Contemporary approaches to the choice of optimal delivery strategies for patients with placenta accreta spectrum
Ignatko I.V., Lebedev V.A., Bogomazova I.M., Timokhina E.V., Belousova V.S., Pashkov V.M., Fedyunina I.A., Sarakhova D.Kh., Samoylova Yu.A., Samara A.B., Askerova S.F., Gutsu V.V.
Objective: To explore the features of delivery in pregnant women with placenta accreta spectrum (PAS) based on clinical and anamnestic data and the course of gestation.
Materials and methods: The study included 107 patients with singleton pregnancies. Among them, 57 women with PAS comprised the main group, and 50 women without placental abnormalities were included in the comparison group. We analyzed the age of patients, somatic, obstetric and gynecological history, the course of current pregnancy, timing and types of delivery, as well as the volume of blood loss using different uterine devascularization techniques.
Results: Analysis of the obstetric and gynecological history showed that patients with PAS were significantly more likely to have two or more uterine scars after cesarean section. Pregnancy-related complications were significantly more often associated with the risk of preterm birth. In patients with PAS, no cases of preeclampsia, placental insufficiency, or fetal growth restriction were observed. Vaginal bleeding in patients with PAS occurred at 22–36.6 weeks of pregnancy. Blood volume loss was higher during emergency delivery in patients with PAS versus planned delivery, regardless of the method used for uterine devascularization. However, the differences were not statistically significant.
Conclusion: Given the absence of statistically significant differences in the volume of blood loss, as well as similar risks of bleeding at different stages of the second half of gestation, delivery of patients with PAS in the absence of episodes of vaginal bleeding should be considered optimal near to full-term pregnancy, in the context of preventing perinatal complications associated with prematurity.
Authors' contributions: Ignatko I.V., Lebedev V.A., Pashkov V.M., Sarakhova D.Kh. – study concept and design; Samara A.B., Samoylova Yu.A., Fedyunina I.A., Askerova S.F. – material collection and processing; Samara A.B., Bogomazova I.M., Gutsu V.V. – statistical data processing; Samara A.B., Bogomazova I.M. – manuscript writing; Ignatko I.V., Timokhina E.V., Belousova V.S., Lebedev V.A. – manuscript editing.
Conflicts of interest: The authors confirm that they have no conflict of interest to declare.
Funding: The study was conducted without any sponsorship.
Ethical Approval: The study was approved by the local Ethics Committee of I.M. Sechenov First Moscow State Medical University, Ministry of Health of Russia (Sechenov University).
Patient Consent for Publication: The patients have signed informed consent for publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available on request from the corresponding author after approval from the principal investigator.
For citation: Ignatko I.V., Lebedev V.A., Bogomazova I.M., Timokhina E.V., Belousova V.S., Pashkov V.M., Fedyunina I.A., Sarakhova D.Kh., Samoylova Yu.A., Samara A.B., Askerova S.F., Gutsu V.V.
Contemporary approaches to the choice of optimal delivery strategies for patients with placenta accreta spectrum.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2026; (3): 75-84 (in Russian)
https://dx.doi.org/10.18565/aig.2025.324
Keywords
Placenta accreta spectrum (PAS) is defined as abnormal trophoblast invasion of a part or all of the placenta into the myometrium of the uterine wall. There is a steady increase in the incidence of placenta accreta in the last decades. In 1970 the incidence of this condition was 1 in 4,000 deliveries. Nowadays the incidence of placenta accreta is 1 in 533, that can be mainly due to the increasing number of cesarean deliveries [1]. The modern theory of the pathogenesis of placenta accreta is based on impaired decidualization of the endometrium due to its atrophy associated with invasive intrauterine manipulations and/or inflammatory processes [2]. It has been found that there is a relationship between the risk of PAS and the number of previous cesarean sections. Also, the probability of the incidence of placenta accreta increases by 35–65 times in the presence of placenta previa. Other risk factors for PAS are maternal age, in vitro fertilization (IVF), a history of low-lying placenta and tight placental attachment. Among the reasons for abnormal placental attachment are clinical risk factors including a history of two or more cesarean sections, placenta previa, intrauterine interventions, bleeding in the first or second trimester and other risk factors for placenta accreta, IVF, as well as the echographic findings of risk factors – abnormal placental shapes, uterine abnormalities, abnormal myometrial vascularity, scar pregnancy after a prior C-section [3]. It should be borne in mind that instrumental research methods – ultrasound (US) and magnetic resonance imaging (MRI) do not provide 100% sensitivity and specificity [1]. According to clinical guidelines “Abnormal placental attachments (placenta previa and placenta accreta)” (2023), approved by the Ministry of Health of Russia, the recommended timing of elective delivery is at 34–36 weeks of gestation in women with placenta accreta without previous vaginal bleeding [1]. Delivery after 36 weeks of gestation is considered dangerous due to a