Use of vaginal ornidazole in women of reproductive age with bacterial vaginosis: results of a multicenter observational study
Apolikhina I.A., Rakhmatulina M.R., Olina A.A., Artymuk N.V., Tarnaeva L.A., Marochko T.Yu.
Objective: To evaluate the clinical and microbiological efficacy and tolerability of a vaginal ornidazole formulation (Dazolic Lady) in women of reproductive age with bacterial vaginosis within real-world clinical practice.
Materials and methods: A prospective, multicenter, uncontrolled, observational study with a before-and-after design was conducted in real-world clinical settings. The study included 100 women aged 18–45 years with a confirmed diagnosis of bacterial vaginosis. Patients were prescribed Dazolic Lady 500 mg intravaginally once daily for 6 days. Treatment efficacy was assessed 4 weeks post-treatment via clinical examination, vaginal pH measurement, Gram-stained smear microscopy, and PCR-based analysis of the vaginal microbiota. Tolerability and treatment satisfaction were evaluated using a Likert scale.
Results: Four weeks after treatment, a significant reduction in the severity of bacterial vaginosis symptoms was observed, with only 3% of patients still reporting complaints. The mean vaginal pH level decreased significantly from 5.43 (0.39) to 4.52 (0.38) (p<0.001). The detection rate of clue cells also decreased significantly from 84% to 8% post-treatment. PCR analysis showed a statistically significant reduction in the detection rates of Gardnerella vaginalis, Fannyhessea vaginae, Mobiluncus spp., and Prevotella spp., along with a concurrent increase in the proportion of Lactobacillus spp. (p<0.05). No adverse events were reported, and 98% of patients rated treatment tolerability as good or excellent.
Conclusion: Vaginal ornidazole (Dazolic Lady) demonstrated high clinical and microbiological efficacy, an absence of adverse events, and good tolerability. These findings support its use as an effective therapeutic option for bacterial vaginosis in outpatient settings.
Authors' contributions: Apolikhina I.A., Tarnaeva L.A. – conception and design of the study, statistical analysis, collection and processing of material, drafting of the manuscript, editing of the manuscript; Rakhmatulina M.R. – conception and design of the study, collection and processing of material; Olina A.A., Artymuk N.V., Marochko T.Yu. – collection and processing of material, editing of the manuscript.
Conflicts of interest: The authors have no conflicts of interest to declare.
Funding: There was no funding for this study.
Ethical approval: The study was reviewed and approved by the Research Ethics Committee of the V.I. Kulakov NMRC for OG&P, Ministry of Health of Russia.
Patient Consent for Publication: All patients provided informed consent for the publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available upon request from the corresponding author after approval from the principal investigator.
For citation: Apolikhina I.A., Rakhmatulina M.R., Olina A.A., Artymuk N.V., Tarnaeva L.A., Marochko T.Yu. Use of vaginal ornidazole in women of reproductive age with bacterial vaginosis: results of a multicenter observational study.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2026; (2): 111-118 (in Russian)
https://dx.doi.org/10.18565/aig.2025.387
Keywords
Bacterial vaginosis (BV) characterized by disruption of the vaginal microbiota is a common vaginal condition in women of reproductive age. This disruption involves a shift away from protective Lactobacillus species and toward an overgrowth of facultative and obligate anaerobic bacteria [1], including Gardnerella vaginae, Fannyhessea vaginae (formerly Atopobium vaginae), Prevotella bivia, Mobiluncus spp., Peptostreptococcus anaerobius, and Megasphaera spp. [2], among others [3, 4]. The clinical significance of BV extends beyond subjective symptoms, as it is associated with an increased risk of pelvic inflammatory disease, pregnancy complications, heightened susceptibility to sexually transmitted infections (STIs), and high recurrence rates [5–8].
The current understanding of the vaginal microbiota highlights the complex pathogenesis of BV and the need for anti-anaerobic agents that are effective against key microorganisms involved in biofilm formation and persistent microbial communities [9].
Although standard BV treatment typically involves metronidazole in various formulations, the persistently high recurrence rate necessitates the exploration of alternative therapeutic approaches [10].
