotherapy on complications and patient-reported outcomes after breast reconstruction. J. Natl. Cancer Inst. 2018; 110(2): 157-65. https://dx.doi.org/10.1093/jnci/djx148.
- Beugels J., Bod L., van Kuijk S.M.J., Qiu S.S., Tuinder S.M.H., Heuts E.M. et al. Complications following immediate compared to delayed deep inferior epigastric artery perforator flap breast reconstructions. Breast Cancer Res. Treat. 2018; 169(2): 349-57. https://dx.doi.org/10.1007/s10549-018-4695-0.
- Ishiba T., Aruga T., Miyamoto H., Ishihara S., Nara M., Adachi M. et al. Short- and long-term outcomes of immediate breast reconstruction surgery after neoadjuvant chemotherapy. Surg. Today. 2022; 52(1): 129-36. https://dx.doi.org/10.1007/s00595-021-02316-3.
- Naoum G.E., Oladeru O.T., Niemierko A., Salama L., Winograd J., Colwell A. et al. Optimal breast reconstruction type for patients treated with neoadjuvant chemotherapy, mastectomy followed by radiation therapy. Breast Cancer Res, Treat. 2020; 183(1): 127-36. https://dx.doi.org/10.1007/s10549-020-05747-7.
- Sabitovic A., Trøstrup H., Damsgaard T.E. The impact of neoadjuvant chemotherapy on surgical outcomes following autologous and implant-based immediate breast reconstruction: a systematic review and meta-analysis. J. Plast. Reconstr. Aesthet. Surg. 2023; 87: 17-23. https://dx.doi.org/10.1016/j.bjps.2023.09.048.
- Yang J.R., Kuo W.L., Yu C.C., Chen S.C., Huang J.J. Reconstructive outcome analysis of the impact of neoadjuvant chemotherapy on immediate breast reconstruction: a retrospective cross-sectional study. BMC Cancer. 2021; 21(1): 522. https://dx.doi.org/10.1186/s12885-021-08256-y.
- Bowen M.E., Mone M.C., Buys S.S., Sheng X., Nelson E.W. Surgical outcomes for mastectomy patients receiving Neoadjuvant chemotherapy: a propensity-matched analysis. Ann. Surg. 2017; 265(3): 448-56. https://dx.doi.org/10.1097/SLA.0000000000001804.
- Beugels J., Meijvogel J.L.W., Tuinder S.M.H., Tjan-Heijnen V.C.G., Heuts E.M., Piatkowski A., van der Hulst R.R.W.J. The influence of neoadjuvant chemotherapy on complications of immediate DIEP flap breast reconstructions. Breast Cancer Res. Treat. 2019; 176(2): 367-75. https://dx.doi.org/10.1007/s10549-019-05241-9.
Received 08.11.2023
Accepted 24.01.2024
Azizjon D. Zikiryakhodzhaev, Dr. Med. Sci., Professor, Head of the Department of Oncology and Reconstructive Plastic Surgery of the Breast and Skin, P.A. Herzen Moscow Research Institute of Oncology – branch of the National Medical Research Center of Radiology, Ministry of Health of Russia, 125284, Russia, Moscow, 2nd Botkinsky passage, 3; Professor of the Department of Oncology, Radiotherapy and Plastic Surgery, Institute of Clinical Medicine, I.M. Sechenov First Moscow State Medical University,
Ministry of Health of Russia (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8-2; Professor of the Department of Oncology and X-ray Radiology named after
V.P. Kharchenko, Medical Institute, RUDN University, 117198, Russia, Moscow, Miklukho-Maklaya str., 6,
azizz@mail.ru, https://orcid.org/0000-0001-7141-2502
Alana R. Bosieva, PhD, Researcher at the Department of Oncology and Reconstructive Plastic Surgery of the Breast and Skin, P.A. Herzen Moscow Research Institute of Oncology – branch of the National Medical Research Center of Radiology, Ministry of Health of Russia, 125284, Russia, Moscow, 2nd Botkinsky passage, 3; Assistant at the Department of Oncology and X-ray Radiology named after V.P. Kharchenko, Medical Institute, RUDN University, 117198, Russia, Moscow, Miklukho-Maklaya str., 6, +7(988)875-10-47,
ms.bosieva@mail.ru, SPIN-code: 1090-7281, https://orcid.org/0000-0003-0993-8866
Corresponding author: Alana R. Bosieva,
ms.bosieva@mail.ru