high risk of bleeding. On the one part, the above timing of delivery is associated with perinatal complications due to preterm birth. On the other part, it is believed that total blood loss during emergency delivery (in case of bleeding) is significantly higher versus blood loss during elective delivery. So, comparative analysis of total blood loss during elective and emergency delivery was performed by Listijono D.R. et al. (2017). They reported lower blood loss in patients, who underwent elective cesarean section – 2200 ml versus 3100 ml, p<0.05) [4]. Also, comparative analysis of blood loss in 255 patients was performed by Thang N.M. et al. (2021). Among them, emergency delivery was in 95 women, and elective delivery was in 160. There were less cases of vaginal bleeding in the group of patients who had elective delivery (29/160 versus 36/95 cases, p<0.001). The patients received similar number of blood transfusions in both groups. However, the number of red blood cell units transfused was higher in the group of patients who had emergency delivery (5.3±0.33 versus 4.5±0.25) [5]. A retrospective multicenter observational study was conducted by Salmanian B. et al. (2022). It included 744 patients with PAS. Of them, 488 (66%) women gave birth at 34–36 weeks, as was indicated, and the remaining women gave birth before 34 or after 36 weeks. Based on the obtained results, it was found that maternal outcomes were similar regardless of the grade of placenta accreta spectrum both in women who gave birth at 34–36 weeks of gestation, and in women who gave birth 2 weeks earlier or later [6]. A multicenter pilot study was conducted by Sukhikh G.T. et al. (2024) in different regions of Russia, and aimed for searching the possibilities to reduce blood loss during delivery in patients with placenta accreta. Based on the results of the study, three main methods to reduce uterine blood flow were presented: complex compression hemostasis, balloon occlusion of internal or common liliac arteries, as well as the aorta, and vascular ligation, including the insertion of uterine balloon tamponade and uterine compression sutures. However, the authors suggest that further prospective study is needed to assess the factors influencing the development of early and late complications, as well as to determine the possibility of subsequent realization of reproductive function [7]. Kulikov I.A. et al. (2022) proposed to use tourniquet hemostasis in combination with vaginal balloon tamponade as the optimal method to reduce uterine blood flow. Significant reduction of blood loss was observed using this method, and subsequent blood transfusion was required compared to the use of isolated ligation of the internal iliac or uterine arteries. [8, 9]. Since placenta accreta poses a serious threat to women's health, Prikhodko A.M. et al. (2024) are of the opinion that effective obstetric management of patients from outpatient care to postpartum care, can significantly reduce the risk of complications and improve reproductive outcomes [10].
The purpose of our study was the following:
- to identify risk group for placenta accreta based on the clinical and anamnestic data and the course of current pregnancy as additional diagnostic criteria (since instrumental research methods (US and MRI) do not provide 100% sensitivity and specificity;
- to determine the optimal timing of delivery in women based on comparison of the number of elective and emergency surgical interventions at different stages of pregnancy and the volume of intraoperative blood loss;
- to determine the most effective methods for reducing uterine blood flow based on comparison of the volume of blood loss during delivery in patients with placenta accreta, and application of various techniques of uterine devascularization.
Material and methods
A single-center retrospective case-control study was conducted. It included 107 patients with singleton pregnancies. The main group comprised 57 patients with the diagnosed placenta accreta. The comparison group included 50 women without abnormal placental attachment. However, all women in the groups had one of the leading risk factors for placental accreta – uterine scar after previous cesarean section. All patients gave birth in S.S. Yudin City Clinical Hospital of Moscow Healthcare Department from 2018 to 2024. Inclusion criteria in the main group was partial or complete placenta accreta regardless of its localization, that was confirmed both intraoperatively and by the results of histological examination. Inclusion criteria in the comparison group were absence of placenta accreta regardless of its localization, and uterine scar after one or several C-sections. Non-inclusion criteria in the study for the patients in both groups were multiple pregnancies.
The patients underwent examination and treatment in accordance with clinical recommendations approved by the Ministry of Health of Russia “Postoperative scar on the uterus, requiring medical care for the mother during pregnancy, childbirth and the postpartum period” (2024) and “Abnormal placental attachments (placenta previa and placenta accreta) (2023).
Analysis of outpatient and inpatient records included patients’ age, somatic, obstetric and gynecological history, the course of current pregnancy, the timing of delivery, types of delivery and indications for delivery, as well as the volume of blood loss using different methods of blood flow reduction.
