Clitoral reduction through resection of the corpora cavernosa with preservation of dorsal and ventral innervation
Makiyan Z.N.
Relevance: Over 90% of newborns with disorders of sex differentiation are assigned female at birth and require feminizing genitoplasty to correct external genitalia. Feminizing plastic surgery of the external genitalia is indicated in cases of pronounced virilization in adulthood. While clinical guidelines address the management of intersex conditions, surgical correction techniques have not been standardized in gynecological practices.
Objective: To optimize the surgical management of clitoromegaly in various forms of female virilization.
Materials and methods: Five patient groups were categorized based on their genome, gonadal morphology, and phenotype (including individuals with a male karyotype) who exhibited similar virilization features on the Prader scale. Surgical correction was performed in 33 female patients presenting with marked virilization of the external genitalia (Prader stages III–V).
Results: Clitoral reduction was achieved by resecting the corpora cavernosa while preserving the dorsal and ventral neurovascular bundles, thereby reducing clitoral size while maintaining erogenous sensitivity. During surgical treatment, the author studied the anatomical features of the genital tubercle, examined embryonic micro-preparations from the Carnegie collection, and compared the findings with current anatomical concepts.
Conclusion: Clitoral reduction via resection of the corpora cavernosa, with preservation of dorsal and ventral innervation, allowed maintenance of erogenous sensitivity. Postoperative evaluations demonstrated satisfactory cosmetic outcomes, a feminine appearance of the genitalia, and preservation of erogenous sensitivity in the clitoris, vaginal vestibule, and labia minora.
Conflicts of interest: The author has no conflicts of interest to declare.
Funding: There was no funding for this study.
Patient Consent for Publication: All patients provided informed consent for the publication of their data.
Author Data Sharing Statement: The data supporting the findings of this study are available upon request from the author.
For citation: Makiyan Z.N. Clitoral reduction through resection of the corpora cavernosa
with preservation of dorsal and ventral innervation.
Akusherstvo i Ginekologiya/Obstetrics and Gynecology. 2026; (2): 86-91 (in Russian)
https://dx.doi.org/10.18565/aig.2025.201
Keywords
Ambivalence or dimorphism?
The genital tubercle is an embryonic organ from which the penis and clitoris develop during sexual differentiation.
Organogenesis of the external genitalia occurs between the 3rd and 12th weeks of embryonic development. Both sexes derive their external genitalia from the genital tubercle, genital ridges, and genital folds [1–5].
In male fetuses, the SRY gene on the Y chromosome produces a factor that initiates the development of the testes (testes-determining factor) and regulates the virilization process. Testosterone, secreted by Leydig cells in the testes, stimulates the transformation of mesonephric ducts into seminiferous tubules and epididymis. Dihydrotestosterone further promotes the development of male sex organs (virilization), including the penis, scrotum, and the prostate gland. The genital ridges (labioscrotal folds) develop into the scrotum, while the genital tubercle elongates to form the penis (phallus), with the shaft of the urethra arising from the fused genital folds, culminating in the head of the penis. The prostate gland is also formed during this process [1–5].
In the absence or inactivation of androgens, the fetus remains in an indifferent developmental stage and becomes phenotypically female. Estrogens stimulate the development of female external genitalia. The genital tubercle transforms into the clitoris, genital ridges develop into the labia majora, and genital folds become the labia minora. The genital tubercle elongates slightly to form a clitoris, which is larger than that in male fetuses during the early stages of development. The urethral groove remains open, forming the vestibule of the vagina [1–6].
Abnormal androgen action during embryogenesis can result in female fetuses exhibiting pronounced signs of virilization (gender differentiation disorder). Insufficient secretion or inactivation of androgens in male fetuses can lead to the development of a female phenotype, resulting in the child being assigned a female sex at birth [7–16].
