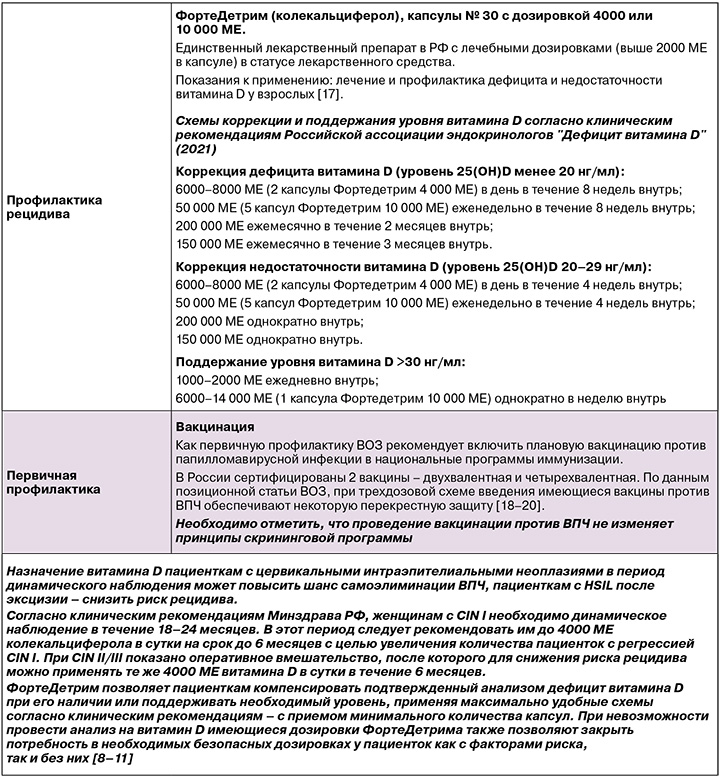
Применение витамина D у пациенток с CIN I/II/III в период динамического наблюдения

Dovletkhanova E.R., Abakarova P.R., Mgeryan A.N., Mezhevitinova E.A., Prilepskaya V.N.
Применение витамина D у пациенток с CIN I/II/III в период динамического наблюдения






1. WHO Guidelines for Screening and Treatment of Precancerous Lesions for Cervical Cancer Prevention. Geneva: World Health Organization; 2013.
2. zur Hausen H. Papillomaviruses in the causation of human cancers – a brief historical account. Virology. 2009; 384(2): 260‑5. https://dx.doi.org/ 10.1016/ j.virol.2008.11.046.
3. Elit L., Levine M.N., Julian J.A., Sellors J.W., Lytwyn A., Chong S. et al. Expectant management versus immediate treatment for low‑grade cervical Intraepithelial neoplasia: randomized trial in Canada and Brazil. Cancer. 2011; 117(7): 1438‑45. https://dx.doi.org/10.1002/cncr.25635.
4. Прилепская В.Н., Сухих Г.Т., ред. Диагностика, лечение и профилактика цервикальных неоплазий. М.: МЕДпресс‑информ; 2020. 80с. [Prilepskaya V.N., Sukhikh G.T., ed. Diagnosis, treatment and prevention of cervical neoplasia. Moscow: MEDpress‑inform; 2020. 80p. (in Russian)].
5. Asiaf A., Ahmad S.T., Mohammad S.O., Zargar M.A. Review of the current knowledge on the epidemiology, pathogenesis, and prevention of human papillomavirus infection. Eur. J. Cancer. Prev. 2014; 23(3): 206‑24. https://dx.doi.org/10.1097/CEJ.0b013e328364f273.
6. Yenigul N.N., Yılmaz F.Y., Ayhan I. Can Serum Vitamin B12 and Folate Levels Predict HPV Penetration in Patients with ASCUS? Nutr. Cancer. 2021; 73(4): 602‑8. https://dx.doi.org/10.1080/01635581.2020.1807030.
7. Громова О.А., Торшин И.Ю., Фролова Д.Е., Лапочкина Н.П., Лиманова О.А. О противовирусных эффектах витамина D. Медицинский Совет. 2020; (3): 152‑8. [Gromova O.A., Torshin I.Yu., Frolova D.E., Lapochkina N.P., Limanova O.A. About antiviral effects of vitamin D. Medical Council. 2020; (3): 152‑8. (in Russian)]. https://doi.org/10.21518/2079‑701X‑2020‑3‑152‑158.
8. Vahedpoor Z., Jamilian M., Bahmani F., Aghadavod E., Karamali M., Kashanian M., Asemi Z. Effects of Long‑Term Vitamin D Supplementation on Regression and Metabolic Status of Cervical Intraepithelial Neoplasia: a Randomized, Double‑Blind, Placebo‑Controlled Trial. Horm. Cancer. 2017; 8(1): 58‑67. https://dx.doi.org/10.1007/s12672‑016‑0278‑x.
9. Vahedpoor Z., Mahmoodi S., Samimi M., Gilasi H.R., Bahmani F., Soltani A. et al. Long‑term vitamin D supplementation and the effects on recurrence and metabolic status of cervical intraepithelial neoplasia grade 2 or 3: A randomized, double‑blind, placebo‑controlled trial. Ann. Nutr. Metab. 2018; 72(2): 151‑60. https://dx.doi.org/10.1159/000487270.
