Neonatal outcomes in patients with placenta ingrowth
Kayumova A.V., Semenov Yu.A., Melkozerova O.A., Kinzhalova S.V., Gaeva A.I., Shakirova K.P., Makarov R.A.
Objective: To study the characteristics of newborns’ adaptation, neonatal complications and outcomes depending on the time of delivery in patients with and without pathological placental invasion.
Materials and methods: The study comprised 69 birth and neonatal records to evaluate the early neonatal period in terms of perioperative management, blood loss volume, and neonatal intensive care, depending on the gestational age and indications for delivery: 43 cases with placenta increta and 26 cases without this pathology. Statistical analysis was based on Fisher's exact test for categorical variables and the Mann–Whitney test for quantitative variables.
Results: No statistically significant differences were found between the subgroups of patients with placenta ingrowth depending on the gestational age for the parameters studied: Apgar score at 1, 5, and 10 minutes; need for and duration of respiratory therapy; duration of antibacterial therapy; length of stay in the neonatal intensive care unit (p>0.05).
Infants born to mothers with pathological placental invasion are substantially more likely to develop severe respiratory distress syndrome, requiring mechanical ventilation and surfactant administration. No statistically significant differences in the use of antibacterial therapy were found between the study groups.
Conclusion: The delivery time in patients with placenta ingrowth depends on maternal safety management. Early elective delivery at 34 weeks of gestation (if possible), is justified, given the nature of life-threatening complications for both mother and fetus that occur with emergency delivery.
Authors’ contributions: Kayumova A.V. – study concept and design; Kayumova A.V., Gaeva A.I., Shakirova K.P. – collection and processing of material; Kayumova A.V., Kinzhalova S.V., Makarov R.A. – manuscript composition; Semenov Yu.A., Melkozerova O.A. – manuscript editing.
Conflicts of interest: The authors declare no conflicts of interest.
Funding: The study had no sponsorship support.
Ethical Approval: The study was approved by the local bioethics committee of the Ural Research Institute of Maternity and Child Care, Ministry of Health of Russia.
Patient Consent for Publication: The patients signed informed consent for the publication of their data.
Authors' Data Sharing Statement: The data supporting the findings of this study are available on request from the corresponding author after approval from the principal investigator.
For citation: Kayumova A.V., Semenov Yu.A., Melkozerova O.A., Kinzhalova S.V., Gaeva A.I.,
Shakirova K.P., Makarov R.A. Neonatal outcomes in patients with placenta ingrowth.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2026; (1): 55-61 (in Russian)
https://dx.doi.org/10.18565/aig.2025.266
Keywords
Placenta accreta spectrum (placenta ingrowth) is an abnormal invasion and penetration of the trophoblast into the myometrium, when part or the entire placenta remains attached to the uterus and cannot separate on its own [1]. According to current data, the incidence of pathological placental invasion varies from 1 in 250 to 500 births and has been increasing in recent decades, which is indicative of the growing rate of cesarean sections as a leading risk factor [2].
Placenta ingrowth is recognized worldwide as a very high-risk obstetric condition associated with massive hemorrhage, disabling surgeries, and maternal mortality. However, there is no general consensus on the management of pregnant women with this condition: the optimal timing of delivery is not strictly defined, and different guidelines report different recommendations on the timing of delivery for patients with pathological placental invasion [3].
To reduce maternal morbidity in women with placenta accreta there should be a tendency towards planned delivery. When deciding on the timing of delivery, it is necessary to balance the maternal risk associated with severe antepartum hemorrhage and emergency surgery by a non-target team of doctors with the risks and benefits for the fetus and neonate [4].
While maternal outcomes of pregnancies complicated by pathological placental invasion are well described, reports on neonatal outcomes are very limited. Some retrospective studies have reported high rates of neonatal intensive care unit (NICU) admission and relatively high need for artificial lung ventilation (ALV), but these studies focused on a limited number of neonatal outcomes and did not compare data on pregnancies without pathological placental invasion [5–7].