Exploration of the clinical outcomes included the following:
- identification of clinical and anamnestic factors indicating or rejecting the probability of placenta accreta;
- determination of the optimal timing of delivery in patients with placenta accreta based on defining at which the stage of pregnancy vaginal bleeding occurs mediating the necessity for emergency operative delivery, and possibly massive blood loss;
- determination of the optimal methods for reducing uterine blood flow based on comparison of the volume of blood loss using different techniques for uterine devascularization.
Statistical analysis
Statistical analysis of the obtained data was performed using Microsoft Excel 2010 with the Statresearch add-inn and SPSS Statistics. The quantitative data were tested for the normality of distribution using the Shapiro–Wilk test. The quantitative data with normal distribution (patients’ age) were described as arithmetic mean (M) and standard deviation (SD). In the absence of normal distribution, the quantitative data were described as median (Me) and the lower and upper quartiles (Q1; Q3). The quantitative variables with normal distribution in the groups with the equal variance assumption were compared using the Student's t-test. The Mann–Whitney U-test was used to compare the quantitative variables with non-normal distribution. The differences were considered significant at p<0.05. The categorical data are presented as absolute values and the percentage. Pearson’s chi-square (χ2) test was used to compare the categorical variables in two independent groups. Fisher's exact test was used when the expected frequencies in the contingency tables were less than 5. Effect size for binary outcomes was calculated as odds ratio (OR) (Haldane–Anscombe correction was used for cases with a zero value). Effect size for continuous data is presented as the difference in medians with 95% confidence interval (CI). When the lower and upper limits of 95% CI were higher or lower than one (1), we concluded that the identified association between the factor and the outcome was statistically significant at p<0.05.
Results
Analysis of patients’ age found no statistically significant differences in age between the groups. The median age of women was 36 (4.17) years (age range 23– 46 years) in the main group, and 35 (4.83) years (age range 26–46 years) in the comparison group. However, it should be noted that the age of the majority of patients in the main group – 40/57 (70.2%) and in the comparison group – 32/50 (64%) was over 35 years. Analysis of parity of pregnancies and births showed that repeated pregnancies were in the vast majority of patients in the main group – 56/57 (98.2%) and multiparity was in 55/57 (96.5%) women , whereas in the comparison group, repeated pregnancies and multiparity were in all 50/50 (100%) patients.
Analysis of somatic history (Table 1) found that lipid metabolism disorders were most often diagnosed in patients in both groups. The frequency of chronic arterial hypertension ranked second; varicose veins of the lower extremities and myopia ranked third; and chronic pyelonephritis and gastritis ranked fourth (in the comparison group). Other diseases were diagnosed in less than 10% of observations. It is noteworthy that the women in the comparison group had chronic hypertension two times more often, and lipid metabolism disorders 1.6 times more often compared with the main group. However, these differences were not statistically significant.
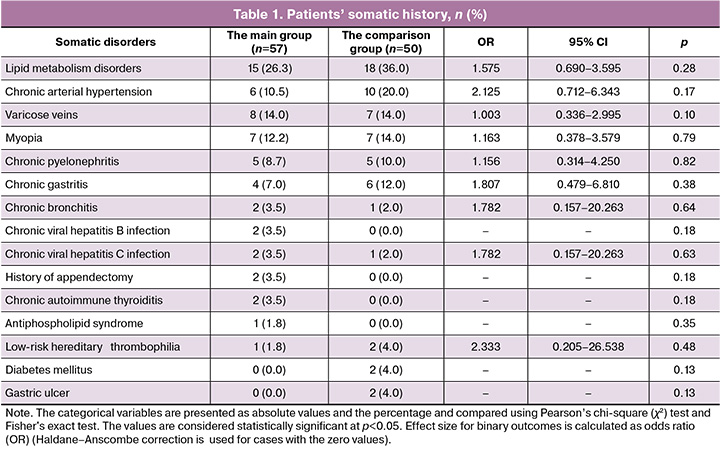
Analysis of the obstetric and gynecological history (Table 2) found that all patients in the comparison group (50/50 (100%)) and 51/57 (89.5%) patients in the main group had uterine scar after cesarean section, which is one of the leading risk factors for placenta accreta. However, the number of patients with a history of two or more C-sections was significantly higher in the main group compared with the comparison group – 27/57 (47.4%) versus 12/50 (24%), OR=2.850; 95% CI (1.241–6.546). Also it is noteworthy that the rate of spontaneous miscarriages and intrauterine interventions in history was higher in patients in the main group. However, the differences were not statistically significant. Uterine abnormalities—septate and bicornuate uterus were observed only in the main group.
