В настоящее время в структуре инфекционных заболеваний женской репродуктивной сферы наиболее распространены бактериальный вагиноз (БВ), вульвовагинальный кандидоз и неспецифический вагинит, этиологическим фактором которых выступают условно-патогенные микроорганизмы (УПМ) и грибы, являющиеся составной частью нормальной микробиоты.
БВ относится к наиболее частым патологическим состояниям, встречающимся в акушерско-гинекологической практике. БВ – это дисбактериоз влагалища или полимикробный невоспалительный синдром, сопровождающийся нарушением баланса индигенной микробиоты с уменьшением биомассы лактобактерий и увеличением облигатных и/или факультативных анаэробов, включая Gardnerella vaginalis, Fannyhessea vaginae (Atopobium vaginae), Prevotella spp. и др. Среди всех вульво-вагинальных инфекций БВ занимает первое место, его распространенность в общей популяции женщин колеблется в широких пределах – от 16 до 84%, а среди беременных достигает 30–45% [1].
Известны неблагоприятные последствия БВ во время беременности на любых сроках. В I триместре БВ является причиной восходящего внутриматочного инфицирования, гибели эмбриона и плода, в 5 и более раз повышает риск неразвивающейся беременности и самопроизвольных выкидышей [2–5]. Частота самопроизвольного прерывания беременности в I триместре у беременных с БВ выше по крайней мере в 2 раза, чем у беременных с эубиозом половых путей [6]. В более поздние сроки беременности (II и III триместры) БВ в 2 раза увеличивает риск преждевременных родов, в 3 раза – хориоамнионита, в 4–5 раз – эндометрита при постановке диагноза до 12 недель [6, 7]. БВ-ассоциированные бактерии способны продуцировать различные протеазы, разрушающие коллаген плодных оболочек, а также фосфолипазы, гидролизующие фосфолипиды в мембранах амниотических оболочек, что приводит к высвобождению арахидоновой кислоты и активации синтеза простагландинов. В исследованиях последних лет установлено, что в развитии преждевременных родов важную роль играют цитокины. Бактериальные эндотоксины, поступающие в амниотическую жидкость, способствуют выработке провоспалительных цитокинов: интерлейкинов 1 и 6, фактора некроза опухоли α, что ведет к образованию арахидоновой кислоты и далее к синтезу простагландинов Е2 и F2α, которые вызывают сокращения миометрия и развитие родовой деятельности [7–9].
В связи с осложненным течением беременности и родов при БВ актуальным остается вопрос его лечения и профилактики. Лечение вагинальных инфекций во время беременности представляет известные трудности в связи с возможным влиянием лекарственных средств на плод. В настоящее время для лечения вагинальных инфекций у беременных женщин нередко используют антибиотики, антисептики, некоторые комбинированные антибактериальные препараты и пробиотики. Однако перечень лекарственных средств для лечения в гестационном периоде ограничен ввиду возможных эмбриотоксических, тератогенных и других неблагоприятных воздействий ряда лекарств на состояние плода. Особенно актуален этот вопрос для I триместра беременности [6]. В ряде исследований показано, что лечение БВ во время беременности способствует не только устранению симптомов заболевания, но и предотвращает ранние потери беременности, снижает частоту преждевременных родов и в целом улучшает результаты [10].
В последние 10–15 лет появилось немало сообщений об эффективности и безопасности молочной кислоты при лечении БВ у небеременных женщин репродуктивного возраста [11–14]. Holst E. и Brandberg A. использовали интравагинальное введение лактат-геля с рН=3,8 с питательными компонентами для лактобацилл по 5 мл перед сном в течение 7 дней у 10 беременных женщин. Авторами был сделан важный вывод, что локальное лечение, которое восстанавливает нормальную кислотность во влагалище, но не имеет системного эффекта, может быть предпочтительным по сравнению с оральной антимикробной терапией, особенно при беременности [15]. Молочная кислота обеспечивает нормализацию рН влагалищной жидкости в пределах 3,5–4,5, что влечет гибель кислотонеустойчивых штаммов [12–14]. Таким образом, происходит физиологическое повышение естественной защиты влагалища от патогенных и чужеродных УПМ при практически полном отсутствии каких-либо побочных явлений, что при беременности имеет не меньшее значение, чем эффективность лекарственного средства.