All of the above mentioned confirms the objective of our study.
Objective: to identify the characteristics of newborn adaptation, neonatal complications and outcomes depending on the time of delivery in patients with and without pathological placental invasion.
Materials and methods
A retrospective analysis of 69 delivery histories and neonatal medical records was conducted to assess the early neonatal period from the perspective of the perioperative management, timing of delivery, volume of blood loss, and medical care in the NICU.
All patients delivered between 330 and 356 weeks of gestation. The patients were divided into two groups: the study group (43 patients who delivered due to placenta accreta spectrum, and their newborns); and the comparison group (26 patients who delivered by cesarean section without placenta accreta, and their newborns).
The first stage of the study involved a comparative analysis of neonatal outcomes for mothers with placenta ingrowth, based on gestational age at delivery, and was presented with the following subgroups: Subgroup 1 – 330–336 weeks (n=6); Subgroup 2 – 340–346 weeks (n=26); Subgroup 3 – 350–356 weeks (n=11).
In the second stage of the study a comparative analysis of neonatal outcomes was conducted in patients with placenta accreta spectrum (the study group) and without this pathology (the comparison group) at similar gestational age of 330–356 weeks. Exclusion criteria: vaginal delivery, multiple pregnancies complicated by twin-to-twin transfusion syndrome and fetal surgery, delivery with a prolonged time between rupture and delivery, and the presence of chorionic amnionitis.
A clinical assessment of the newborns’ health status was made using a statistical chart. All data were obtained from electronic delivery records and newborn medical records. The analysis included the time of delivery, type of anesthesia, volume of blood loss, minute of fetal extraction, Apgar score at 1, 5 and 10 minutes, types of respiratory support for newborns and its duration, presence of infectious disease, use of antibacterial therapy, surfactant therapy and duration of stay in the NICU.
Statistical analysis
Statistical analysis was performed with Excel 2010 and SPSS Statistics version 26.0. Statistical comparisons of quantitative variables were made by calculating medians (Me), lower and upper quartiles (Q1; Q3). Significance was assessed using the Mann–Whitney test for two comparison groups; for three comparison groups we used the Kruskal–Wallis test. Categorical or qualitative variables are presented as absolute frequency of occurrence of the characteristic (n) and percentages (%). Fisher's exact test was used to assess the significance of frequencies. Risks were identified by the odds ratio (OR) with the value of one of the indicators in the comparison group accounted for zero and with the relative risk (RR) with a 95% confidence interval (CI). When comparing three groups, the critical level was reduced to 0.017 using the Bonferroni correction.
Results
Between 2024 and 2025, 48 patients with placenta accreta were delivered at the Ural Research Institute for Maternal and Child Care of the Ministry of Health of Russia at 254–364 weeks of gestation, resulting in 49 births. The cohort of patients at 330–356 weeks of gestation was of particular interest, with the need to determine the optimal gestational age in terms of neonatal complications and outcomes. Therefore, only 42 patients and 43 newborns were included in the main study group. The comparison group included all patients who delivered in 2025 at the above-mentioned gestational period. The exceptions comprised the following complications: delivery of multiple pregnancies with twin-to-twin transfusion syndrome and the use of fetal surgery during pregnancy, delivery with a prolonged time between rupture and delivery, and the presence of chorioamnionitis.
At the first stage of the study we assessed the frequency and structure of neonatal complications depending on the gestational age at the time of delivery in patients with placenta accreta spectrum.
The 1st subgroup (330–336 weeks) consisted of 6 newborns, the 2nd (340–346 weeks) – of 26 children, the 3rd (350–356 weeks) – of 11. It is worth noting that the 34.0–34.6 subgroup was the largest.
Despite statistically significant differences between the subgroups regarding the timing of delivery, no differences in the body weight of newborns were noted: in the 1st subgroup the body weight of newborns was 2420.0 (2225.0; 2540.0) g, in the 2nd – 2465.0 (2157.5; 2707.5) g, while in the 3rd subgroup it accounted for 2490.0 (2325.0; 2870.0) g, p>0.05.