Sex formation disorders occur in 0.018% of newborn girls (1.8 per 10,000 live births), whereas the frequency of sex differentiation disorders in individuals with a 46,XY karyotype is estimated to be 1 per 20,000 live-born boys. More than 90% of patients with sex differentiation disorders, both those with a 46,XX karyotype (congenital adrenal hyperplasia) and those with a 46,XY karyotype (androgen insensitivity syndrome, testicular dysgenesis), are assigned female sex at birth. Consequently, they often require feminizing surgery [7–18].
Feminizing surgery of the external genitalia is performed in cases of pronounced virilization (Prader grade III–V), often alongside introitoplasty (creation of a vaginal opening) in the presence of a urogenital sinus [9–18].
While clinical guidelines address issues related to the treatment of intersex disorders, relevant surgical correction methods have not been standardized for gynecological practice [9–14].
This study aimed to optimize the surgical treatment of clitoromegaly in various cases of female virilization.
Materials and methods
Between 2007 and 2025, 135 patients aged 18–45 years with various types of sex differentiation disorders underwent surgery at the Department of Operative Gynecology of the V.I. Kulakov NMRC for OG&P, Ministry of Health of the Russian Federation [18–20]. Among these, 33 patients exhibited pronounced virilization of the external genitalia and clitoromegaly, prompting clitoral reduction. Patients with a 46,XY karyotype underwent gonadectomy.
Comprehensive clinical and laboratory examinations were conducted following the systematization of sex differentiation disorders. This included the determination of karyotype, gonadal morphology, and degree of virilization of the external genitalia [20].
Results
The degree of virilization of the external genitalia was assessed according to the Prader classification stages I–V [14, 15].
Prader stages I–II indicate mild virilization, characterized by clitoral enlargement with normal vaginal opening development. Surgical intervention was not required.
Prader stages III–V reflect indeterminate or intersexual genital development in patients classified as female, accompanied by significant virilization.
In Prader stage III, clitoromegaly is moderately pronounced, with a clitoral length ranging from 15 to 30 mm. The vaginal opening may be normal or narrowed due to the fusion of the labia minora.
In Prader stage IV, clitoromegaly is pronounced, with a clitoral length between 30 and 50 mm. The vaginal entrance may be narrowed due to labial fusion, potentially forming a urogenital sinus with the external opening positioned high in the vestibule of the vagina.
In Prader stage V, clitoromegaly is most severe, with a clitoral length exceeding 50 mm and a clitoral glans measuring more than 10 mm in diameter. The clitoris resembles the structure of the male penis. The vaginal entrance is closed due to the complete fusion of the labia minora, resulting in a stem urethra with the external urethral opening located at the clitoral head.
Feminizing plastic surgery of the external genitalia included clitoral reduction in 33 patients and introitoplasty in 17. Clitoral reduction was performed in 33 patients with pronounced clitoromegaly (Prader virilization grades III–V) [9–16, 20]. In cases of mild-to-moderate virilization (Prader grades I–II), feminizing plastic surgery was deemed unnecessary, and no surgical treatment was performed.
For patients exhibiting pronounced virilization (Prader grades III–IV), feminizing plastic surgery included clitoral reduction in 33 patients and creation of a vaginal opening (introitoplasty) in 17.
Severe clitoromegaly has been identified in the following types of sex differentiation disorders:
- congenital adrenal hyperplasia (karyotype 46,XX) – 17 patients;
- 46,XY testicular feminization syndrome: 7 patients
- 46,XY gonadal dysgenesis: 5 patients
- 45,X/46XY Turner syndrome: 2 patients
- 46,XX/46,XY ovotesticular sex differentiation disorder: 1 patient.
Before performing feminizing surgery on patients with a 46,XX karyotype and congenital adrenal hyperplasia (12 patients with the salt-wasting form and 5 with the virilizing form), hormone therapy was selected and adjusted.