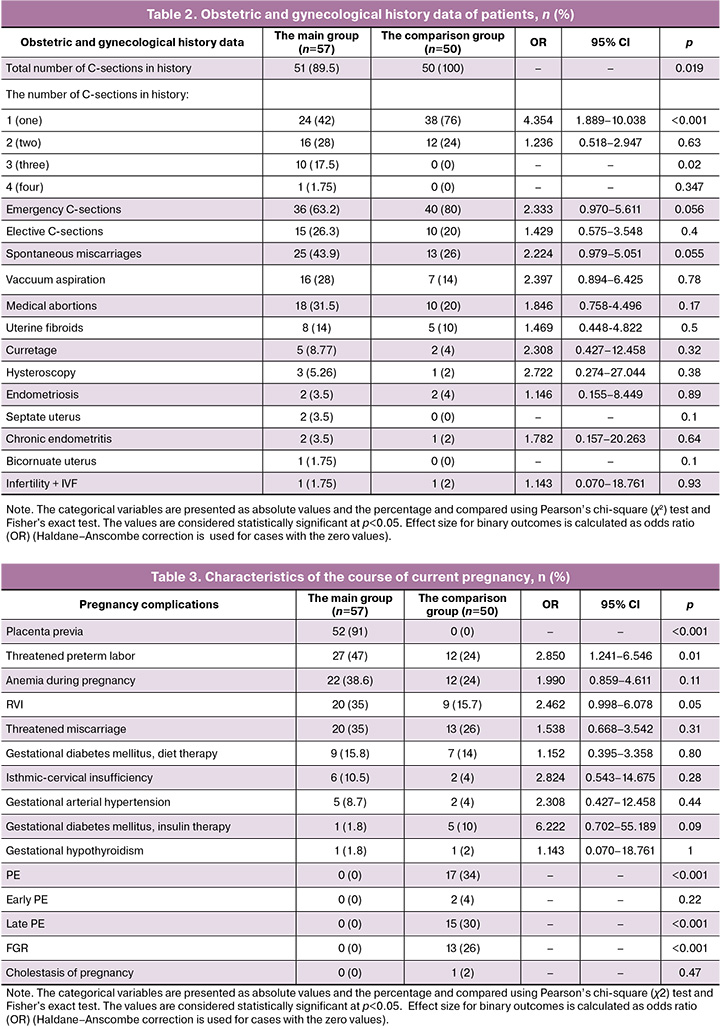
Comparison of the course of current pregnancies (Table 3) showed that threatened preterm labor was significantly more often in patients in the main group. The prevalence of anemia, acute respiratory viral infections (RVI) during pregnancy, and threatened miscarriage was insignificant in the main group.
Placenta previa was identified only in patients in the main group, whereas preeclampsia (PE) and fetal growth restriction were identified only in patients in the comparison group. It is interesting to note that none of the patients in the main group had a serious complication of pregnancy, such as preeclampsia.
Cesarean deliveries were performed in 52/57 (91%) patients in the main group and in 49/50 49/50 (98%) patients in the comparison group. Placenta previa was an indication for cesarean delivery in patients in the main group. Uterine scars after two or more previous C-sections were indications for cesarean delivery in 18/50 (36%) patients in the comparison group, as well as after one cesarean section in combination with other indications: severe preeclampsia – 15/50 (30%), premature rupture of membranes and cervical incompetence – 7/50 (14%), placental insufficiency and FGR – 5/50 (10%), abnormal fetal presentation – 1/50 (2%), uterine scar insufficiency – 1/50 (2%), intrahepatic cholestasis – 1/50 (2%), and preterm placental abruption – 1/50 (2%).
Elective cesarean deliveries were performed in 28/57 (49.1%) patients in the main group at 34–36 weeks according to the clinical recommendations, and emergency deliveries were performed in 24/57 (42.1%) women due to vaginal bleeding before 34 weeks of gestation – 19/57 (33.3%) women, the symptoms of preterm labor was – 3/57 (5.26%), antenatal fetal death – 1/57 (1,75%), and the symptoms of uterine scar insufficiency – 1/57 (1.75%)
Vaginal deliveries were in 5/57 (8.8%) patients without placenta previa in the main group, and in 1/50 (2%) patient in the comparison group.