Исходя из сказанного, становится очевидным необходимость разработки новых вариантов лечения БВ у беременных женщин на разных сроках.
Цель настоящего исследования заключалась в сравнительном анализе клинико-лабораторной эффективности, а также безопасности лечения первичного БВ у беременных женщин при применении лекарственного препарата, содержащего 100 мг молочной кислоты, в форме вагинальных суппозиториев, и традиционной терапии антисептиками или метронидазолом.
Материалы и методы
Настоящее открытое контролируемое проспективное когортное исследование эффективности, безопасности и переносимости молочной кислоты 100 мг в терапии первичного БВ у беременных женщин в I, II и III триместрах проведено в период с января 2019 г. по декабрь 2022 г. в амбулаторных и стационарных (дневной стационар) условиях клинических подразделений ФГБУ «НМХЦ им. Н.И. Пирогова» Минздрава России.
На этапе скрининга с учетом критериев включения/невключения была обследована и включена в исследование 141 беременная женщина с впервые диагностированным БВ.
Критерии включения
1. Одноплодная беременность, I, II или III (до 37 недель включительно) триместр для 1, 2 и 3 исследуемой групп. Срок беременности рассчитывается по первому дню последней менструации и результатам ультразвукового исследования (УЗИ) плода до 16 недель.
2. Диагноз БВ, установленный по критериям Amsel (общий балл ≥3) и шкале Nugent (общий балл >6).
3. Возраст от 18 до 40 лет.
4. Согласие воздерживаться от половых контактов в течение всего периода участия в исследовании.
5. Наличие подписанного информированного согласия на участие в исследовании.
6. Пациентки, способные понимать цели настоящего исследования и соблюдать требования Протокола.
Критерии невключения
В исследование не могли быть включены пациенты, которые удовлетворяли хотя бы одному из нижеследующих критериев:
1. указание в анамнезе на индивидуальную непереносимость и/или проявления лекарственной аллергии к препаратам, содержащим молочную кислоту, или другим компонентам суппозиториев «Фемилекс»;
2. возраст моложе 18 и старше 40 лет;
3. часто рецидивирующий БВ (более двух раз в год);
4. выявление сопутствующих инфекций, передающихся половым путем (гонорея, трихомониаз, хламидиоз, микоплазмоз) методом полимеразной цепной реакции (ПЦР) и в мазке, окрашенном по Граму (для Neisseria gonorrhоaea, Trichomonas vaginalis);
5. кандидозный кольпит, выявленный на основании обнаружения нитевидных форм или спор дрожжеподобных грибов в мазке, окрашенном по Граму;
6. наличие атипических клеток в мазке с шейки матки;
7. многоплодная беременность;
8. беременность, наступившая с применением методов ВРТ (ЭКО, ИКСИ, искусственная инсеминация, суррогатное материнство);
9. предлежание плаценты II–IV степени (по данным УЗИ);
10. патология пуповины (по данным УЗИ): оболочечное прикрепление пуповины, единственная артерия пуповины, гематома или истинный узел пуповины;
11. истмико-цервикальная недостаточность с наложением шва на шейку матки;
12. несостоятельность рубца на матке (по данным УЗИ) для пациентов группы 3 (III триместр до 33 недели включительно);
13. рвота беременных тяжелой степени с обезвоживанием (потеря веса >10% от исходного, измеренного при постановке на учет, Ht>40%, диурез <700 мл, кетонурия 3+/4+);
14. угрожающие преждевременные роды (укорочение и открытие наружного и/или внутреннего зева шейки матки при влагалищном исследовании, и/или кровянистые выделения из половых путей, и/или пролабирование плодных оболочек во влагалище, и/или подтекание околоплодных вод, подтвержденное лабораторным исследованием, и/или наличие регулярных схваток);
15. признаки внутриутробной инфекции (по данным УЗИ), и/или наличие IgM в крови беременной или 4-кратного повышения титра IgG с интервалом 3–4 недели к антигенам TORCH-комплекса;
16. острая плацентарная недостаточность или хроническая суб- и декомпенсированная фетоплацентарная недостаточность (антенатальная гипоксия плода, нарушение кровотока в системе мать-плацента-плод II–III степени и плодового кровотока по данным допплерометрии плода);