There were no statistically significant differences between the subgroups for other indicators: Apgar score at 1, 5, and 10 minutes; need and duration of respiratory support; duration of antibacterial therapy; length of stay in the NICU, p>0.05 (Table 1).
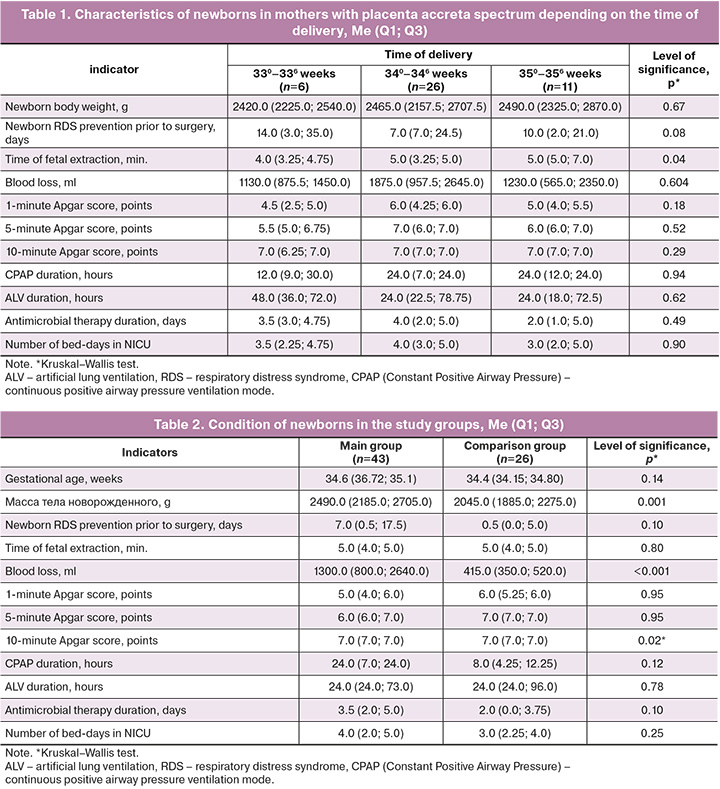
All infants whose mothers delivered with placenta ingrowth required admission to the NICU for respiratory support. This indicates that preterm and late preterm infants require intensive care and stabilization due to morphofunctional immaturity.
Thus, our results indicate the absence of statistically significant differences in major clinical characteristics of late preterm newborns born from mothers with placenta ingrowth.
Comparative analysis of the major parameters of the newborns from mothers with placenta accreta spectrum (main group) and newborns delivered by caesarean section without placenta accreta (comparison group) in their mothers are presented in Table 2.
Despite the absence of differences in the term of delivery, newborns in the comparison group had a statistically significant lower birth weight: in the main group it was 2490.0 (2185.0; 2705.0) grams, while in in the comparison group it accounted for 2045.0 (1885.0; 2275.0) grams, p<0.001. This may be due to the fact that the comparison group included mainly patients with severe preeclampsia and fetal growth retardation.
Newborns in the study group had significantly lower 10-minute Apgar score and were more likely to receive ALV; however, this did not result in a prolonged respiratory support. In the main group ALV was used in 23/43 (53.8%) newborns, compared to only 4/26 (15.4%) in the comparison group, p=0.002 (Table 3).