Ornidazole, a nitroimidazole, has a longer elimination half-life and exhibits significant activity against anaerobic bacteria, including Gardnerella vag. and Fannyhessea vag. Studies suggest that vaginal ornidazole formulations can provide sufficient local drug exposure and may offer pharmacokinetic advantages over metronidazol. Specifically, data indicate that at pH 7, ornidazole is absorbed more slowly because of its higher lipophilicity, ensuring prolonged local activity against anaerobic microorganisms [11].
Early clinical studies have confirmed the efficacy of ornidazole in restoring vaginal flora. Lindner J.G. et al. [12] first demonstrated the impact of ornidazole on vaginal microbiocenosis, observing the restoration of normal vaginal and cervical canal microbiota after a five-day course of therapy. Despite its age, this study remains fundamental as a starting point for investigating the local effects of 5-nitroimidazoles. Based on accumulating evidence, some researchers have proposed incorporating ornidazole into alternative BV treatment regimens. For example, Vazquez F. et al. [9] presented a treatment regimen that included vaginal ornidazole (500 mg) and a combination of oral and vaginal therapy, an approach potentially justified in severe or recurrent cases. Similarly, Saraçoğlu F. et al. [13] demonstrated that combination therapy achieved high clinical cure rates [13].
Comparative studies have further supported the efficacy of ornidazole in treating BV. Thulkar J. et al. [14] reported a clinical cure rate of 100% one week after ornidazole therapy, which remained at 97.7% after four weeks. Russian data also support the activity of ornidazole: Rakhmatulina M.R. [15] showed that ornidazole was non-inferior to metronidazole in terms of efficacy and superior in terms of tolerability in some subgroups. A systematic review and meta-analysis by Fan Y. et al. [16], encompassing 42 studies and 8,382 patients, suggested that ornidazole is an effective alternative to first-line treatments.
These findings indicate a growing trend toward expanding the therapeutic options for BV.
In Russia, interest in vaginal ornidazole formulations has increased following the publication of multicenter data from an observational program evaluating "Dazolyk Lady" (vaginal ornidazole). This program, which involves four research centers (V.I. Kulakov NMRC for OG&P, D.O. Ott RI for OGR and a medical center in Kemerovo), demonstrated the drug's clinical efficacy, high tolerability, and ease of use [17], as reported by Olina A.A. et al. (2025).
Therefore, a multicenter, uncontrolled observational study to evaluate the clinical efficacy and tolerability of vaginal ornidazole in women of reproductive age with BV is warranted.
This study aimed to assess the clinical efficacy and tolerability of vaginal ornidazole (Dazolyk Lady) in women of reproductive age with bacterial vaginosis in a real-world clinical setting.
Materials and methods
This prospective, multicenter, uncontrolled observational study employed a before–after design and was conducted in real-world clinical settings.
Because the study was observational and performed in routine clinical settings without interference in prescribed therapy, a control group was not included. The primary endpoint was the assessment of the tolerability of vaginal ornidazole therapy (Dazolik Lady) after 6 days of treatment and during the 3 days after therapy completion. The secondary endpoint was the evaluation of the clinical effects of therapy 4 weeks after treatment completion. The study population comprised 100 women of reproductive age with a confirmed diagnosis of BV. The study was conducted within routine outpatient gynecological practices in three cities of the Russian Federation: Moscow (n=40), Saint Petersburg (n=30), and Kemerovo (n=30). Standardized clinical data were collected using electronic individual case report forms. The observation period for each participant was at least 5 weeks and included two visits.
The inclusion criteria were as follows: age 18–45 years; confirmed diagnosis of BV (clinically and/or based on microscopy and polymerase chain reaction [PCR]); absence of systemic or local antibacterial therapy within the preceding 3 months; outpatient status; and signed informed consent. The exclusion criteria were as follows: pregnancy, lactation, aerobic or mixed vaginitis, contraindications to ornidazole use, and inability to adhere to the prescribed treatment regimen.
At the first visit, a clinical examination, vaginal pH measurement, gram-stained smear microscopy, vaginal microbiota PCR testing (Femoflor/Florocenosis), assessment of vaginal discharge, and recording of subjective symptoms were performed. After confirming the diagnosis, the patients were prescribed Dazolik Lady at a dosage of one 500 mg vaginal tablet once daily at bedtime for six days. This prescription was made in accordance with current clinical guidelines prior to discussing the participation in the observational program. The second visit, which took place 4 weeks after therapy completion, included reevaluation of symptoms, pH measurement, repeat smear microscopy, PCR testing, and assessment of drug tolerability and treatment satisfaction. Tolerability, general well-being, ease of use, and accessibility of the medication were rated on a Likert scale by both the physician and the patient.