All patients with sex differentiation disorders and a 46,XY karyotype classified as female (including those with testicular feminization syndrome, 46,XY gonadal dysgenesis, and ovotesticular sex differentiation disorder) underwent laparoscopy and bilateral adnexectomy to prevent the development of malignant neoplasms in the gonads [8–13, 20].
Several important nuances should be considered. The clitoris is innervated by sensory nerves that traverse the dorsal clitoral and genital nerves originating from the S2–S5 segments. Surgical removal of the clitoral head and cavernous bodies may damage innervation, resulting in a complete loss of erogenous sensitivity.
Clitoral reduction involves the resection of cavernous bodies while preserving the dorsal and ventral innervation. The stages of operation are as follows:
A semilunar incision was made around the corona of the clitoral glans, with an indentation of 5–8 mm (Fig. 1a). The entire shaft (body) of the clitoris was exposed up to the pubic symphysis (Fig. 1b). Longitudinal-lateral incisions were made in Buck's fascia and the tunica albuginea on both sides of the clitoral shaft. The cavernous bodies were mobilized and sharply separated, starting from the clitoral glans to the bifurcation of the clitoral legs (Fig. 1c), and cut off directly above the pubic bone (Fig. 1d). The isolated dorsal and ventral neurovascular bundles were mobilized and preserved while maintaining their connection to the clitoral glans. The clitoral head, which is attached to the dorsal neurovascular bundle, is sutured to the stump of the resected cavernous bodies. The skin incision of the prepuce was closed with fine sutures above the clitoral shaft (Fig. 1e). The final appearance of the external genitalia after surgery is shown in Figure 1e.
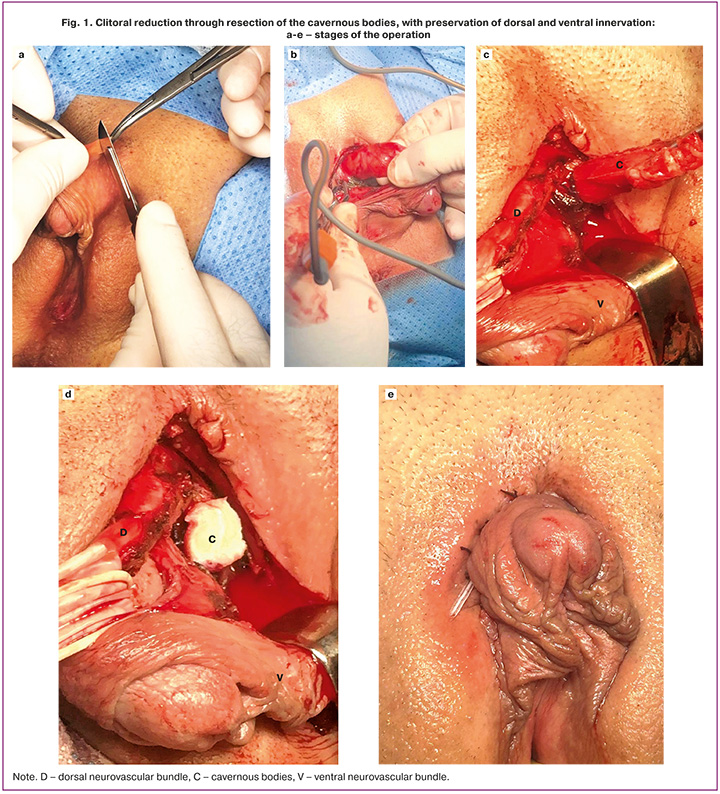
Postoperative examination revealed satisfactory cosmetic results, a feminine appearance of the genitals, and preservation of erogenous sensitivity in the clitoris, vaginal vestibule, and labia minora [21].