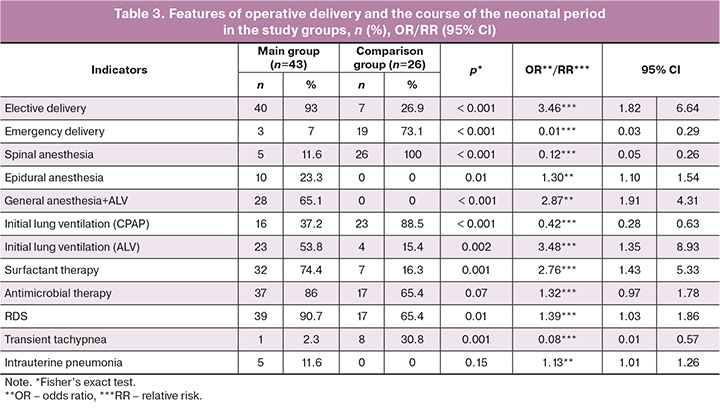
It can be assumed that a lower Apgar score and initial ALV were associated with a more frequent use of general anesthesia with ALV in women from the main group – 28/43 (65.1%); though in the comparison group this method of anesthesia was not applied, p<0.001 (Table 3). The choice of anesthesia type depends on the degree of placenta ingrowth and the expected massive blood loss associated with this pathology, which comprised 1300.0 (800.0; 2640.0) ml in the main group, while in the comparison group it was 415.0 (350.0; 520.0) ml, p<0.001.
There were no statistically significant differences in the duration of antimicrobial therapy and the length of stay in the NICU in the groups, p>0.05 (Table 2).
A detailed analysis of the course of the neonatal period in the study groups is presented in Table 3.
The severity of the newborns’ condition at birth was caused by respiratory disorders, which can develop as a result of respiratory distress syndrome (RDS) and transient tachypnea of the newborn or intrauterine pneumonia. There were no statistically significant differences in the impact of intrauterine pneumonia on respiratory failure in the study groups: 5/43 (11.6%) – in the main group, while in comparison group there were no children with intrauterine pneumonia, p>0.05. When analyzing respiratory disorders, we found that in the main group RDS was recorded more often – in 39/43 (90.7%) newborns, while in the comparison group it was detected in 17/26 (65.4%), p=0.01. In the comparison group, on the contrary, transient tachypnea prevailed (in 8/26 (30.8%) newborns,), while in the main group this complication was observed in only 1/43 (2.3%) children, p=0.001.
Newborns from both study groups required respiratory support in the delivery room so far. To stabilize respiratory function, the infants were provided with AVL significantly more often – 23/34 (53.8%), compared to the comparison group (4/26 which is 15.4%), p=0.002. On the contrary, respiratory disorders in the comparison group had a more favorable course, since the disease resulted from transient tachypnea of the newborn. Stabilization of the condition due to non-invasive respiratory support with the CPAP method within this group happened statistically more often than in the main group: 23/36 (88.5%) versus 16/43 (37.2%), p<0.001.
Surfactant therapy was provided significantly more often in children from the main study group – 32/43 (74.4%), than in those patients of the comparison group – 7/26 (16.3%), p=0.001, which was also associated with the higher frequency rate of severe forms of RDS in newborns from the main study group (Table 3).
After analyzing the use of antibacterial therapy, no statistically significant differences were found in the study groups, p>0.05.
Discussion
The choice of the optimal time for delivery in patients with placenta accreta spectrum is a crucial obstetrical challenge in the context of finding a balance between the risks of neonatal complications associated with the functional immaturity of newborns during preterm delivery and the risks of emergency cesarean section, often accompanied by life-threatening conditions in mothers.
The clinical guidelines of the Royal College of Obstetricians and Gynecologists recommend to provide a delivery at 350–366 weeks of gestation in women with placenta accreta, if there are no risk factors for preterm birth. It is believed that such timing is associated with the best correlation between fetal lung maturity and the risk of unplanned delivery [8].
National guidelines of Ireland suggest considering delivery for women with suspected placenta accreta starting from 34 weeks of gestation and no later than 366 weeks. Each case should be discussed at a medical board meeting to identify the most appropriate timing of delivery, given individual risk factors [9]. Similar recommendations are provided by international professional organizations, including ACOG and SMFM [10].
The FIGO international guidelines encourage providing delivery from 340 weeks. However, it is reasonable to wait until 366 weeks if the woman has not previously experienced antenatal bleeding or risk factors for preterm birth [11]. Colombian colleagues share similar opinion [12].