The tolerability of therapy (primary endpoint) was evaluated based on the presence or absence of adverse events during the 6-day treatment period and within 3 days after its completion. These included local reactions such as itching, burning, mucosal irritation, increased discharge, or allergic reactions, as well as treatment discontinuation and adherence to the prescribed dosing regimen. Additionally, tolerability, convenience of use, and subjective treatment outcomes were recorded using a Likert scale based on both physician and patient assessments.
The clinical efficacy of therapy (secondary endpoint) was assessed 4 weeks after the end of treatment by analyzing the dynamics of BV clinical symptoms, vaginal pH, and laboratory findings. The efficacy criteria included (1) presence/absence and severity of typical BV symptoms, (2) change in vaginal pH, (3) microscopic signs of bacterial vaginosis (including the presence of "clue cells"), and (4) PCR test results.
Statistical analysis
The statistical data presentation complied with the SAMPL guidelines. Continuous variables with a normal distribution are presented as the mean (standard deviation), M (SD); variables with a non-normal distribution are presented as the median (interquartile range), Me (Q1; Q3). Categorical variables are reported as counts and percentages (n (%)). The normality of the distribution was tested using the Kolmogorov–Smirnov test. The Wilcoxon test was applied to compare paired continuous variables before and after treatment. McNemar’s test was used to compare related binary proportions (before/after). Cochran's Q test was applied when harmonization across multiple categorical indicators was required. The level of statistical significance was set at p<0.05. The predetermined minimum sample size was 77 patients, and the actual study population comprised 100 participants.
Results
The study results are presented according to the prespecified primary and secondary endpoints. The primary endpoint, treatment tolerability, was assessed based on adverse events (AEs) and Likert scale data recorded during the 6-day treatment period and for three days post-treatment. The secondary endpoint, clinical efficacy, was evaluated 4 weeks after treatment completion and included an analysis of clinical symptom dynamics, vaginal pH levels, and microbiological parameters. The study enrolled 100 women of reproductive age who met the inclusion criteria and had laboratory-confirmed bacterial vaginosis (BV). The mean age of the patients was 31.69 (SD 7.75) years, and the body mass index (BMI) was 24.24 (SD 4.63) kg/m². The majority of the women had regular menstrual cycles. All participants fully completed the prescribed 6-day course of Dazolik Lady and attended the follow-up visit 4 weeks post-treatment, demonstrating high treatment adherence and minimizing factors that could affect the completeness of observations.
At baseline, all patients (100/100, 100%) presented with typical BV clinical manifestations. Among the reported complaints (n=309), the most frequent were persistent vaginal discharge with an unpleasant "fishy" odor (96/309, 31.07%), vulvar discomfort (40/309, 12.94%), dyspareunia (34/309, 11.0%), and increased discharge following unprotected sexual intercourse (24/309, 7.77%) (Figure).
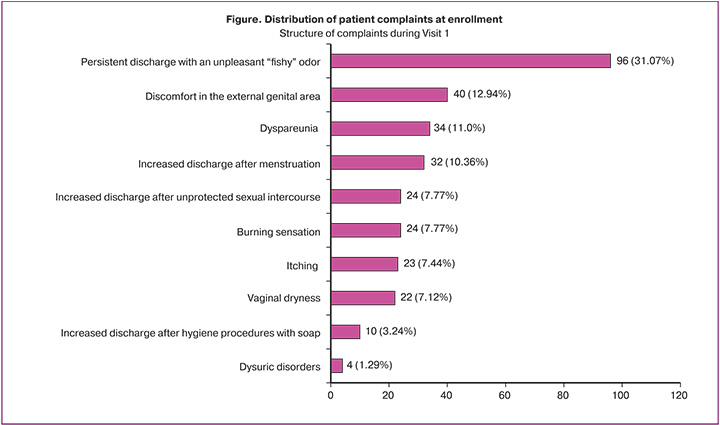
At the follow-up visit, only 3/100 (3.0%) patients reported any complaints compared to 100/100 (100%) at enrollment (p<0.001). Pathological discharge upon examination was observed in 1/100 (1.0%) patients compared to 100/100 (100.0%) at baseline (p<0.001). During the interviews, patients reported significant symptomatic improvement as early as the completion of the 6-day treatment course; however, objective symptom assessment was conducted only at Visit 2 (4 weeks post-treatment) in accordance with the study protocol. Consequently, most women presented no BV-related complaints at the follow-up examination.