Table 4 shows patient distribution in the main group depending on the grade of placenta accreta according to FIGO classification based on the results of morphological examination and topographic classification (Table 5)
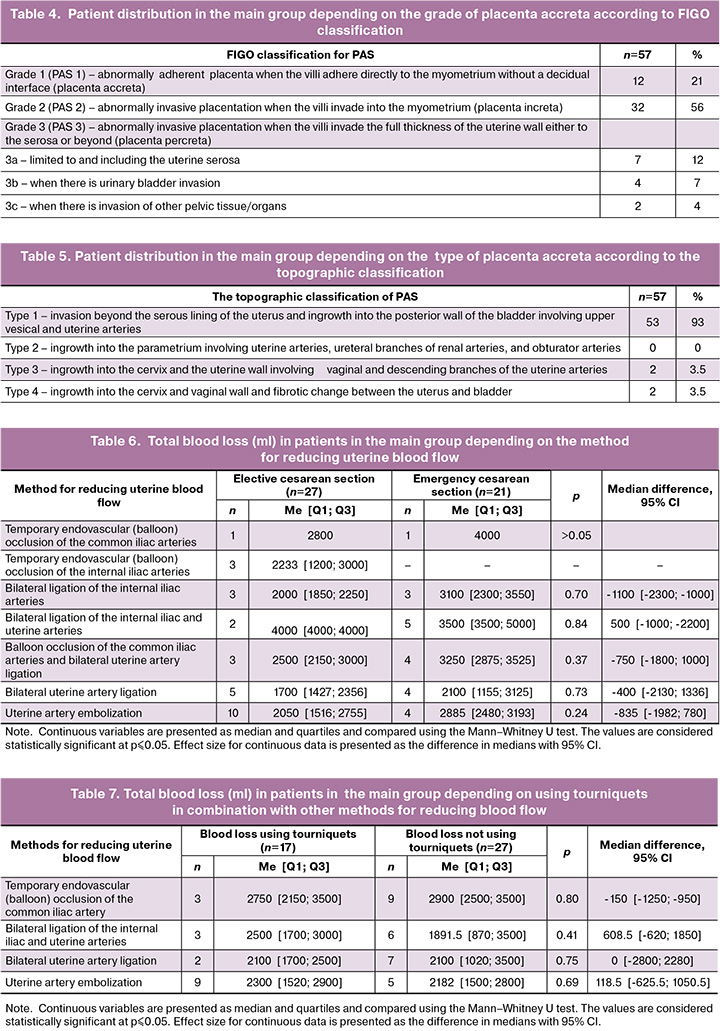
Total blood loss was 5000 ml in patients in the main group, and 1100 ml in patients in the comparison group. Table 6 presents maximum blood loss in patients in the main group during elective and emergency deliveries, depending on the method used to reduce uterine blood flow.
Comparison of total blood loss in patients in the main group depending on using or not using tourniquets as an additional method for reduction of blood loss is presented in Table 7.
Discussion
Based on this study, it should be noted that patients’ age, parity of pregnancies and births, as well as somatic history did not make it possible to identify the group of patients at risk for PAS.
Analysis of obstetric and gynecological history showed that patients with PAS had two or more uterine scars after previous cesarean sections and underwent intrauterine interventions significantly more often compared with patients without PAS. At the same time, chronic endometritis was observed in a small number of patients in both groups. There were no cases of specific urogenital infections (sexually transmited infections) among the examined patients in both groups.
Analysis of current pregnancy-related complications found that in women with PAS pregnancies were complicated with threatened preterm labor in the second trimester. At the same time, there were no statistically significant differences found in the incidence of threatened spontaneous miscarriage in the first trimester in pregnant women with or without vaginal bleeding in both groups. Furthermore, no cases of PE, placental insufficiency, or FGR, as well as no cases of high risk for these complications according to biochemical screening test in the first trimester were observed in patients with PAS. Accordingly, it can be suggested, that both the presence of PE and placental insufficiency, and identification of patients with a high risk for developing these complications is incompatible with the diagnosis of PAS.
Analysis of indications for delivery in patients in the main group showed that only one third of women – 19/57 (33.3%) had emergency delivery due to bleeding before 34 weeks of pregnancy. Among them, 9/19 (47.4%) women had pregnancy complications – threatened spontaneous miscarriage with bleeding, and preterm labor. In the remaining 10/19 (52.6%) the course of pregnancy was without the signs of threatened miscarriage at any pregnancy stage. Therefore, it is not appropriate to consider these pregnancy complications as mandatory predictors of bleeding in placenta accreta and placenta previa. It is also of interest that bleeding occurred at different stages of pregnancy. In 3/57 (5.3%) observations, bleeding occurred at 22–27.6 weeks of gestation, in 7/57 (12.3%) at 28–31.6 weeks, in 3/57 (5.3%) at 32–33.6 weeks, and in 6/57 (10.5%) at 34–36.6 weeks.