17. задержка внутриутробного развития плода, симметричный и асимметричный типы (отставание размеров тела на 2 и более недели от нормы, физиологичной для срока беременности); множественные или грубые аномалии развития плода;
18. подозрение на наличие хромосомных аномалий у плода;
19. резус-иммунизация беременной (титр антител 1:16 и более) и/или признаки гемолитической болезни плода при любом титре антител (толщина плаценты 6–8 см, многоводие, гепатомегалия, асцит у плода);
20. гестоз II половины беременности средней и тяжелой степени тяжести (сумма баллов >7 по шкале Г.М. Савельевой), преэклампсия, эклампсия, выявленная при беременности артериальная гипертензия (ДАД >100 мм рт. ст.);
21. суб- и декомпенсированный гестационный сахарный диабет (гликемия натощак >6,0 ммоль/л, постпрандиальная гликемия >8,0 ммоль/л);
22. аномалии развития половых органов у женщины;
23. беременность на фоне внутриматочного средства (ВМС);
24. подозрение на трофобластическую болезнь;
25. применение любых интравагинальных препаратов на момент включения в исследование;
26. острый инфекционный процесс любой локализации или обострение хронического процесса в течение месяца до скрининга, в т.ч. бессимптомная бактериурия (>100 000 бактерий в 1 мл минимум в 2 пробах мочи без признаков инфекции), воспалительные заболевания органов малого таза, мастит;
27. наличие заболеваний или состояний, способствующих повышению концентрации молочной кислоты в крови (тяжелые физические нагрузки, анемия тяжелой степени, онкологические заболевания, гликогенозы и др.);
28. курение во время беременности (по данным анамнеза);
29. отсутствие подписанного информированного согласия;
30. индекс массы тела (ИМТ) ≤17,5 или ≥40 кг/м2;
31. наличие в анамнезе иммунодефицитных состояний (ВИЧ-инфекция, СПИД, лечение цитостатиками, системными глюкокортикоидами и др.);
32. обнаружение в крови маркеров ВИЧ-инфекции, гепатита В и С, сифилиса на скрининге;
33. выявленные до беременности экстрагенитальные заболевания: сахарный диабет любой степени, артериальная гипертензия любой степени, заболевания почек или другая тяжелая экстрагенитальная патология, затрудняющая оценку эффективности лечения;
34. наличие в анамнезе патологии гемостаза (тромбоцитопатия, коагулопатия);
35. наличие серьезных заболеваний у новорожденного, в том числе сопровождающихся хронической гипоксией;
36. любые другие состояния и обстоятельства, затрудняющие, по мнению исследователя, участие в исследовании.
Критерии преждевременного завершения исследования (исключения из исследования):
1. несоответствие критериям включения;
2. впервые выявленные состояния и/или заболевания, описанные в критериях невключения;
3. неразвивающаяся беременность (отсутствие сердцебиения у плода), антенатальная гибель плода;
4. преждевременные роды, преждевременный разрыв плодных оболочек (до 37 недель);
5. нежелательные явления и серьезные нежелательные явления, при развитии которых, по мнению исследователя, дальнейшее участие в исследовании может навредить здоровью или благополучию пациентки либо или плода;
6. отсутствие динамического наблюдения за показателями, указанными в протоколе исследования (пропуск планового визита без уважительных причин и оповещения врача-исследователя, пропуск более одного планового отбора проб крови и мочи, или одного микроскопического исследования вагинальной флоры, или отбора материала для ПЦР-диагностики, или ультразвукового исследования матки и плода);
7. прием неразрешенных в данном исследовании препаратов;
8. административные причины, в том числе несоблюдение пациентом процедур, предусмотренных протоколом, способные повлиять на результаты исследования; планируемый во время беременности переезд или выбор иного места родов, которые сделают невозможным сбор информации об исходах беременности;
9. желание пациентки прекратить участие в исследовании.