Vaginal pH measurements also followed a similar trend. At baseline, vaginal acidity corresponded to values characteristic of BV (mean 5.43 (SD 0.39)). Four weeks after treatment completion, the pH decreased to 4.52 (SD 0.38), a statistically significant change from baseline (p<0.001), indicating the restoration of an acidic environment favorable for Lactobacillus growth. The proportion of women with a physiological pH ≤ 4.5 increased from 9% at baseline to 90% at the follow-up visit, reflecting the recovery of the protective properties of the vaginal ecosystem.
Gram-stained smear microscopy also revealed marked positive changes. Prior to therapy, "clue cells" characteristic of BV were detected in 84/100 (84%) women, whereas one month post-treatment, the proportion of patients positive for clue cells decreased to 8/100 (8%). An increase in Lactobacillus counts and a decrease in epithelial "granularity" were observed, indicating the normalization of the morphological profile of the biotope. The leukocyte reaction remained within the normal range at all observation stages, consistent with BV being a non-inflammatory process (Table 1).
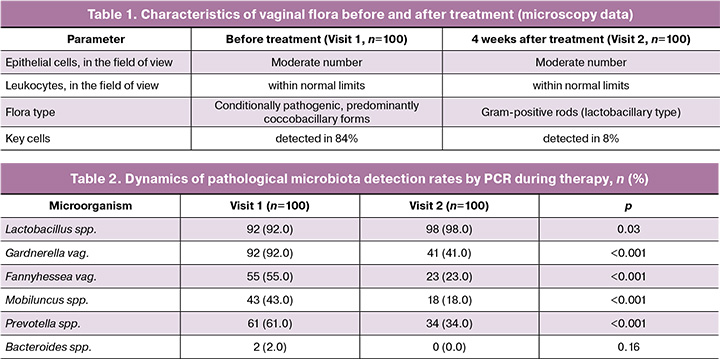
PCR performed at baseline (Visit 1) and 4 weeks post-treatment (Visit 2) revealed statistically significant changes in the vaginal microbiota structure, indicating the restoration of eubiosis and a reduction in the proportion of anaerobic consortia characteristic of BV.
Following therapy, an increase in the detection frequency of Lactobacillus spp. was noted, from 92/100 (92.0%) patients at baseline to 98/100 (98.0%) at the follow-up visit (p=0.03). Concurrently, a significant decrease in the detection frequency of BV-associated anaerobic microorganisms was recorded: Gardnerella vaginalis decreased from 92/100 (92.0%) at Visit 1 to 41/100 (41.0%) at Visit 2 (p<0.001); Fannyhessea (Atopobium) vaginae from 55/100 (55.0%) to 23/100 (23.0%) (p<0.001); Mobiluncus spp. from 43/100 (43.0%) to 18/100 (18.0%) (p<0.001); and Prevotella spp. from 61/100 (61.0%) to 34/100 (34.0%) (p<0.001) (Table 2). The detection frequency of Bacteroides spp. decreased from 2/100 (2.0%) to 0/100 (0%), although these differences were not statistically significant (p=0.16) (Table 2).
Overall, the PCR data demonstrated a pronounced shift in microbiota structure from anaerobic dysbiosis to normocenosis, consistent with the clinical outcomes, vaginal pH normalization, and microscopy findings. The significant reduction in the prevalence of Gardnerella vaginalis, Fannyhessea vaginae, Mobiluncus spp., and Prevotella spp., combined with the growth of Lactobacillus spp., confirms both the clinical and microbiological efficacy of the therapy.
The therapy was characterized by high tolerability, with no adverse events recorded during the treatment period or within 3 days post-completion (primary endpoint), as confirmed by both physician observations and patient self-assessment. On the Likert scale, 98/100 (98.0%) patients rated the drug's tolerability as "good" or "excellent", 94/100 (94.0%) noted the convenience of use, and 96/100 (96.0%) reported a marked improvement in their general well-being. Virtually all participants indicated a willingness to recommend this treatment to other women experiencing similar symptoms.