Assessment of blood loss during elective and emergency delivery showed that the volume of blood loss was higher during emergency delivery versus elective delivery regardless of the method used to reduce blood flow. However, the differences were not statistically significant. Whereas comparison of blood loss with or without using tourniquets as additional method for reducing blood flow showed that blood loss was lower in patients with temporary endovascular (balloon) occlusion of the internal iliac artery in combination with tourniquets compared with not using tourniquet hemostasis. Conversely, blood loss was higher using tourniquets in patients with bilateral ligation of the internal iliac and uterine arteries, as well as uterine artery embolization. However, the differences were not statistically significant.
Thus, the results of our study showed that patients with PAS experiencing placenta previa before 34 weeks of gestation have 33% higher risk of bleeding, that is primarily mediated by placenta previa in combination with formation of the lower uterine segment during pregnancy. In the presence of sonographic signs of PAS in the presence of placenta previa, the risk of bleeding was s is at 24–26 weeks of gestation (when formation of the lower uterine starts), or in some cases at 28–31.6 and 34–36.6 weeks. For example, with placental attachment at the site of the previous C-section scar, but without placenta previa, the risk of bleeding was clinically significant only in the third stage of labor.
Conclusion
Given the above, it is important to understand that, in the absence of statistically significant differences between blood loss during elective and emergency deliveries, as well as with similar risks of bleeding in the second trimester of pregnancy, the optimal timing for delivery is 36 weeks – 36 weeks and 6 days in the absence of vaginal bleeding in women with PAS, who give birth in the third-level maternity hospitals. This can help to prevent perinatal complications due to preterm birth. In the absence of statistically significant differences in the volume of blood loss by using different techniques of uterine devascularization to reduce blood flow, any method is appropriate and justified. However, due to a small number of observations (n=57), further research is needed to evaluate properly the advantages and shortcoming of each method.
References
- Министерство здравоохранения Российской Федерации. Клинические рекомендации. Патологическое прикрепление плаценты (предлежание и врастание плаценты). 2023. [Ministry of Health of the Russian Federation. Clinical guidelines. Pathological attachment of the placenta (placenta previa and placenta accreta). 2023 (in Russian)].
- De Mucio B., Serruya S., Alemán A., Castellano G., Sosa C.G. A systematic review and meta-analysis of cesarean delivery and other uterine surgery as risk factors for placenta accrete. Int. J. Gynecol. Obstet. 2019. 147(3): 281-91. https://dx.doi.org/10.1002/ijgo.12948
- Einerson B.D., Weiniger C.F. Placenta accreta spectrum disorder: updates on anesthetic and surgical management strategies. Int. J. Obstet. Anesth. 2021; 46: 102975. https://dx.doi.org/10.1016/j.ijoa.2021.102975
- Listijono D.R., Chuah S.C., Rahimpanah F. Management of placenta accreta in an Australian tertiary referral centre: a ten-year experience. Clin. Exp Obstet. Gynecol. 2017; 44(3): 374-8.
- Thang N.M., Anh N.T.H., Thanh P.H., Linh P.T., Cuong T.D. Emergent versus planned delivery in patients with placenta accreta spectrum disorders: a retrospective study. Medicine (Baltimore). 2021; 100(51): e28353. https://dx.doi.org/10.1097/MD.0000000000028353
- Salmanian B., Einerson B.D., Carusi D.A., Shainker S.A., Nieto-Calvache A.J., Shrivastava V.K. et al. Timing of delivery for placenta accreta spectrum: the Pan-American Society for the placenta accreta spectrum experience. Am. J. Obstet. Gynecol. MFM. 2022; 4(6): 100718. https://dx.doi.org/10.1016/j.ajogmf.2022.100718