Все пациентки по вариантам получаемой терапии были распределены в 2 группы. 1-ю (основную) группу составили 82 беременные, которым лечение БВ проводилось монотерапией суппозиториями с молочной кислотой 100 мг. Во 2-ю группу (сравнения) вошли 59 женщин, лечение которым проводилось антисептиками или метронидазолом. В 1-й группе из наблюдения по различным причинам выбыли 12 человек, во второй – 9. Полностью закончили исследование 70 женщин 1-й группы, которые были обследованы в различные сроки беременности: 24 пациентки в I триместре, 28 – во II триместре, 18 – в III триместре. Аналогичным образом были обследованы женщины 2-й группы (n=50): 19 пациенток в I триместре, 19 – во II триместре, 12 – в III триместре. С учетом отсутствия статистически значимых различий в каждой из групп по всем триместрам беременности, дальнейшее сравнение эффективности и безопасности лечения проводилось между группами вне зависимости от триместра.
Диагноз БВ выставлялся по критериям Амселя: визуально у всех пациенток имелись обильные патологические выделения из влагалища. рН-метрия вагинальной жидкости выполнялась с применением отечественных лакмусовых полосок «Кольпо-тест рН» (Биосенсор АН, Россия); микроскопия мазков вагинальной жидкости проводилась стандартно после окраски по Граму с оценкой наличия ключевых клеток и морфотипов микроорганизмов; аминотест выполняли с помощью 10% раствора гидроокиси калия, положительным результатом которого считали появление или усиление неприятного запаха типа «гнилой рыбы». Дополнительно в 1-й группе при исследовании использовали метод ПЦР в реальном времени (Фемофлор-16, ДНК-Технология, Россия). Всем включенным в исследование пациенткам 1-й группы назначали вагинально официнальный препарат в форме вагинальных суппозиториев, содержащий 100 мг молочной кислоты, по 1 суппозиторию 1 раз в сутки в течение 10 дней. Во 2-й группе 27 беременным был назначен деквалиния хлорид 10 мг по 1 вагинальной таблетке в течение 6 дней; 14 – хлоргексидин 16 мг по 1 суппозиторию интравагинально 2 раза в сутки в течение 7 дней; 9 женщин принимали метронидазол 500 мг по 1 вагинальному суппозиторию 1 раз в день в течение 7 дней.
Для всех включенных в исследование беременных женщин запланировано 4 обязательных визита: 0-й – скрининговый, 1-й – день назначения препарата и начала лечения, 2-й – через 1–2 дня после окончания лечения и 3-й – через 30 дней после окончания лечения. На 0, 2 и 3-м визитах выполнялись все клинические и дополнительные методы обследования. Образцы вагинальной жидкости для ПЦР-диагностики в реальном времени у пациенток 1-й группы отбирали на скрининговом (для оценки состояния микробиоты влагалища до лечения) и 3-м (для оценки микробиологической эффективности лечения БВ) визитах. Длительность участия пациенток в исследовании составила весь период до окончания беременности. Период наблюдения одной пациентки, в зависимости от срока беременности составлял от 32 до 180 дней.