Discussion
This article addresses the surgical treatment of a relatively rare congenital pathology characterized by virilization, specifically clitoromegaly, in female patients. Current theories regarding the origin of the genital tubercle from the mesenchyme fail to account for its complex anatomical structure, innervation, and blood supply, as well as the mechanisms of virilization under androgen influence. These factors are crucial for the diagnosis, differentiation, and execution of clitoral reduction in cases of CAH. The study identified five distinct groups differing in genome, gonadal morphology, and patient phenotype (including those with a male karyotype), all exhibiting similar virilization manifestations on the Prader scale. During surgical treatment, the author examined the anatomical features of the genital tubercle and micro-preparations of embryos from the Carnegie collection, presenting a comparative analysis with the contemporary understanding.
Clitoral reduction via resection of the cavernous bodies while preserving dorsal and ventral innervation allowed the maintenance of erogenous sensitivity. Survey results indicate that most patients are satisfied with the appearance of their genitals, although they report a lack of pronounced clitoral erection during intimate contact.
What constitutes the genital tubercle, and how does androgen exposure affect it? How does hormonal influence contribute to the growth and enlargement of the genital tubercle? These questions remain unanswered.
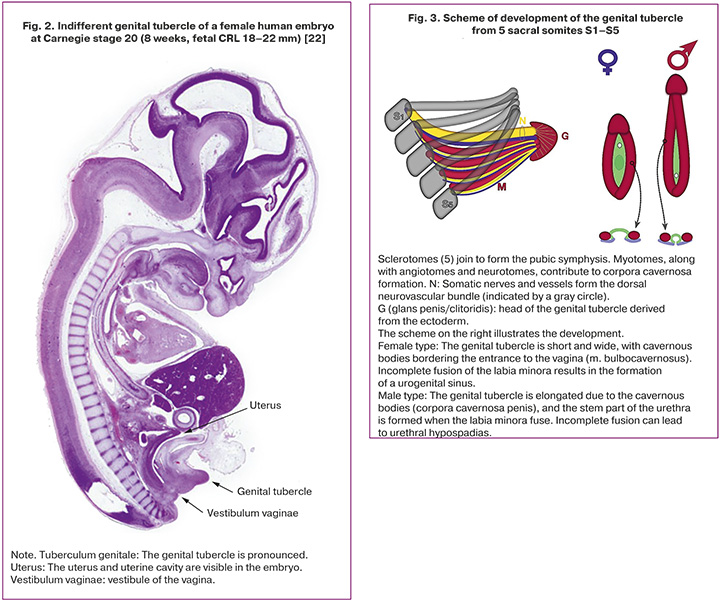
Figure 2 illustrates a microscopic examination of a female embryo (abortus) in the sagittal plane at 8 weeks of gestation (Carnegie stage 20). The image depicts a genital tubercle from which the clitoris develops, positioned above the entrance to the vaginal vestibule. A similar male embryo at Carnegie stage 21, corresponding to 9 weeks of gestation, showed an already formed penis [22].
Current understanding suggests that the genital tubercle arises from mesenchymal tissue; however, mesenchymal cells are dispersed throughout the embryo and lack specific androgen receptors [23–26].
By the end of the fifth week, the human embryo exhibits a segmental structure consisting of 42–44 pairs of somites: 4 occipital, 8 cervical, 12 thoracic, 5 lumbar, 5 sacral, and 8–10 coccygeal pairs. Each somite comprises five components: myotome (muscle component), sclerotome (cartilage and bone component), and dermatome (which forms the skin). Each myotome and dermatome retains its own innervation and vascularization from its original segments, the neurotome and angiotome, respectively [1–5].
The spine and ribs maintain their segmental structure, developing from the sclerotic compartments of the somites and connecting to the ventral part of the body to form the body cavity. Each segment includes five components: a vertebra linked to a rib (sclerotome), intercostal muscles (myotome), its own nerve (neurotome), a vessel (angiotome), and the skin (dermatome). The first occipital and last 5–7 coccygeal somites later disappear, while the remaining somites form the axial skeleton [1].