- Сухих Г.Т., Шмаков Р.Г., Курцер М.А., Баринов С.В., Чупрынин В.Д., Михеева А.А., Амирасланов Э.Ю., Григорьян А.М., Коноплев Б.А., Кутакова Ю.Ю., Карапетян Т.Э., Васильченко О.Н., Забелина Т.М., Серова О.Ф., Пырегов А.В., Куликов И.А., Белоусова Т.Н., Плахотина Е.Н., Пенжоян Г.А., Макухина Т.Б., Андреева М.Д., Шаповалова О.А., Каменских Г.В., Лобач С.В., Позднякова Т.И., Савельева И.В., Бухарова Е.А., Носова Н.В., Цхай В.Б., Распопин Ю.С., Гарбер Ю.Г., Веккер И.Р., Журлова О.Н., Нечаева М.В., Белоконева Т.С., Протопопова Н.В., Дудакова В.Н., Новичков Д.А., Хворостухина Н.Ф., Владимирова Н.Ю., Федорова К.В., Ринчиндоржиева М.П., Тудупова Б.Б., Панова Т.В., Гусева О.И., Мануйленко О.В., Брум О.Ю., Дорфман О.В., Веровская Т.А., Бабаева Л.К., Рымашевский А.Н., Хвалина Т.В., Дмитриева С.Л., Горев С.Н., Малышкина А.И., Песикин О.Н., Зубенко В.Б., Лобач Н.В., Брюхачева Т.В., Караваева А.А., Беженарь В.Ф., Аракелян Б.В., Ральникова А.Ю., Курлеева Т.Ю., Вологодская Е.В., Дикарёва Л.В., Малышева И.П., Идам-Сюрюн А.М., Шакурова Е.Ю., Семёнов Ю.А., Шерстобитов А.В., Фортыгин А.Я., Зюзин Е.С., Кашлевская Н.Л., Шамина М.С., Густоварова Т.А., Хаванский В.А., Щегольков М.Е., Кулакова С.А., Клишина В.В. Хирургическое лечение при врастании плаценты в Российской Федерации (пилотное многоцентровое исследование). Акушерство и гинекология. 2024; 1: 50-66. [Sukhikh G.T., Shmakov R.G., Kurtser M.A., Barinov S.V., Chuprynin V.D., Mikheeva A.A., Amiraslanov E.Yu., Grigoryan A.M., Konoplev B.A., Kutakova Yu.Yu., Karapetyan T.E., Vasilchenko O.N., Zabelina T.M., Serova O.F., Pyregov A.V., Kulikov I.A., Belousova T.N., Plakhotina E.N., Penzhoyan G.A., Makukhina T.B., Andreeva M.D., Shapovalova O.A., Kamenskikh G.V., Lobach S.V., Pozdnyakova T.I., Saveljeva I.V., Bukharova E.A., Nosova N.V., Tskhay V.B., Raspopin Yu.S., Garber Yu.G., Wecker I.R., Zhurlova O.N., Nechaeva M.V., Belokoneva T.S., Protopopova N.V., Dudakova V.N., Novichkov D.A., Khvorostukhina N.F., Vladimirova N.Yu., Fedorova K.V., Rinchindorzhieva M.P., Tudupova B.B., Panova T.V., Guseva O.I., Manuilenko O.V., Brum O.Yu., Dorfman O.V., Verovskaya T.A., Babaeva L.K., Rymashevsky A.N., Khvalina T.V., Dmitrieva S.L., Gorev S.N., Malyshkina A.I., Pesikin O.N., Zubenko V.B., Lobach N.V., Briukhacheva T.V., Karavaeva A. A.A., Bezhenar V.F., Arakelyan B.V., Ralnikova A.Yu., Kurleeva T.Yu., Vologodskaya E.V., Dikareva L.V., Malysheva I.P., Idam-Syuryun A.M., Shakurova E.Yu., Semenov Yu.A., Sherstobitov A.V., Fortygin A.Ia., Ziuzin E.S., Kashlevskaya N.L., Shamina M.S., Gustovarova T.A., Khavansky V.A., Shchegolkov M.E., Kulakova S.A., Klishina V.V. Surgical management of placenta accreta spectrum in the Russian Federation (a pilot multicenter study). Obstetrics and Gynecology. 2024; (1): 50-66 (in Russian)]. https://dx.doi.org/10.18565/aig.2023.306
- Куликов И.А., Шмаков Р.Г., Белоусова Т.Н., Плахотина Е.Н., Низяева Н.В., Гейлис И.А., Искаков Д.Д., Милютина Е.Р., Вдовиченко Е.А., Прочаковский Д.В. Эффективность метода дистального компрессионного гемостаза с применением турникетов в сочетании с баллонной тампонадой влагалища вагинальным катетером Жуковского при родоразрешении беременных с врастанием плаценты. Акушерство и гинекология. 2022; 10: 58-66. [Kulikov I.A., Shmakov R.G., Belousova T.N., Plakhotina E.N., Nizyaeva N.V., Geilis I.A., Iskakov D.D., Milyutina E.R., Vdovichenko E.A., Prochakovsky D.V. Efficacy of the tourniquet hemostasis combined with vaginal balloon tamponade using a Zhukovsky vaginal catheter during delivery in placenta accreta spectrum. Obstetrics and Gynecology. 2022; (10): 58-66 (in Russian)]. https://dx.doi.org/10.18565/aig.2022.10.58-66
- Куликов И.А., Белоусова Т.Н., Плахотина Е.Н., Искаков Д.Д., Милютина Е.Р. Возможности уменьшения объема кровопотери при выполнении органосохраняющих операций у беременных с врастанием плаценты. Российский вестник акушера-гинеколога. 2022; 22(6): 54-60. [Kulikov I.A., Belousova T.N., Plakhotina E.N., Iskakov D.D., Milutina E.R. Possibilities to reduce blood loss during organ-preserving surgery in pregnant women with placenta ingrowth. Russian Bulletin of Obstetrician-Gynecologist. 2022; 22(6): 54-60 (in Russian)]. https://dx.doi.org/10.17116/rosakush20222206154