Статистический анализ
На этапе первичной статистической обработки данных для контроля качества исходной информации применен однофакторный дисперсионный анализ, который позволил выделить показатели, статистически значимо отличающиеся в группах (р<0,05) по F-критерию Фишера. Кроме того, данный метод помог определить величину влия¬ния конкретного фактора на каждый из изучаемых показателей. Полученные цифровые материалы обрабатывались статистически с помощью адаптированных под поставленные задачи пакетов программы Statistica 14.0 и прикладных статистических программ BMDP (Biological Medical Program), Statgraphics, ориентированных на анализ биомедицинских данных. Применялись непараметрические методы. Рассчитывались средние, относительные величины. Для сравнения величин использовались тесты Краскела–Уоллиса, хи-квадрат, двухсторонний точный Фишера, Манна–Уитни, Вилкоксона, метод расчета МакНеймера. Для обозначения центрального положения и абсолютного разброса данных использовались среднее значение (M) и стандартное отклонение (SD) в формате M±SD, а для оценки относительного разброса применялся коэффициент вариации V, который описывает однородность показателя и позволяет сопоставлять однородность разных переменных, независимо от их масштаба и единиц измерения. Принято считать, что, если уровень вариации меньше 10%, то степень разброса данных незначительная, от 10 до 20% – средняя, больше 20% и меньше или равно 33% – значительная; если значение коэффициента вариации не превышает 33%, совокупность считается однородной, если больше 33% – неоднородной. Для исследования структуры данных по каждому показателю применялись медиана и квартили в формате Me [Q1; Q3] и минимум и максимум для оценки диапазона разброса значений показателя в формате (Min; Max). Все группы были не связаны между собой. Множественных сравнений не проводилось. Критическим значением было р=0,05.
Результаты
Обе группы были стратифицированы между собой на начальном этапе (0-й визит) по основным параметрам (возраст, рост, вес и др., Mе [Q1; Q3]), а также по всем признакам БВ: выделения, их запах, зуд, боль, жжение, гиперемия, микроскопические показатели, рН, аминотест и др. (табл. 1, 2). Как видно из представленных данных, характеристики пациенток исследуемых групп существенно не отличались, то есть изначально были выбраны равнозначные группы. Общая характеристика состояния и анамнеза беременных женщин с БВ свидетельствует о репрезентативности сравниваемых, в первую очередь, по триместрам групп, что обеспечивает высокий уровень статистического анализа и сильную достоверность полученных результатов.
При изучении кислотности (рН-метрия) влагалища установлено, что до начала лечения высокий уровень рН >4,7 выявлен у 65/70 (92,86%) женщин 1-й группы и 47/50 (94%) 2-й группы (р=0,8). В дальнейшем, сразу после лечения (2-й визит) рН>4,7 выявлен только у 2/70 (2,86%) беременных 1-й группы и 14/50 (28%) – 2-й группы (р<0,0001). Через 30 дней после окончания лечения (3-й визит) нормальная кислотность влагалища сохранялась у 69/70 (98,6%) беременных 1-й группы, тогда как во 2-й группе только у 22/50 (44%) (р<0,0001). Эти результаты свидетельствуют, что молочная кислота эффективно восстанавливает кислотность влагалища при лечении БВ у беременных женщин.
До лечения ключевые клетки были обнаружены в 1-й группе у 67/70 (95,7%) беременных, во 2-й – у 44/50 (88,0%) (р=0,1137). Сразу после лечения и через 1 месяц после его окончания ключевые клетки не были обнаружены ни в одном наблюдении пациенток в основной группе. В группе сравнения ключевые клетки обнаружены в 11/50 (22%) и 28/50 (56%) случаях соответственно (р<0,0001). Таким образом, по показателю «ключевые клетки» отмечена значимо более высокая эффективность лечения БВ у беременных при применении молочной кислоты (табл. 3).
Аналогичная положительная динамика отмечена и в отношении аминотеста.
В таблице 3 приведены данные по основным симптомам БВ и клиническим признакам в динамике после лечения (на 2-м и 3-м визитах, по сравнению с 1-м визитом). Как видно из представленных данных, результаты лечения молочной кислотой оказались значимо более эффективными, чем в группе сравнения, практически по всем показателям.
У беременных 1-й группы оценивали микробиологическую эффективность по результатам количественной ПЦР в реальном времени дважды: до лечения и через 30 дней после окончания лечения.