National clinical guidelines in Australia and New Zealand do not specify precise intervals but advise that the timing of delivery in women with suspected or confirmed pathological placental ingrowth should be based on clinical findings and the need to provide fetal lung maturation [13].
Literature review showed that 34 weeks of pregnancy is the optimal time, given the ability of large medical institutions to handle neonatal complications at this period. At 36 weeks of pregnancy and over approximately half of women with placenta accreta require emergency delivery, which is associated with a high risk of maternal complications [14].
According to the research data, there are no large randomized trials on neonatal outcomes in placenta accreta in the Russian Federation. Belousova T.N. et al. report a decline in the need for respiratory support and NICU stay with increasing gestational age [15]. Other authors also confirm that the incidence rate of neonatal complications depends on gestational age: the greater the gestational age, the fewer complications [16].
Colleagues from other countries have found that the incidence rate of adverse neonatal outcomes was significantly higher in the group with placenta accreta compared to the group without pathological placental invasion [17]. According to various authors, there was a lower Apgar score, more frequent AVL, and RDS cases, which is consistent with the results obtained in our study [18–20].
Furthermore, we detected significantly higher birth weight in neonates born to mothers with placenta accreta spectrum compared to those without placental ingrowth. Liang Y. et al. report that abnormal fetal growth in pregnancies complicated by placenta accreta may be the result of abnormal placental implantation [21]. In our study, the comparison group included patients requiring emergency delivery due to severe pre-eclampsia or decompensated feto-placental insufficiency, which obviously points to lower birth weight in newborns at the time of delivery [22].
Conclusion
The timing of delivery should be chosen according to maternal safety factors. Elective early delivery (34 weeks) is recommended given the nature of life-threatening complications for both mother and fetus that may occur with emergency delivery.
Infants born to mothers with placenta accreta are more likely to develop severe RDS, requiring more frequent AVL and surfactant administration. This may be due to the greater morphofunctional immaturity of lung tissue at birth, resulting from pathological placentation and decreased tissue perfusion. Further research is needed for better understanding of the pathogenesis.
References
- Nieto-Calvache A., Palacios-Jaraquemada J., Hussein A., Jauniaux E., Milani Coutinho C., Rijken M. Management of placenta accreta spectrum in low- and middle-income countries. Best Pract. Res. Clin. Obstet. Gynaecol. 2024; 94: 102475. https://dx.doi.org/10.1016/j.bpobgyn.2024.102475
- Zhong W., Zhu F., Li S., Chen J., He F., Xin J. et al. Maternal and neonatal outcomes after planned or emergency delivery for placenta accreta spectrum: a systematic review and meta-analysis. Front. Med. 2021; 28(8): 731412. https://dx.doi.org/10.3389/fmed.2021.731412
- Bonanni G., Lopez-Giron M., Allen L., Fox K., Silver R.M., Hobson S.R. et al. Guidelines on placenta accreta spectrum disorders: a systematic review. JAMA Netw Open. 2025; 8(7): e2521909. https://dx.doi.org/10.1001/jamanetworkopen.2025.21909