Discussion
The results of this multicenter observational study demonstrate the clinical and microbiological efficacy of a vaginal formulation of ornidazole (Dazolic Lady) in treating BV in women of reproductive age. The integration of clinical, microscopic, and molecular genetic data suggests that local ornidazole therapy effectively normalizes the vaginal environment and restores the vaginal microbiota.
These findings align with the current understanding of BV pathogenesis, which centers on an imbalance of lactobacillary flora and the overgrowth of anaerobic bacteria. Eradicating key anaerobic microbiota members, including Gardnerella vaginalis and Fannyhessea vaginae, is challenging with standard metronidazole-based regimens, contributing to persistent biofilms and disease recurrence [18]. Therefore, the search for alternative therapeutic approaches with prolonged local activity remains a clinical priority. The pharmacokinetic properties of ornidazole, including its slower absorption under elevated vaginal pH conditions and sustained therapeutic concentrations, may provide more effective activity against anaerobic communities [11].
The observed shift in vaginal microbiocenosis toward eubiosis is particularly noteworthy. This shift, characterized by reduced abundance of anaerobic microorganisms and restored lactobacillary dominance, reflects both the suppression of pathogenic bacteria and the creation of conditions conducive to stable microbiological remission. Restoring normal biocenosis is considered a key factor in reducing the risk of BV recurrence, a common clinical problem associated with systemic antibacterial regimens.
The absence of registered adverse events and the high tolerability ratings reported by both patients and physicians are consistent with previous Russian studies on 5-nitroimidazole drugs [15].
The vaginal route of administration minimizes systemic exposure and enhances therapy acceptability, which is crucial for treatment adherence in outpatient settings. This high patient adherence further validates the clinical applicability of our proposed regimen.
These results suggest that vaginal ornidazole formulations are a promising option for local BV therapy, including in patients with recurrent disease. While metronidazole and clindamycin remain first-line agents in international clinical guidelines, accumulating evidence supports the expansion of therapeutic options with drugs possessing alternative pharmacokinetic and pharmacodynamic profiles.
It is important to note that this study was conducted in a real-world clinical practice setting and lacked randomization and a control group, representing a design limitation. Consequently, the observed changes cannot be definitively attributed to therapy. However, the multicenter design, standardized outcome assessments, and objective microbiological control methods, including gram staining and PCR diagnostics, enhance the representativeness of the data across a broad clinical population. The strong correlation between clinical and microbiological outcomes underscores the potential of vaginal ornidazole as an effective treatment for primary and recurrent BV.
Conclusion
This multicenter observational study demonstrated the high clinical and microbiological efficacy of the vaginal formulation of ornidazole (Dazolic Lady) for treating BV in women of reproductive age. Four weeks after completing therapy, most patients experienced resolution of primary symptoms, normalization of vaginal pH, a significant reduction in "clue cell" detection, and a marked restoration of the vaginal microbiocenosis structure based on PCR data, including an increased proportion of normocenosis and decreased levels of BV-associated anaerobic bacteria.
The drug exhibited a favorable safety profile with no recorded adverse events during the study. Patients and physicians rated the tolerability as high, and adherence to therapy was 100%. The local administration route and ease of use contributed to high patient satisfaction with treatment.
Collectively, these data support Dazolic Lady as an effective, safe, and clinically justified agent for BV therapy, providing sustained normalization of the vaginal environment and restoration of microbiocenosis. The results confirm the value of vaginal ornidazole formulations and support their use in outpatient gynecological settings.