10. Зароченцева Н.В., Джиджихия Л.К. Влияние приема витамина D у пациенток с ВПЧ‑ассоциированными заболеваниями шейки матки. Вопросы практической кольпоскопии. Генитальные инфекции. 2022; (4): 17‑23. [Zarochentseva N.V., Dzhidzhikhiya L.K. The effect of vitamin D in patients with HPV‑associated cervical diseases. Issues of Practical Colposcopy & Genital Infections. 2022; (4): 17‑23. (in Russian)]. https://dx.doi.org/10.46393/ 27826392_2022_4_17.
11. Li D., Liu Y., Kong D., Papukashvili D., Rcheulishvili N., Zhao H. et al. Vitamin D Receptor Gene Polymorphisms and the Risk of CIN2+ in Shanxi Population. Biomed Res. Int. 2022; 2022: 6875996. https://dx.doi.org/10.1155/2022/6875996.
12. Клинические рекомендации. Цервикальная интраэпителиальная неоплазия, эрозия и эктропион шейки матки. 2020–2021–2022 (31.05.2021). Утверждены Минздравом РФ. [Clinical guidelines. Cervical intraepithelial neoplasia, cervical erosion and ectropion. 2020‑2021‑2022 (31.05.2021). Approved by the Ministry of Health of the Russian Federation. (in Russian)].
13. Castle P.E., Gage J.C., Wheeler C.M., Schiffman M. The clinical meaning of a cervical intraepithelial neoplasia grade 1 biopsy. Obstet. Gynecol. 2011; 118(6): 1222‑9. https://dx.doi.org/10.1097/AOG.0b013e318237caf4.
14. Massad L.S., Einstein M.H., Huh W.K., Katki H.A., Kinney W.K., Schiffman M., Solomon D., Wentzensen N., Lawson H.W.; 2012 ASCCP Consensus Guidelines Conference. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. J. Low Genit. Tract Dis. 2013; 17(5 Suppl 1): S1‑S27. https://dx.doi.org/10.1097/LGT.0b013e318287d329.
15. Cox J.T., Schiffman M., Solomon D.; ASCUS‑LSIL Triage Study (ALTS) Group. Prospective follow‑up suggests similar risk of subsequent cervical intraepithelial neoplasia grade 2 or 3 among women with cervical intraepithelial neoplasia grade 1 or negative colposcopy and directed biopsy. Am. J. Obstet. Gynecol. 2003; 188(6): 1406‑12. https://dx.doi.org/10.1067/mob.2003.461.
16. Дедов И.И., Мельниченко Г.А., Мокрышева Н.Г., Пигарова Е.А., Поваляева А.А., Рожинская Л.Я., Белая Ж.Е., Дзеранова Л.К., Каронова Т.Л., Суплотова Л.А., Трошина Е.А. Проект федеральных клинических рекомендаций по диагностике, лечению и профилактике дефицита витамина D. Остеопороз и остеопатии. 2021; 24(4): 4‑26. [Dedov I.I., Mel’nichenko G.A., Mokrysheva N.G., Pigarova E.A., Povaliaeva A.A., Rozhinskaya L.Ya. et al. Draft Federal Clinical Practice Guidelines for the diagnosis, treatment, and prevention of vitamin D deficiency. Osteoporosis and Bone Diseases. 2021; 24(4): 4‑26. (in Russian)]. https://doi.org/10.14341/osteo12937.
17. Инструкция по медицинскому применению препарата ФортеДетрим. [Instructions for medical use of the drug ForteDetrim. (in Russian)].
18. WHO. Vaccines in national immunization programme update. 10 October 2019. https://www.who.int/immunization/monitoring_surveillance/en/
19. Баранов А.А., Намазова-Баранова Л.С., Таточенко В.К., Вишнёва Е.А., Федосеенко М.В., Селимзянова Л.Р., Чемакина Д.С., Лобзин Ю.В., Харит С.М., Брико Н.И., Лопухов П.Д., Сухих Г.Т., Уварова Е.В., Прилепская В.Н., Поляков В.Г., Гомберг М.А., Краснопольский В.И., Зароченцева Н.В., Костинов М.П., Белоцерковцева Л.Д., Мельникова А.А., Батыршина Л.Р. Вакцинопрофилактика заболеваний, вызванных вирусом папилломы человека: позиции доказательной медицины. Обзор клинических рекомендаций. Вопросы современной педиатрии. 2017; 16(2): 107‑17. [Baranov A.А., Namazova‑Baranova L.S., Tatochenko V.K., Vishneva E.А., Fedoseenko M.V., Selimzianova L.R., Chemakina D.S., Lobzin Yu.V., Kharit S.М., Briko N.I., Lopukhov P.D., Sukhikh G.Т., Uvarova E.V., Prilepskaya V.N., Polyakov V.G., Gomberg M.А., Krasnopolskiy V.I., Zarochentseva N.V., Kostinov M.P., Belotserkovtseva L.D., Melnikova A.А., Batyrshina L.R. Vaccinal Prevention of the Diseases Caused by Human Papillomavirus: Evidence‑Based Medicine. Review of Clinical Guidelines. Current Pediatrics. 2017; 16(2): 107‑17. (in Russian)]. https://dx.doi.org/10.15690/ vsp.v16i2.1711.
20. Вакцины против вируса папилломы человека: документ по позиции ВОЗ – май 2017. [Human papillomavirus vaccines: WHO position paper ‑ May 2017]. https://www.who.int/immunization/position_papers/position_paper_process.pdf