According to a new concept, the genital tubercle develops from five sacral (S1–S5) somites (Fig. 3): five sclerotomes fuse to create the pelvic bones and pubic symphysis, while five fused myotomes, along with their innervating neurotomes and angiotomes, extend along the pubic symphysis, forming the cavernous bodies of the genital tubercle. The head of the genital tubercle is derived from the ectoderm, with somatic innervation and blood supply from the dorsal neurovascular bundle [19].
Confirmation of the development of the genital tubercle from the sacral somites is evident in the preservation of innervation from the S1–S5 segments.
A new theory posits that the growth of the genital tubercle follows the same mechanisms as that of limb development. However, the genitogenic somites fuse along the midline above the pubis, similar to rib arches, marking a key difference between the two.
It is likely that the initiation of genital tubercle development at different stages relies on growth factors involved in limb development. Androgens appear to stimulate the masculinization of the indifferent genital tubercle through cavernous body hypertrophy and inductive effects on neurotomes, activating nerve growth factors.
The paradox lies in the fact that sexual differentiation of the external genitalia is the final stage of embryogenesis, even though the indifferent sexual tubercle, formed from five sacral somites, begins to develop at the earliest stages, prior to the formation of the gonads and internal genital organs.
Conclusion
Clitoral reduction through resection of the cavernous bodies while preserving dorsal and ventral innervation succeeded in maintaining erogenous sensitivity. Postoperative evaluation demonstrated satisfactory cosmetic results, a feminine appearance of the genitals, and preservation of erogenous sensitivity in the clitoris, vestibule, and the labia minora.
References
- Hill M.A. Embryology BGD Lecture - Sexual Differentiation. 2025. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/BGD_Lecture_-_Sexual_Differentiation
- Jost A. A new look at the mechanisms controlling sex differentiation in mammals. Johns Hopkins Med. J. 1972; 130(1): 38-53.
- Wilhelm D., Palmer S., Koopman P. Sex determination and gonadal development in mammals. Physiol. Rev. 2007; 87(1): 1-28. https://dx.doi.org/10.1152/physrev.00009.2006
- Cohn M.J. Development of the external genitalia: conserved and divergent mechanisms of appendage patterning. Dev. Dyn. 2011; 240(5): 1108-15. https://dx.doi.org/10.1002/dvdy.22631
- Munger S.C., Natarajan A., Looger L.L., Ohler U., Capel B. Fine time course expression analysis identifies cascades of activation and repression and maps a putative regulator of mammalian sex determination. PLoS Genet. 2013; 9(7): e1003630. https://dx.doi.org/10.1371/journal.pgen.1003630
- Mieszczak J., Houk C.P., Lee P.A. Assignment of the sex of rearing in the neonate with a disorder of sex development. Curr. Opin. Pediatr. 2009; 21(4): 541-7. https://dx.doi.org/10.1097/MOP.0b013e32832c6d2c
- Sax L. How common is intersex?: a response to Anne Fausto-Sterling. J. Sex Res. 2002; 39(3): 174-8. https://dx.doi.org/10.1080/00224490209552139
- Hughes I.A., Houk C., Ahmed S.F., Lee P.A. Consensus statement on management of intersex disorders. Arch. Dis. Child. 2006; 91(7): 554-63. https://dx.doi.org/10.1136/adc.2006.098319
- Cools M., Nordenström A., Robeva R., Hall J., Westerveld P., Flück C. et al.; COST Action BM1303 working group 1. Caring for individuals with a difference of sex development (DSD): a Consensus Statement. Nat. Rev. Endocrinol. 2018; 14(7): 415-29. https://dx.doi.org/10.1038/s41574-018-0010-8
- Pasterski V., Prentice P., Hughes I.A. Consequences of the Chicago consensus on disorders of sex development (DSD): current practices in Europe. Arch. Dis. Child. 2010; 95(8): 618-23. https://dx.doi.org/10.1136/adc.2009.163840
- Houk C.P., Lee P.A. Update on disorders of sex development. Curr. Opin. Endocrinol. Diabetes Obes. 2012; 19(1): 28-32. https://dx.doi.org/10.1097/MED.0b013e32834edacb
- Ahmed S.F., Achermann J.C., Arlt W., Balen A.H., Conway G., Edwards Z.L. et al. UK guidance on the initial evaluation of an infant or an adolescent with a suspected disorder of sex development. Clin. Endocrinol. (Oxf). 2011; 75(1): 12-26. https://dx.doi.org/10.1111/j.1365-2265.2011.04076.x
- Ahmed S.F., Armstrong K., Cheng E.Y., Cools M., Harley V., Mendonca B.B. et al. Differences of sex development. Nat. Rev. Dis. Primers. 2025; 11(1): 54. https://dx.doi.org/10.1038/s41572-025-00637-y
- Prader A. [Genital findings in the female pseudo-hermaphroditism of the congenital adrenogenital syndrome; morphology, frequency, development and heredity of the different genital forms]. Helv Pediatr Acta.: 1954, 9(3); 231-48. [Article in German].