References
- Muzny C.A., Cerca N., Elnaggar J.H., Taylor C.M., Sobel J.D., Van Der Pol B. State of the art for diagnosis of bacterial vaginosis. J. Clin. Microbiol. 2023; 61(8): e0083722. https://dx.doi.org/10.1128/jcm.00837-22
- Рахматулаева М.М. Эффект полиморфизма гена интерлейкина-10 в формировании глубоких нарушений микробиоценоза влагалища при бактериальном вагинозе. Научные результаты биомедицинских исследований. 2024; 10(1): 156-70. [Rakhmatullaeva M.M. The effect of interleukin-10 gene polymorphism in the formation of deep disorders of vaginal microbiocenosis in bacterial vaginosis. Research Results in Biomedicine. 2024; 10(1): 156-70 (in Russian)]. https://dx.doi.org/10.18413/2658-6533-2024-10-1-0-10
- Воропаева Н.М., Белькова Н.Л., Немченко У.М., Григорова Е.В., Данусевич И.Н. Микроорганизмы, ассоциированные с бактериальным вагинозом: разнообразие и клинико-диагностическое значение. Acta biomedica scientifica. 2021; 6(3): 17-30. [Voropaeva N.M., Belkova N.L., Nemchenko U.M., Grigorova E.V., Danusevich I.N. Microorganisms associated with bacterial vaginosis: diversity and clinical and diagnostic significance. Acta biomedica scientifica. 2021; 6(3): 17-30 (in Russian)]. https://dx.doi.org/10.29413/ABS.2021-6.3.2
- Горбунова Е.А., Аполихина И.А. К вопросу о диагностике и лечении рефрактерных и рецидивирующих вагинозов и вагинитов в практике гинеколога. Медицинский совет. 2024; 18(4): 75-82. [Gorbunova Е.А., Apolikhina I.A. On the diagnosis and treatment of refractory and recurrent vaginosis and vaginitis in the practice of a gynecologist. Medical Council. 2024; 18(4): 75-82 (in Russian)]. https://dx.doi.org/10.21518/ms2024-165
- Leitich H., Kiss H. Asymptomatic bacterial vaginosis and intermediate flora as risk factors for adverse pregnancy outcome. Best Pract. Res. Clin. Obstet. Gynaecol. 2007; 21(3): 375-90. https://dx.doi.org/10.1016/j.bpobgyn.2006.12.005
- Atashili J., Poole C., Ndumbe P.M., Adimora A.A., Smith J.S. Bacterial vaginosis and HIV acquisition: a meta-analysis of published studies. AIDS. 2008; 22(12): 1493-1501. https://dx.doi.org/10.1097/QAD.0B013E3283021A37
- Redelinghuys M.J., Geldenhuys J., Jung H., Kock M.M. Bacterial vaginosis: current diagnostic avenues and future opportunities. Front. Cell. Infect. Microbiol. 2020; 10: 354. https://dx.doi.org/10.3389/FCIMB.2020.00354
- Peebles K., Velloza J., Balkus J.E., McClelland R.S., Barnabas R.V. High global burden and costs of bacterial vaginosis: a systematic review and meta-analysis. Sex. Transm. Dis. 2019; 46(5): 304-11. https://dx.doi.org/10.1097/OLQ.0000000000000972
- Vazquez F., Fernández-Blázquez A., García B. Vaginosis. Vaginal microbiota. Enferm. Infecc. Microbiol. Clin. (Engl. Ed). 2019; 37(9): 592-601. English, Spanish. https://dx.doi.org/10.1016/j.eimc.2018.11.009
- Миханошина Н.В., Припутневич Т.В., Григорян И.Э. Вагиниты и вагиноз: подходы к диагностике и лечению в свете действующих клинических рекомендаций. Акушерство и гинекология. 2022; 11: 172-8. [Mikhanoshina N.V., Priputnevich T.V., Grigorian I.E. Vaginitis and vaginosis: diagnostic and treatment approaches in the light of the current clinical practice guidelines. Obstetrics and Gynecology. 2022; (11): 172-8 (in Russian)]. https://dx.doi.org/10.18565/aig.2022.11.172-178
- Ozyazici M., Gökçe E., Hizarcioglu S.Y., Taner M.S., Köseoglu K., Ertan G. Dissolution and vaginal absorption characteristics of metronidazole and ornidazole. Pharmazie. 2006; 61(10): 855-61.