- Hobson S., Kingdom J., Murji A., Windrim R.C., Carvalho J.C.A., Singh S.S. et al. No. 383-screening, diagnosis, and management of placenta accreta spectrum disorders. J. Obstet. Gynaecol. Can. 2019; 41(7): 1035-49. https://dx.doi.org/10.1016/j.jogc.2018.12.004
- Morlando M., Schwickert A., Stefanovic V., Gziri M.M., Pateisky P., Chalubinski K.M. et al. Maternal and neonatal outcomes of elective and emergency cesarean section in the placenta accreta spectrum: a multinational database study. Acta Obstet. Gyneco.l Scand. 2021; 100(S1): 41-9. https://dx.doi.org/10.1111/AOGS.14120
- Jauniaux E., Alfirevic Z., Bhide A.G., Belfort M.A., Burton G.J., Collins S.L. et al.; Royal College of Obstetricians and Gynaecologists. Placenta praevia and placenta accreta: diagnosis and management: Green-top Guideline No. 27a. BJOG. 2019; 126(1): e1-e48. https://dx.doi.org/10.1111/1471-0528.15306
- Bartels H.C., Walsh J.M., Ní Mhuircheartaigh R., Brophy D., Moriarty J., Geoghegan T. et al. National Clinical Practice Guideline: Diagnosis and management of placenta accreta spectrum. National Women and Infants Health Programme and the Institute of Obstetricians and Gynaecologists. December 2022. Available at: https://www.hse.ie/eng/about/who/acute-hospitals-division/woman-infants/clinical-guidelines
- Salmanian B., Einerson B., Carusi D., Shainker S.A., Nieto-Calvache A.J., Shrivastava V.K. et al. Timing of delivery for placenta accreta spectrum: the Pan-American Society for the Placenta Accreta Spectrum experience. Am. J. Obstet. Gynecol. MFM. 2022; 4(6): 100718. https://dx.doi.org/10.1016/j.ajogmf.2022.100718
- Allen L., Jauniaux E., Hobson S., Papillon-Smith J., Belfort M.A.; FIGO Placenta Accreta Diagnosis and Management Expert Consensus Panel. FIGO consensus guidelines on placenta accreta spectrum disorders: Nonconservative surgical management. Int. J. Gynaecol. Obstet. 2018; 140(3): 281-90. https://dx.doi.org/10.1002/ijgo.12409
- Nieto-Calvache A.J., Sanín-Blair J.E., Buitrago-Leal H.M., Benavides-Serralde J.A., Maya-Castro J., Rozo-Rangel A.P. et al. Colombian Consensus on the Treatment of Placenta Accreta Spectrum (PAS). Rev. Colomb. Obstet. Ginecol. 2022; 73(3): 283-316. https://dx.doi.org/110.18597/rcog.3877
- RANZCOG Women’s Health Committee. Placenta accreta spectrum (PAS) (C-Obs 20). 2024. Available at: https://ranzcog.edu.au/wp-content/uploads/Placenta-Accreta-Spectrum.pdf
- Obstetric Care Consensus No. 7: Placenta accreta spectrum. Obstet. Gynecol. 2018; 132(6): 259-75. https://dx.doi.org/10.1097/AOG.0000000000002983
- Белоусова Т.Н., Князева Н.Ю., Голосная Г.С., Куликов И.А., Новиков М.Ю., Холичев Д.А. Особенности течения неонатального периода при врастании плаценты. Эффективная фармакотерапия. 2023; 19(44): 20-4. [Belousova T.N., Knyazeva N.Yu., Golosnaya G.S., Kulikov I.A.,Novikov M.Yu., Kholichev D.A. Features of the course of the neonatal period with placenta accretion. Effective Pharmacotherapy. 2023; 19(44): 20-4 (in Russian)]. https://dx.doi.org/10.33978/2307-3586-2023-19-44-20-24
- Макухина Т.Б., Пенжоян Г.А., Модель Г.Ю., Донцова М.В. Исходы беременностей высокого риска врастания плаценты в зависимости от порядка оказания помощи при родоразрешении. Женское здоровье и репродукция. 2023; 4(59): 1-9. [Makukhina T.B., Penzhoyan G.A., Model G.Yu., Dontsova M.V. Outcomes of high-risk pregnancies with placenta accreta, depending on the order of delivery. Women's Health and Reproduction. 2023; 4(59): 1-9 (in Russian)]. https://dx.doi.org/10.31550/2712-8598-ZhZiR-2023-4-1