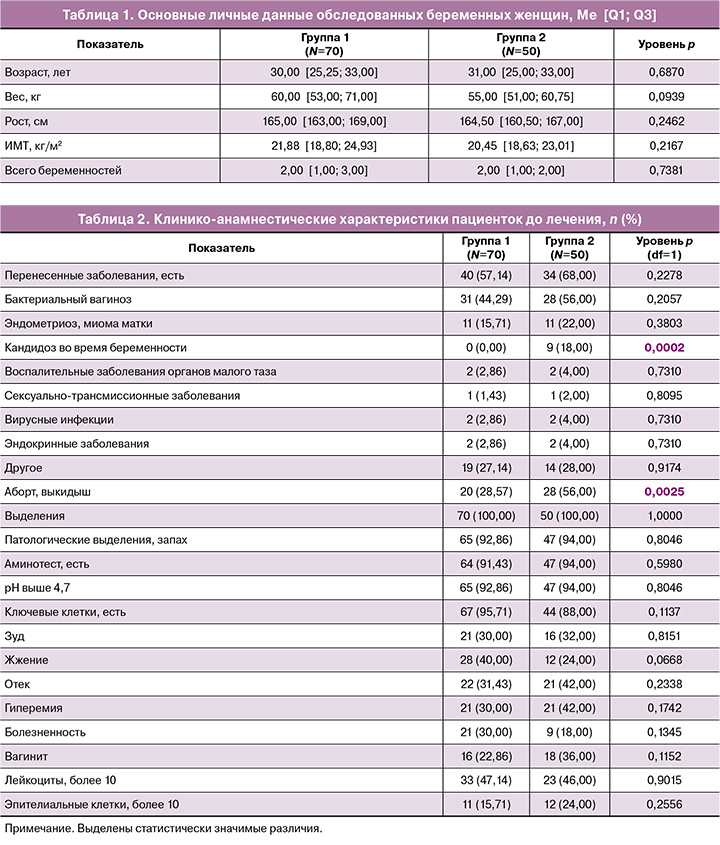
Анализ полученных данных показал, что для всех беременных с I по III триместр было характерным уменьшение количества лактобактерий до lg 105,21±2,02 в I триместре, lg 104,75±2,43 – во II и lg 104,89±2,32 – в III. На этом фоне наблюдалось увеличение количественных показателей группы Gardnerella vaginalis/Prevotella bivia/Porphyromonas spp. – до lg 105,54±1,47, lg 105,93±1,51, lg 106,28±0,83 в I, II и III триместрах соответственно. Аналогично увеличилась концентрация Eubacterium spp. – до lg 105,12±2,13, lg 104,67±2,39 и lg 104,89±2,22 соответственно I, II и III триместрам беременности. Эти данные свидетельствуют об уменьшении числа лактобактерий у беременных женщин с БВ и увеличении у них количественных показателей БВ-ассоциированных микроорганизмов – гарднерелл и эубактерий. Другие бактерии, характерные для БВ, также присутствовали во влагалище у беременных женщин, однако их величины не превышали клинически значимых уровней и были <104. Наименьшее количество лактобактерий и наибольшее количество БВ-ассоциированных микроорганизмов наблюдалось в III триместре беременности, что может быть косвенным признаком высокого риска преждевременных родов.
Через 1 месяц после лечения молочной кислотой на фоне некоторого уменьшения общей бактериальной массы (влияние молочной кислоты на анаэробную микробиоту) по сравнению с предыдущим визитом увеличилась медиана количества лактобактерий. Наблюдалось значимое увеличение количества лактобактерий по триместрам беременности: в I – с 105,21±2,02 до 106,58±0,72 (р=0,0051); во II – с 104,75±2,43 до 106,50±0,79 (р=0,0051) и в III – с 104,89±2,32 до 106,67±0,69. Прирост составил 26,4, 36,84 и 36,36% соответственно I, II и III триместрам (р=0,0051). Суммарно динамика количества лактобактерий и БВ-ассоциированных микроорганизмов представлена на рисунке 1. Контроль взятия материала для ПЦР во всех случаях был адекватным, что свидетельствует о достоверности полученных результатов.
При анализе течения наблюдаемых беременностей и исходов родов было установлено, что после лечения молочной кислотой ни у одной пациентки не было рецидивов БВ в отдаленном периоде. Иными словами, после лечения БВ в I триместре в дальнейшем вплоть до родов рецидивы БВ не возникали. Аналогичная ситуация наблюдалась среди беременных женщин, получивших лечение во II и III триместрах.