- Lindner J.G., Plantema F.H., De Vos N.M., Hoogkamp-Korstanje A.A. Reaction of the vaginal flora to ornidazol in patients with cervicitis. Chemotherapy. 1979; 25(4): 243-8. https://dx.doi.org/10.1159/000237846
- Saraçoǧlu F., Göl K., Şahin Ï., Türkkani B., Atalay C., Öztopçu C. Treatment of bacterial vaginosis with oral or vaginal ornidazole, secnidazole and metronidazole. Int. J. Gynecol. Obstet. 1998; 62(1): 59-61. https://dx.doi.org/10.1016/S0020-7292(98)00029-0
- Thulkar J., Kriplani A., Agarwal N. A comparative study of oral single dose of metronidazole, tinidazole, secnidazole and ornidazole in bacterial vaginosis. Indian. J. Pharmacol. 2012; 44(2): 243-5. https://dx.doi.org/10.4103/0253-7613.93859
- Рахматулина М.Р. Опыт применения 5-нитроимидазолов в терапии больных с бактериальным вагинозом. Российский вестник акушера-гинеколога. 2015; 15(2): 92-7. [Rakhmatulina M.R. Experience with 5-nitroimidazoles in the therapy of bacterial vaginosis. Russian Bulletin of Obstetrician-Gynecologist. 2015; 15(2): 92-7 (in Russian)]. https://dx.doi.org/10.17116/rosakush201515292-97
- Fan Y., Gu Y., Xian Y., Li Q., He Y., Chen K. et al. Efficacy and safety of different drugs for the treatment of bacterial vaginosis: a systematic review and network meta-analysis. Front. Cell. Infect. Microbiol. 2024; 14: 1402346. https://dx.doi.org/10.3389/fcimb.2024.1402346
- Олина А.А., Савичева А.М., Будиловская О.В., Метелева Т.А. Конкурентные преимущества орнидазола в лечении бактериального вагиноза. Вопросы гинекологии, акушерства и перинатологии. 2025; 24(4): 166-73. [Olina A.A., Savicheva A.M., Budilovskaya O.V., Meteleva T.A. Competitive advantages of ornidazole in the treatment of bacterial vaginosis. Gynecology, Obstetrics and Perinatology. 2025; 24(4): 166-73 (in Russian)]. https://dx.doi.org/10.20953/1726-1678-2025-4-166-173
- Swidsinski S., Moll W.M., Swidsinski A. Bacterial vaginosis – vaginal polymicrobial biofilms and dysbiosis. Dtsch. Arztebl. Int. 2023; 120(20): 347-54. https://dx.doi.org/10.3238/ARZTEBL.M2023.0090
Received 29.12.2025
Accepted 02.02.2026
About the Authors
Inna A. Apolikhina, Dr. Med. Sci., Professor, Head of the Department of Aesthetic Gynecology and Rehabilitation, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparina str., 4; Professor at the Department of Obstetrics, Gynecology, Perinatology and Reproductology of the Institute of Vocational Education, I.M. Sechenov First Moscow State Medical University, Ministry of Health of Russia (Sechenov University), apolikhina@inbox.ru, https://orcid.org/0000-0002-4581-6295Margarita R. Rakhmatulina, Dr. Med. Sci., Professor, Deputy Director for Organizational and Analytical Work, State Research Center of Dermatovenereology and Cosmetology, Ministry of Health of Russia, 107076, Russia, Moscow, Korolenko str., 3, bldg. 6, rahmatulina@cnikvi.ru, https://orcid.org/0000-0003-3039-7769
Anna A. Olina, Dr. Med. Sci., Professor, Deputy Director for Development, D.O. Ott Research Institute of Obstetrics, Gynecology and Reproductology, 199034, Russia,
St. Petersburg, Mendeleevskaya Line, 3; Professor at the Department of Obstetrics and Gynecology, Academician E.A. Wagner Perm State Medical University, Ministry of Health of Russia; Professor at the Department of Obstetrics and Gynecology named after S.N. Davydov, I.I. Mechnikov North-Western State Medical University, Ministry of Health of Russia, olina29@mail.ru, https://orcid.org/0000-0001-9101-7569
Natalia V. Artymuk, Dr. Med. Sci., Professor, Head of the Department of Obstetrics and Gynecology named after Prof. G.A. Ushakova, Kemerovo State Medical University, Ministry of Health of Russia, 650056, Russia, Kemerovo, Voroshilova str., 22a, +7(3842)73-48-56, artymuk@gmail.com, https://orcid.org/0000-0001-7014-6492
Liana A. Tarnaeva, Obstetrician-Gynecologist, PhD Student, Academician V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology and Perinatology, Ministry of Health of Russia, 117997, Russia, Moscow, Ac. Oparina str., 4, +7(915)112-77-52, li.tarnaeva@ya.ru, https://orcid.org/0009-0006-7500-7516
Tatiana Yu. Marochko, PhD, Associate Professor at the Department of Obstetrics and Gynecology named after Prof. G.A. Ushakova, Kemerovo State Medical University, Ministry of Health of Russia, 650056, Russia, Kemerovo, Voroshilova str., 22a, +7(3842)73-48-56, marochko.2006.68@mail.ru, https://orcid.org/0000-0001-5641-5246