- Prader A., Gurtner H.P. [The syndrome of male pseudohermaphrodism in congenital adrenocortical hyperplasia without overproduction of androgens (adrenal male pseudohermaphrodism)]. Helv. Paediatr. Acta. 1955; 10(4): 397-412. [Article in German].
- Creighton S.M. Long-term outcome of feminization surgery: the London experience. BJU Int. 2004; 93(S. 3): 44-6. https://dx.doi.org/10.1111/j.1464-410X.2004.04708.x
- Sugiyama Y., Mizuno H., Hayashi Y., Imamine H., Ito T., Kato I. et al. Severity of virilization of external genitalia in Japanese patients with salt-wasting 21-hydroxylase deficiency. Tohoku J. Exp. Med. 2008; 215(4): 341-8. https://dx.doi.org/10.1620/tjem.215.341
- Makiyan Z. Studies of gonadal sex differentiation. Organogenesis. 2016; 12(1): 42-51. https://dx.doi.org/10.1080/15476278.2016.1145318
- Makiyan Z. Systematization of ambiguous genitalia. Organogenesis. 2016; 12(4): 169-82. https://dx.doi.org/10.1080/15476278.2016.1210749
- Makiyan Z. Systematization for female genital anatomic variations. Clin. Anat. 2021; 34(3): 420-30. https://dx.doi.org/10.1002/ca.23668
- Makiyan Z. Female sexuality. Lulu Publishing (USA). 2013. Available at: https://www.lulu.com/shop/zohrab-makiyan/female-sexuality/ebook/product-21025371.html
- Hill M.A. Embryology. Carnegie Stage 20. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Carnegie_stage_20#/media/File:K15818_Stage_20_sagittal_02.jpg
- Lin C., Yin Y., Veith G.M. Fisher A.V., Long F., Ma L. Temporal and spatial dissection of Shh signaling in genital tubercle development. Development. 2009; 136(23); 3959-67. https://dx.doi.org/10.1242/dev.039768
- Yang J.H., Menshenina J., Cunha G.R. Place N., Baskin L.S. Morphology of mouse external genitalia: implications for a role of estrogen in sexual dimorphism of the mouse genital tubercle. J. Urol. 2010; 184(4S): 1604-9. https://dx.doi.org/10.1016/j.juro.2010.03.079
- Tschopp P., Sherratt E., Sanger T.J., Groner A.C., Aspiras A.C., Hu J.K. et al. A relative shift in cloacal location repositions external genitalia in amniote evolution. Nature. 2014; 516(7531): 391-4. https://dx.doi.org/10.1038/nature13819
- Herrera A.M., Cohn M.J. Embryonic origin and compartmental organization of the external genitalia. Sci. Rep. 2014; 4: 6896 https://dx.doi.org/10.1038/srep06896
Received 23.07.2025
Accepted 26.11.2025