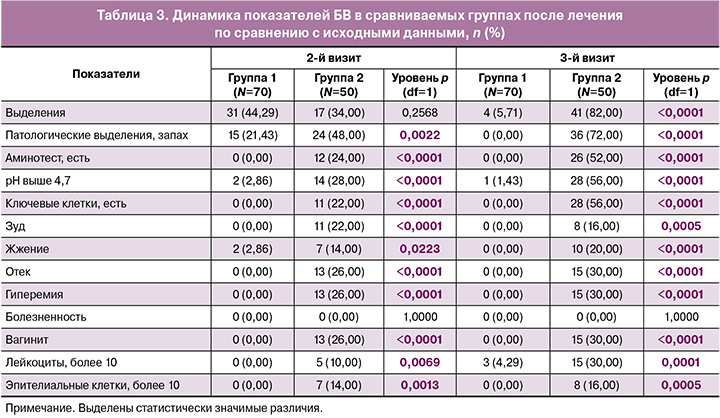
Кроме того, при анализе течения беременностей и исходов родов отмечены достоверно отличающиеся результаты, которые представлены в таблице 4 и на рисунке 2.
Так, во 2-й группе в 20% случаев наблюдались рецидивы БВ, потребовавшие повторного лечения; значимо чаще наблюдалось прерывание беременности (12%) и преждевременные роды (8%).
Кроме того, ни в одном наблюдении в 1-й группе не было самопроизвольного прерывания беременности на ранних сроках или преждевременных родов в более позднем периоде гестации. У 52 (74,3%) женщин основной группы состоялись своевременные роды естественным путем, кесарево сечение (КС) выполнено у 18 (25,7%) пациенток. В группе сравнения у 33 (75%) женщин прошли своевременные роды естественным путем, КС выполнено у 11 (25%) пациенток.
Мы проанализировали полученные данные и провели корреляционный анализ, в результате которого было установлено, что из всей выборки риск КС составил 25,4%. Наибольший риск КС наблюдался у пациенток с ИМТ >22,9 кг/м2; обнаруживаемыми во влагалище высокими титрами гарднерелл и эубактерий; малым количеством лактобактерий.
Обсуждение
В настоящее время в арсенале акушеров-гинекологов имеется ограниченный перечень лекарственных средств, имеющих доказанную эффективность и безопасность применения при беременности, особенно на ранних ее сроках. Данная проблема актуальна и для лечения БВ [16]. С этих позиций изучение эффективности и безопасности альтернативных антибиотикам схем лечения с применением более физиологичных средств у беременных женщин является весьма перспективным. Теоретическое обоснование применения молочной кислоты как наиболее физиологического компонента в качестве самостоятельного средства или в комплексном лечении БВ нашло свое отражение в проведенном исследовании.