- Toussia-Cohen Sh., Castel E., Friedrich L., Mor N., Ohayon A., Levin G. et al. Neonatal outcomes in pregnancies complicated by placenta accreta- a matched cohort study. Arch. Gynecol. Obstet. 2024; 310(1): 269-75. https://dx.doi.org/10.1007/s00404-023-07353-6
- Kasraeian M., Hashemi A., Hessami K., Alamdarloo S.M., Vahdani R., Vafaei H. et al. A 5-year experience on perinatal outcome of placenta accreta spectrum disorder managed by cesarean hysterectomy in southern Iranian women. BMC Womens Health. 2021; 21(1): 243. https://dx.doi.org/10.1186/S12905-021-01389-Z
- Palacios-Jaraquemada J., Basanta N., Fiorillo A., Labrousse C., Martínez M. Neonatal outcome after conservative-reconstructive surgery for placenta accreta spectrum disorders. J. Matern Fetal. Neonatal. Med. 2022; 35(25): 4994-6. https://dx.doi.org/10.1080/14767058.2021.1873944
- Bartal M., Papanna R., Zachariaset N., Soriano-Calderon N., Limas M., Blackwell S.C. et al. Planned versus unplanned delivery for placenta accreta spectrum. Am. J. Perinatol. 2022; 39(3): 252-8. https://dx.doi.org/10.1055/s-0040-1714676
- Liang Y., Zhang L., Bi S., Chen J., Zeng S., Huang L. et al. Risk factors and pregnancy outcome in women with a history of cesarean section complicated by placenta accreta. Matern. Fetal Med. 2022; 4(3): 179-85. https://dx.doi.org/10.1097/fm9.0000000000000142
- Huang Y.C., Yang C.C. Impact of planned versus emergency cesarean delivery on neonatal outcomes in pregnancies complicated by abnormal placentation: a systematic review and meta-analysis. Medicine (Baltimore). 2023; 102(32): e34498. https://dx.doi.org/10.1097/MD.0000000000034498
Received 25.09.2025
Accepted 09.12.2025
About the Authors
Alena V. Kayumova, PhD, Associate Professor, Deputy Director in Obstetrics and Gynecology, Ural Research Institute of Maternity and Child Care,Ministry of Health of Russia, 620028, Russia, Yekaterinburg, Repin str., 1, +7(982)62-32-047, kaum-doc@mail.ru, https://orcid.org/0000-0003-2685-4285
Yuri A. Semenov, Dr. Med. Sci., Associate Professor, Director, Ural Research Institute of Maternity and Child Care, Ministry of Health of Russia,
620028, Russia, Yekaterinburg, Repin str., 1, U-sirius@mail.ru, https://orcid.org/0000-0002-3268-7981
Oksana A. Melkozerova, Dr. Med. Sci., Deputy Director for Science, Ural Research Institute of Maternity and Child Care, Ministry of Health of Russia,
620028, Russia, Yekaterinburg, Repin str., 1, abolmed1@mail.ru, https://orcid.org/0000-0002-4090-0578
Svetlana V. Kinzhalova, Dr. Med. Sci., Associate Professor, Head of the Department of Intensive Care and Reanimation, Ural Research Institute of Maternity and Child Care, Ministry of Health of Russia, 620028, Russia, Yekaterinburg, Repin str., 1, sveking@mail.ru, https://orcid.org/0000-0003-2576-6742
Alexandra I. Gaeva, Deputy Director for Pediatrics, Ural Research Institute of Maternity and Child Care, Ministry of Health of Russia,
620028, Russia, Yekaterinburg, Repin str., 1, gaeva_al@mail.ru, https://orcid.org/0009-0003-5690-0593
Ksenia P. Shakirova, PhD, Head of the Neonatal Intensive Care Department, Ural Research Institute of Maternity and Child Care, Ministry of Health of Russia,
620028, Russia, Yekaterinburg, Repin str., 1, ksushamova@gmail.com, https://orcid.org/0000-0001-8183-0089
Roman A. Makarov, PhD, Senior Researcher, Department of Intensive Care and Reanimation, Ural Research Institute of Maternity and Child Care,
Ministry of Health of Russia, 620028, Russia, Yekaterinburg, Repin str., 1, r_makarov_ekb@mail.ru, https://orcid.org/0000-0002-8067-5643;
Corresponding author: Alena V. Kayumova, kaum-doc@mail.ru