- Приходько А.М., Амирасланов Э.Ю., Чупрынин В.Д., Кан Н.Е., Баев О.Р. Алгоритм медицинской помощи при врастании плаценты. Акушерство и гинекология. 2024; 11: 200-6. [Prikhodko A.M., Amiraslanov E.Yu., Chuprynin V.D., Kan N.E., Baev O.R. Algorithm for providing medical care to patients with placenta percreta. Obstetrics and Gynecology. 2024; (11): 200-6 (in Russian)]. https://dx.doi.org/10.18565/aig.2024.179
Received 10.11.2025
Accepted 03.03.2026
About the Authors
Irina V. Ignatko, Dr. Med. Sci., Corresponding Member of the Russian Academy of Sciences, Professor of the Russian Academy of Sciences, Professor, Head of the Department of Obstetrics, Gynecology, and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine, I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2, ignatko_i_v@staff.sechenov.ru,https://orcid.org/0000-0002-9945-3848
Vladimir A. Lebedev, Dr. Med. Sci., Professor, Professor at the Department of Obstetrics, Gynecology, and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine,
I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2, lebedev_v_a@staff.sechenov.ru, https://orcid.org/0000-0002-3302-5533
Irina M. Bogomozova, PhD, Associate Professor, Associate Professor at the Department of Obstetrics, Gynecology and Perinatology, N.V. Sklifosovsky Institute of
Clinical Medicine, I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2, +7(926)305-04-03, bogomazova_i_m@staff.sechenov.ru, https://orcid.org/0000-0003-1156-7726
Elena V. Timokhina, Dr. Med. Sci., Associate Professor, Professor at the Department of Obstetrics, Gynecology, and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine, I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2, timokhina_e_v@staff.sechenov.ru, https://orcid.org/0000-0001-6628-0023
Vera S. Belousova, Dr. Med. Sci., Associate Professor, Professor at the Department of Obstetrics, Gynecology, and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine, I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2, belousova_v_s@staff.sechenov.ru, https://orcid.org/0000-0001-8332-7073
Vladimir M. Pashkov, Dr. Med. Sci., Professor, Professor at the Department of Obstetrics, Gynecology and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine,
I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8,
bldg. 2, pashkov_v_m@staff.sechenov.ru, https://orcid.org/0000-0003-3768-7822
Irina A. Fedyunina, PhD, Associate Professor at the Department of Obstetrics, Gynecology, and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine,
I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2, fedyunina_i_a@staff.sechenov.ru, https://orcid.org/0000-0002-9661-5338
Dzhamilya Kh. Sarakhova, PhD, Deputy Chief Physician for Obstetrics and Gynecology, S.S. Yudin City Clinical Hospital, Moscow Healthcare Department, 115446, Russia, Moscow, Kolomenskiy proezd, 4, bldg. 2, dzh2010@yandex.ru, https://orcid.org/0009-0008-0531-0899
Yulia A. Samoylova, PhD, Head of the 1st Obstetrics Department of Pregnancy Pathology, S.S. Yudin City Clinical Hospital, Moscow Healthcare Department, 115446, Russia, Moscow, Kolomenskiy proezd, 4, bldg. 2, Samoylova2005@yandex.ru, https://orcid.org/0000-0001-7448-515X
Alina B. Samara, Resident at the Department of Obstetrics, Gynecology and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine, I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2, linaasamaraa@gmail.com,
https://orcid.org/0000-0001-8266-6524
Sevda F. Askerova, PhD student at the Department of Obstetrics, Gynecology and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine, I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2, askerova_sevda12@mail.ru, https://orcid.org/0000-0002-4925-594X
Vladimir V. Gutsu, PhD student at the Department of Obstetrics, Gynecology, and Perinatology, N.V. Sklifosovsky Institute of Clinical Medicine, I.M. Sechenov First Moscow State Medical University, Ministry of Health of the Russian Federation (Sechenov University), 119991, Russia, Moscow, Trubetskaya str., 8, bldg. 2; Obstetrician-Gynecologist at the Consultative and Diagnostic Department of Maternity Hospital, S.S. Yudin City Clinical Hospital, Moscow Healthcare Department, 115446, Russia, Moscow, Kolomenskiy proezd, 4, bldg. 2, gutsu_vladimir@mail.ru, https://orcid.org/0009-0000-3712-3280
Corresponding author: Irina M. Bogomazova, bogomazova_i_m@staff.sechenov.ru