Появившиеся в последнее десятилетие публикации о роли молочной кислоты в поддержании локального биологического гомеостаза влагалища, отсутствии ее влияния на состав нормальной микробиоты, локальный иммунологический статус и др., подтверждают важность дальнейших исследований [11–16]. Первое сообщение об использовании молочной кислоты для лечения вагинальных инфекций, в том числе и у беременных женщин, принадлежит, вероятно, Альберту Додерлейну (1892). Об этом красноречиво уже на следующий год (1893) написал в своей диссертации русский акушер-гинеколог В.В. Строганов: «Исходя из приведенных наблюдений, что кислая реакция благоприятствует развитию нормальной флоры в вагине, он [А. Додерлейн] применил молочную кислоту, в виде спринцеваний 1% раствором, при лечении беременных с патологическим секретом. Результат оказался лучшим, чем при всех других методах лечения. Работа Додерлейна чрезвычайно интересна и поучительна» [17]. Впоследствии этот способ на многие годы был забыт, а вместо молочной кислоты стали широко использовать антифизиологичные содовые спринцевания, что не могло не сказаться на ухудшении женского здоровья в целом. Только через столетие, но уже не спринцевания, а инстилляции 1–2% раствором молочной кислоты во влагалище получили применение для лечения БВ у беременных женщин [1]. Было констатировано, что инстилляции влагалища молочной кислотой являются оптимальным и физиологически оправданным способом в комплексном лечении БВ. Благодаря их назначению уменьшается рН содержимого влагалища (восстанавливается кислая среда), создавая тем самым неблагоприятные условия для размножения строгих анаэробов, ассоциированных с БВ, и напротив, оптимизируя влагалищную жидкость и среду для роста лактобактерий. Кроме того, слабые растворы молочной кислоты обладают выраженным антисептическим действием, а также обеспечивают оптимальные условия для восстановления лактобациллярной нормобиоты. С этой позиции представляется перспективным и патогенетически обоснованным применение местных антисептических средств, снижающих рН вагинальной жидкости. Основным эффектом кислотосодержащих препаратов является непосредственное снижение рН среды влагалища. В результате окисления вагинальной жидкости происходит выраженное подавление роста анаэробных бактерий и в то же время восстановление нормальной лактобациллярной микробиоты, для которой кислая среда является оптимальной. В настоящее время применение кислотосодержащих препаратов является, по мнению многих отечественных и зарубежных авторов, новой парадигмой в лечении БВ, представляющей реальную альтернативу стандартным схемам системной и местной антибиотикотерапии.
Andersch В. et al. [18] для лечения БВ предложили местное применение лактат-геля (рН=3,5) по 5 мл в течение 7 дней. По мнению исследователей, этот метод обладал почти такой же эффективностью, как прием метронидазола 500 мг 2 раза в день в течение недели. В 2015 г. нами также была опубликована статья, посвященная применению молочной кислоты в сочетании с орнидазолом при лечении БВ [20], в которой была показана эффективность данной комбинации.
В настоящем исследовании доказана высокая результативность монотерапии БВ у беременных женщин суппозиториями со 100 мг молочной кислоты, эффективность и безопасность которой в настоящее время подтверждена в нескольких рандомизированных клинических исследованиях, выполненных с соблюдением международных стандартов и обладающих высокой доказательностью. Также изучено in vitro влияние молочной кислоты на некоторые УПМ, грибы Candida albicans, протективные лактобактерии и образование биопленок [21]. Эти данные способны повлиять в перспективе на выбор молочной кислоты для лечения БВ и других вагинальных инфекций у беременных женщин. Полученные результаты расширяют акушерам-гинекологам возможности в выборе терапии БВ в I, II и III триместрах беременности.
В соответствии с государственным реестром лекарственных средств в Российской Федерации зарегистрирован лекарственный препарат в форме суппозиториев вагинальных, содержащий 100 мг молочной кислоты, под торговым наименованием «Лактодепантол» (ранее «Фемилекс»). Его основное действие направлено на снижение рН влагалища до физиологических значений (3,5–4,5), восстановление собственной микробиоты, образование защитных и разрушение патогенных биопленок. Применяется по 1 суппозиторию 1 раз в сутки, курс лечения 10 дней [21, 22].
Заключение
Молочная кислота (100 мг) в виде вагинальных суппозиториев является эффективным и безопасным средством лечения БВ во время беременности, включая лечение беременных женщин в I триместре. Применение молочной кислоты (100 мг) эффективно снижает количество БВ-ассоциированных микроорганизмов, таких как гарднереллы и эубактерии, на 93–100%. В то же время отмечается рост лактобактерий на 26–37%. Высокая клиническая и микробиологическая эффективность молочной кислоты, а также отсутствие побочных реакций, хорошая переносимость, комплаентность и безопасность позволяют рекомендовать ее в качестве средства первой линии и реальной альтернативы антибиотикам, антисептикам и пробиотикам при лечении БВ во время беременности. Результаты данного исследования показали высокую эффективность, безопасность и хорошую переносимость такого лечения у беременных женщин, что делает суппозитории с молочной кислотой препаратом выбора для лечения БВ во время беременности.



